Chapter 1 1 Thermal Measurements Temperature and heat



















- Slides: 22

Chapter 1 1 Thermal Measurements Temperature and heat (thermal energy) are two different concepts. A spark that is a few hundred degrees may not burn you but a bowl of boiling hot water will certainly give you a nasty blistering burn. Why is this so?

Chapter 11: Thermal Measurements

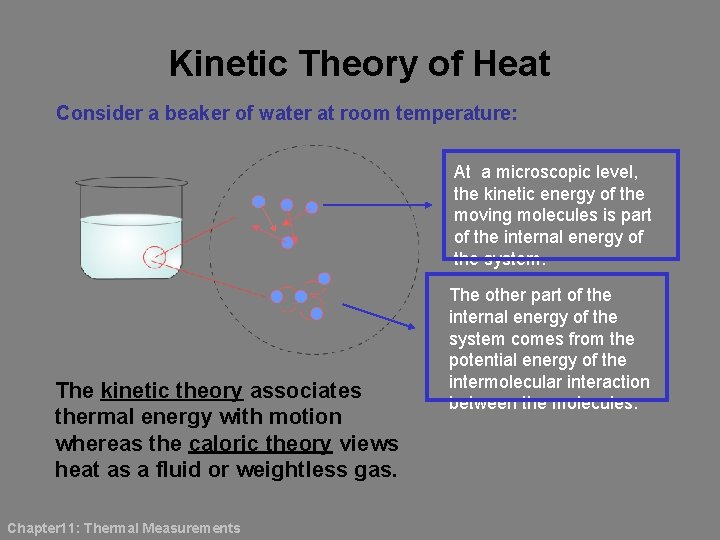
Kinetic Theory of Heat Consider a beaker of water at room temperature: At a microscopic level, the kinetic energy of the moving molecules is part of the internal energy of the system. The kinetic theory associates thermal energy with motion whereas the caloric theory views heat as a fluid or weightless gas. Chapter 11: Thermal Measurements The other part of the internal energy of the system comes from the potential energy of the intermolecular interaction between the molecules.

Caloric Theory of Heat was considered a fluid called caloric that flows from hotter to colder bodies. As heat was thought of as a material substance in caloric theory, it was assumed that it could neither be created nor destroyed. However, while investigating a cannon being bored, Count Rumford (1753 – 1814) observed that an endless supply of heat could be produced by friction. Therefore, heat cannot be a substance that is conserved. Chapter 11: Thermal Measurements

Chapter 11: Thermal Measurements

Sir James Joule (1818 – 1889) studied the relation between heat and mechanical work. He found that the rise in temperature is directly proportional to the amount of work done by the drop in the weights. The experiment showed that mechanical energy can be converted into thermal or heat energy. The unit for thermal energy is called the calorie (cal). One cal is the amount of thermal energy required to raise the temperature of 1 g of water by 1 °C. Chapter 11: Thermal Measurements

Chapter 11: Thermal Measurements

Q θ Heat capacity (C) of an object is defined as the amount of thermal energy required to raise the temperature of the object by 1 K or 1 °C. The SI unit for heat capacity is J K -1 or J °C-1 C=Q÷θ Q = Cθ where Q = thermal energy absorbed or released in J θ = change in temperature in K or °C θ Example: Q When a piece of iron is cooled from 70 °C to 40 °C, thermal energy given out is 690 J. What is the heat capacity of the piece of iron? C = 690 J ÷ (70 - 40) °C = 23 J °C-1 Chapter 11: Thermal Measurements

Q θ Specific heat capacity (c) of a substance is defined as the amount of thermal energy required to raise the temperature of 1 kg of the substance by 1 K or 1°C. The SI unit for specific heat capacity is J kg-1 K-1 or J kg-1 °C-1 c = C ÷ mass = Q ÷ mθ ⇒ Q = mcθ where C is the heat capacity Q is the amount of thermal energy θ is the temperature change Chapter 11: Thermal Measurements

Example 1. An electric kettle contains s an immersion heater of power 2. 0 k. W. It takes 5 minutes to heat 2. 0 kg of water from 30 °C to 100 °C. Assuming no thermal energy is lost to the surroundings, what is the specific heat capacity of water? Power P = 2 000 W Time t = 5 min = 300 s Thermal energy supplied by heater = P × t = 2 000 × 300 = 600 000 J Thermal energy absorbed by water = mcθ =2×c× (100 – 30) J = 140 c J Thermal energy absorbed by water = Thermal energy supplied by heater 140 c = 600 000 ÷ 140 = 4 286 J kg -1 °C-1 Chapter 11: Thermal Measurements

2. 0. 5 kg of lime juice at 30 °C is placed in the refrigerator which can remove heat at an average rate of 20 J s-1. How long will it take to cool the lime juice to 5 °C? (The specific heat capacity of lime juice is 4 200 J kg -1 °C-1) Thermal energy removed by refrigerator in time t s = ( 20 × t ) J Thermal energy lost by lime juice, Q = = = mcθ 0. 5 × 4 200 × (30 - 5) J 0. 5 × 4 200 × 25 J Thermal energy removed by refrigerator = Thermal energy lost by lime juice 20 × t = 0. 5 × 4 200 × 25 t = (0. 5 × 4 200 × 25) ÷ 20 s = 2 625 s = 43. 8 min Hence the time taken to cool the lime juice from 30 °C to 5 °C is 43. 8 min Chapter 11: Thermal Measurements

Determining Specific Heat Capacity by Electrical Methods Specific Heat Capacity of a Solid A sample of a solid block of mass, m, is obtained. The temperature of the solid before heating is θ 1. A heater of power, P, is turned on for a period of time, t, and the new temperature, θ 2, of the block is recorded. Based on the principle of conservation of energy, Heat loss by the heater is heat gained by the solid block. This is assuming that the heat lost to the surroundings is negligible. The specific heat capacity of the solid, c, is as follows: Pt = mc(θ 2 − θ 1) c= Pt m(θ 2 − θ 1) Chapter 11: Thermal Measurements

Specific Heat Capacity of Liquid The mass of the empty calorimeter and the specific heat capacity of the calorimeter must be known. The temperatures of the liquid before and after turning on the electric heater for a period of time were recorded. Based on the principle of conservation of energy, Pt = mcliquid(θ 2 − θ 1) + Mccalorimeter(θ 2 − θ 1) c= Pt m(θ 2 − θ 1)ccalorimeter Mccalorimeter m Where: P = power of the electric heater (W) t = time taken for temperature to rise from θ 1 to θ 2 (s) θ = temperature of liquid (K) m = mass of liquid (kg) M = mass of calorimeter (kg) cliquid = specific heat capacity of liquid (J kg− 1 K− 1) ccalorimeter = specific heat capacity of calorimeter (J kg− 1 K− 1) Chapter 11: Thermal Measurements

Chapter 11: Thermal Measurements

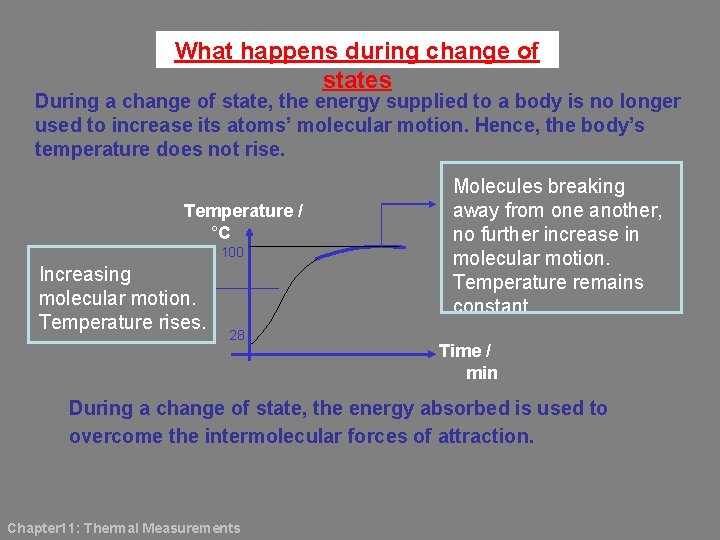
What happens during change of states During a change of state, the energy supplied to a body is no longer used to increase its atoms’ molecular motion. Hence, the body’s temperature does not rise. Temperature / °C 100 Increasing molecular motion. Temperature rises. 28 Molecules breaking away from one another, no further increase in molecular motion. Temperature remains constant. Time / min During a change of state, the energy absorbed is used to overcome the intermolecular forces of attraction. Chapter 11: Thermal Measurements

Specific Latent Heat The specific latent heat of fusion (lf) of a substance is the quantity of heat needed to change a unit mass of the substance from solid state to liquid state without a temperature change The specific latent heat of vaporization (lv) of a substance is the quantity of heat needed to change a unit mass of the substance from liquid state to vapour state without a temperature change General equation : Chapter 11: Thermal Measurements Q=m×l

Evaporatio E nvaporation is the change in state from liquid to gas that takes place at the surface of a liquid. More energetic molecules escape Less energetic molecules remain in the liquid Consequently: ⇒ Average kinetic energy of the molecule remaining in the water is lowered. ⇒ Evaporation produces cooling effect. Molecules in the liquid differ from one another in that some are more energetic, while the others are less energetic. As the more energetic liquid molecules escape into the air, they carry with them their energy. As a result, the average energy of the remaining water is now lowered. Chapter 11: Thermal Measurements

Differences between Boiling and Evaporation Chapter 11: Thermal Measurements

Chapter 11: Thermal Measurements

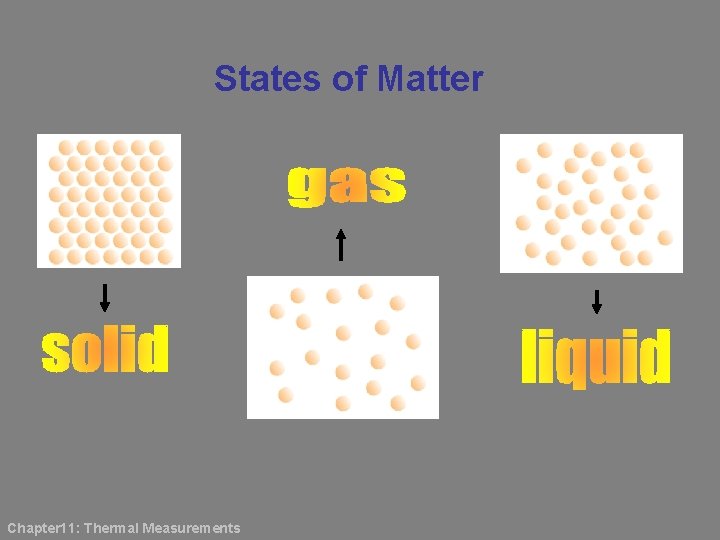
States of Matter Chapter 11: Thermal Measurements

In a solid, the molecules are arranged close together and in a regular pattern. Strong attractive intermolecular forces hold the molecules in position such that they can only vibrate in their fixed positions. In a liquid, the molecules are not arranged in a regular pattern. They are slightly further apart than in solids. The attractive intermolecular forces are not very strong hence the molecules are able to move more freely as compared to the molecules in solids. Chapter 11: Thermal Measurements

In a gas, the molecules are far apart. The molecules move about randomly with high speed. The intermolecular forces are weak. Chapter 11: Thermal Measurements