Chapter 1 Basic Concepts 1 A gas in

Chapter 1 Basic Concepts 1. A gas in a piston-cylinder device is compressed, and as a result its temperature rises. Is this a heat or work interaction? 2. On a P-v diagram, what does the area under the process curve represent? 3. Determine the energy required to accelerate a 2000 -kg car from 20 to 70 km/h on an uphill road with a vertical rise of 40 m.

4. Consider a device with one inlet and one exit. If the volume flow rates at the inlet and at the exit are the same, is the flow through the device necessarily steady? Why? 5. A 2 m 3 rigid tank contains nitrogen gas at 500 k. Pa and 300 K. Now heat is transferred to the nitrogen in the tank and the pressure of nitrogen rises to 800 k. Pa. The work done during this process is (A) 600 k. J (B) 1000 k. J (C) 0 k. J (D) 500 k. J (E) 1600 k. J

6. A 2 m 3 rigid tank contains nitrogen gas at 500 k. Pa and 300 K. Now heat is transferred to the nitrogen in the tank and the pressure of nitrogen rises to 800 k. Pa. The work done during this process is (A) 600 k. J (D) 500 k. J (B) 1000 k. J (E) 1600 k. J (C) 0 k. J

CHAPTER 2 PROPERTIES OF PURE SUBSTANCES 2. 1 A rigid tank contains 5 kg of an ideal gas at 4 atm and 40℃. Now a valve is opened, and half of mass of the gas is allowed to escape. If the final pressure in the tank is 1. 5 atm. the final temperature in the tank is [a] -38 ℃ [e]144 ℃ [b]30 ℃ [c]40 ℃ [d] 53 ℃ 2. 2 A rigid vessel contains steam at 20 MPa and 400 ℃. The mass of the steam is [a] 5. 0 kg [e]680 kg [b] 0. 322 kg [c] 322 kg [d] 503 kg

2. 3 A rigid tank is filled with saturated liquid-vapor mixture of water at 200 k. Pa. If 20 percent of the mass is liquid and the 80 percent of the mass is vapor , the total mass in the tank is [a] 705 kg [b] 500 kg [c]258 kg [d] 635 kg [e]2809

Chapter 3 The first Law of Thermodynamics o A reversible cycle plots as a perfect circle on a T-S diagram with maximum and minimum temperatures 600 K and 300 K and a maximum and minimum entropy of 600 k. J/K and 300 k. J/K.

a) What is the net work of this cycle? b) Indicate the path in the cycle along which heat is rejected. How do you know? (A sentence or two is required, perhaps bolstered by an equation). c) What is thermodynamic efficiency of this cycle?

o 2. The sketch below shows a perfectly insulated container with two compartments separated by a non-adiabatic, frictionless piston. Both compartments are at the same pressure (PA= PB) and contain an equal amount of the same gas ( m. A =m. B ). Initially compartment A is at a higher temperature than compartment B(TA>TB).
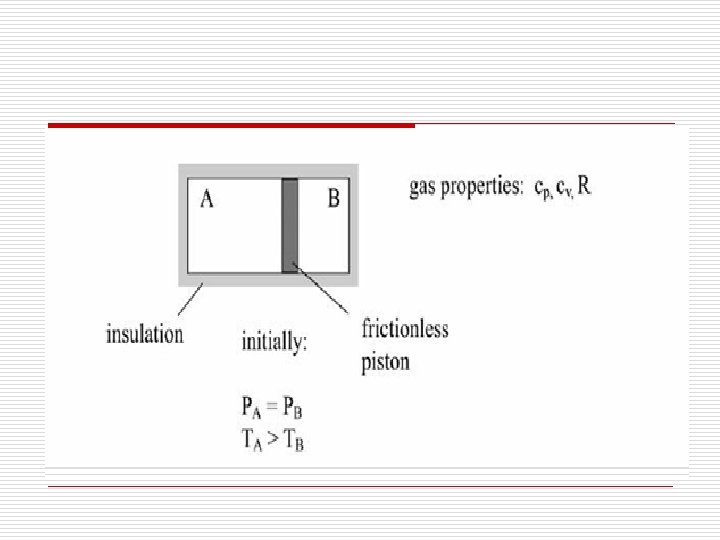

o a) Which way, if at all, does the piston move? Indicate by an arrow on a drawing in your exam book (A sentence or two is expected, perhaps bolstered by some equations). o b) Is the final internal energy Ufinal of the system (both compartments together) lower, equal or higher than initial internal energy Uinitial of the system? Why? (A sentence or two is expected, perhaps bolstered by some equations. )

o o c) What is the final temperature of the system Tfinal expressed in terms of TA and TB ? b) What is the change in entropy of compartment A? What is the change in entropy of compartment B? e) What is the change in entropy of the surroundings? f) Is this process reversible or irreversible? How do you know? (Note: you do not have to prove your answer mathematically to get full credit. Explain in a sentence how you know whether the process is reversible or irreversible)

o 10 kg of air contained in a piston cylinder apparatus at 5 bar, 300 K, expands isothermally until the pressure is 1 bar. Determine: o (i) the work done. o (ii) the internal energy change of the air. o (iii) the heat transfer. o Assume that air behaves as an ideal gas and pressure variation is gradual and continuous.

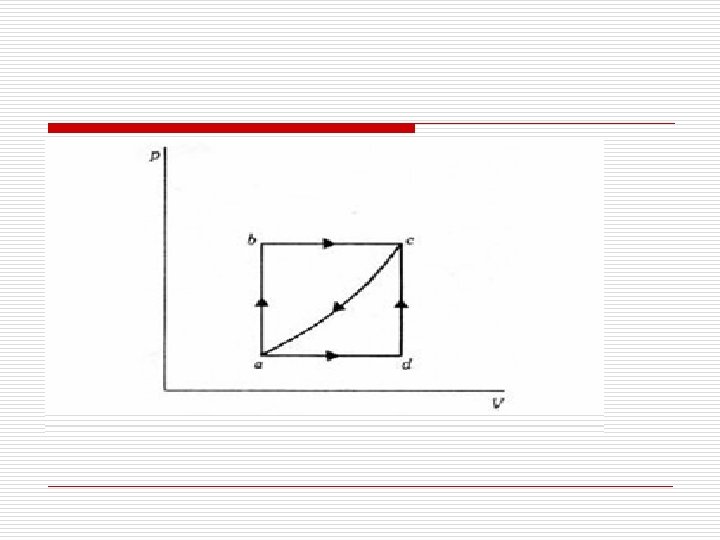
o A system is capable of executing a cyclic process as indicated in the P V sketch; it may be executed either clockwise abca or counterclockwise adca. (a) When going clockwise to state c, 80 KJ of heat flow from the system. Find the work along the path ca. (b) When going counterclockwise to state c, 70 KJ of heat flow from the system. Find the work during the process adc.

CHAPTER 5 THE SECOND LAW OF THERMODYNAMICS 5. 1 The drinking water needs of an office are met by cooling tab water in a refrigerated water fountain from 22℃ to 8 ℃ at an average rate of 8 kg/h. If the COP of this refrigerator is 3. 1, the required power input to this refrigerator is [a] 28 W [b] 42 W [c] 88 W [d] 130 W [e] 403 W

5. 2 A heat engine cycle is executed with steam in the saturation dome. The pressure of steam 1 MPa during heat addition, and 0. 5 MPa during heat rejection. The highest possible efficiency of this heat engine is [a] 6. 2% [b] 15. 6 % [c] 50. 0 % [d] 93. 8 % [e] 100 %

5. 3 A heat engine receives heat from a source at 100 ℃ and rejects the waste heat to a sink at 50 ℃. If heat is supplied to this engine at a rate of 100 kj/s, the maximum power this heat engine can produce is [a] 25. 4 k. W [c] 174. 6 k. W [e] 100. 0 k. W [b] 55. 4 k. W [d] 95. 0 k. W

5. 4 Steam is compressed from 8 MPa and 300 ℃ to 10 MPa isentropically. The final temperature of the steam is [a] 290 ℃ [b] 300 ℃ [c] 320 ℃ [d] 330 ℃ [e] 340 ℃

5. 5 Helium gas is compressed from 1 atm and 25 ℃ to a pressure of 10 atm adiabatically. The lowest temperature of helium after compression is [a] 25 ℃ [c] 250 ℃ [e] 476 ℃ [b] 63 ℃ [d] 384 ℃

5. 6 Steam expands in an adiabatic turbine from 10 MPa and 500 ℃ to 0. 1 MPa at a rate of 8 kg/s. If steam leaves the turbine as saturated vapor, the power output of the turbine is [a] 5586 kw [c] 2136 kw [e]26, 990 kw [b] 698 kw [d] 12, 452 kw

5. 7 A unit mass of a substance an irreversible process from state 1 to state 2 while gaining heat from the surroundings at temperature T in the amount of q. If the entropy of the substance is at state 1, and at state 2, the entropy change of the substance s during this process is [a] [b] [c] [d] [e]

5. 8 Air is compressed from room conditions to a specified pressure in a reversible manner by two compressor : one isothermal and the other adidbatic. If the entropy change of air s during the reversible adiabatic compression, the correct statement regarding entropy change of air per unit mass is [a] [b] [c] [d] [e]

5. 9 Heat is lost through a plane wall steadily at a rate of 600 W. If the inner and outer surface temperature of the wall are 20 ℃ and 5℃, respectively, the rate of entropy generation within the wall is [a] 0. 11 W/K [c] 2. 10 W/K [e] 90. 0 W/K [b] 4. 21 W/K [d] 142. 1 W/K

5. 10 Air at 15 ℃ is compressed steadily and isothermally from 100 KPa to 700 KPa at a rate of 0. 12 kg/s. The minimum power input to the compressor is [a] 1. 0 k. W [e] 16. 1 k. W [b] 11. 2 k. W [c] 25. 8 k. W [d] 19. 3 k. W

5. 11 Air is to be compressed steadily and isentropically from 1 atm to 16 atm by a two-stage compressor. To minimize the total compression work, the intermediate pressure between the two stages must be [a] 2 atm [c] 8 atm [e] 12 atm [b] 4 atm [d] 10 atm

5. 12 Liquid water enters an adiabatic piping system at 15 ℃ at a rate of 5 kg/s. If the water temperature rises by 0. 5 ℃ during flow due to friction. the rate of entropy generation in the pipe is [a] 36 W/K [c] 685 W/K [e] 8370 W/K [b] 29 W/K [d] 920 W/K

5. 13 Steam enters an adiabatic turbine at 4 MPa and 500 ℃ at a rate of 15 kg/s, and exits at 0. 2 MPa and 300 ℃. The rate of entropy generation in the turbine is [a] 0. 8 k. W/K [c] 12. 0 k. W/K [e] 17. 4 k. W/K [b] 1. 2 k. W/K [d] 15. 1 k. W/K

5. 13 o o A quantity of air undergoes a thermodynamic cycle consisting of three processes in series. Process 1 -2: constant-volume heating from P 1=0. 1 MPa, T 1=288 K, V 1=0. 02 to P 2=0. 42 MPa; o Process 2 -3: constant-pressure cooling ; o Process 3 -1: o isothermal heating to the initial state ; Employing the ideal gas model with Cp=1 k. J/kg. K, evaluate the change in entropy for each process. Sketch the cycle on p-v coordinates.

CHAPTER 8 THERMODYNAMICS OF HIGH-SPEED GAS FLOW 8. 1 In which medium will sound travel fastest for a given temperature: air, helium, or argon? 8. 2 Is it possible to accelerate a gas to a supersonic velocity in a converging nozzle? 8. 3 Air enters a converging-diverging nozzle at a pressure of 1. 2 MPa with negligible velocity. What is the lowest pressure that can be obtained at the throat of the nozzle?

8. 4 Air enters a nozzle at 0. 2 MPa, 350 K, and a velocity of 150 m/s. Assuming isentropic flow, determine the pressure and temperature of air at a location where the air velocity equals the velocity of sound. What is the ratio of the area at this location to the entrance area? 8. 5 Air enters a converging-diverging nozzle at 0. 8 MPa with negligible velocity. Assuming the flow to be isentropic, determine the back pressure that will result in an exit Mach number of 1. 8.

Air enters an adiabatic nozzle at 800 K and 140 KPa and emerges at 7 KPa. The isentropic efficiency of the nozzle is known to be 0. 96. Determine the discharge velocity. Sketch the process on a T-S diagram and label the states. You may assume the working fluid is a perfect gas with Cp=1 k. J/kg. K.

8. 6 Steam enters an converging nozzle at 30. MPa and 600 ℃ with a negligible velocity, and it exits at 1. 8 MPa. For a nozzle exit area of 24 c㎡, determine the exit. Velocity, mass flow rate, and exit Mach number if the nozzle (a) is isentropic and (b) has an efficiency of 90 percent

CHAPTER 9 GAS POWER CYCLES 9. 1 A Carnot cycle operates between the temperature limits of 300 K and 1500 K, and produces 600 kw of net power. The rate of entropy change of the working fluid during the heat addition process is [a] 0 [c] 0. 5 k. W/K [e] 5. 0 k. W/K [b] 0. 4 k. W/K [d] 2. 0 k. W/K

o a) A simple engine uses a perfect gas as the working fluid in a piston-cylinder system. The gas is first heated at constant pressure from state 1 to state 2, then cooled at constant volume to state 3 where T 3=T 1, and then cooled at constant temperature, thereby returning to state 1. Derive expressions for the amounts of energy transfer as work and heat (per kg of gas) for each process in terms of the temperatures and pressures at each state and the constants of the gas. Suppose T 1=300 K, P 1=0. 2 MPa, T 2=800 K, K=Cp/Cv=1. 4. Calculate the cycle efficiency (net work output/energy input as heat)

o b) When the engine of part (a) is reversed, it becomes a refrigeration device. Calculate the amount of energy transfer as heat from the cold space for this cycle (per kg of gas), and the cycle COP (energy transfer as heat from cold space/net work input). You can assume air for the working fluid.

CHAPTER 10 REFRIGERATION CYCLES 10. 1 A refrigerator removes heat from a refrigerated space at-5 ℃ at a rate of 0. 35 kj/s and rejects it to an environment at 20 ℃. The minimum requires power input is [a] 30 W [b] 33 W [c] 56 W [d] 124 W [e] 350 W

10. 2 An ideal vapor compression refrigeration cycle with R-134 a as the working fluid operates between the pressure limits of 140 k. Pa and 900 kpa. The mass fraction of the refrigerant that is in the liquid phase at the inlet of the evaporator is [a] 0. 75 [c] 0. 50 [e] 0. 25 [b] 0. 65 [d] 0. 35

10. 3 Consider a refrigeration that operates on the vapor compression refrigerant cycle with R-134 a as the working fluid. The refrigerant enters the compressor as saturated vapor at 140 k. Pa , and exits at 800 k. Pa and 60 ℃, and leaves the condenser as saturated liquid at 800 k. Ps. The coefficient of performance of this refrigerator is [a] 0. 41 [c] 1. 8 [e] 3. 4 [b] 0. 1 [d] 2. 5

o There are 528 k. J/min of heat removed from a body by a refrigerator operating between the limits of 244. 5 K and 305. 5 K. If its coefficient of performance is three fourths of that of a Carnot refrigerator working between the same temperature limits, find (a) the heat rejected and (b) the work input, kw. (c) What are the COP and the heat if this device is used to deliver heat ?
- Slides: 39