Chapter 02 Basic Chemistry Basic Chemistry Review You

Chapter 02 Basic Chemistry

Basic Chemistry Review You should already know 90% of this information! Do NOT write down everything, only what you don’t know. 2

Basic Chemistry Chemical Elements Matter: Refers to anything that has mass and occupies space Only 92 naturally occurring fundamental types of matter – 92 Elements Organisms composed primarily (98%) of only six elements Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, Sulfur CHNOPS 3

Crust versus Organisms Basic Chemistry 4

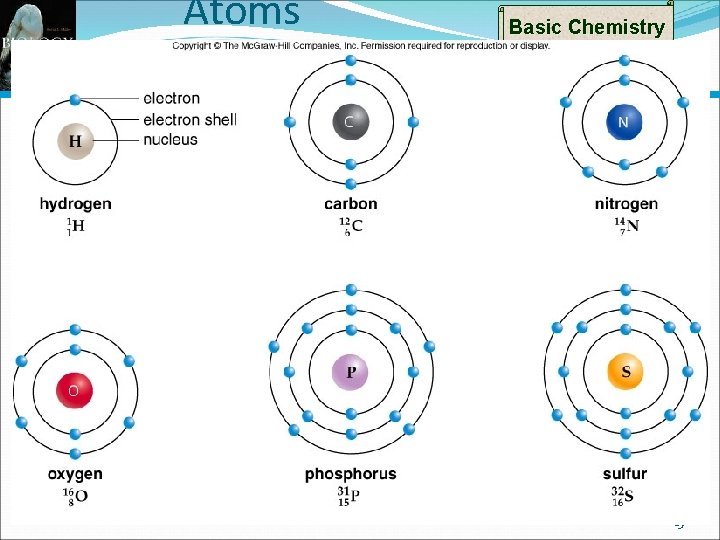
Basic Chemistry Atomic Structure Atoms - Smallest particles of elements Atoms composed of three types of subatomic particles Protons Positive charge In nucleus Neutrons Neutral charge Also found in nucleus Electrons Negatively charge Orbit nucleus 5
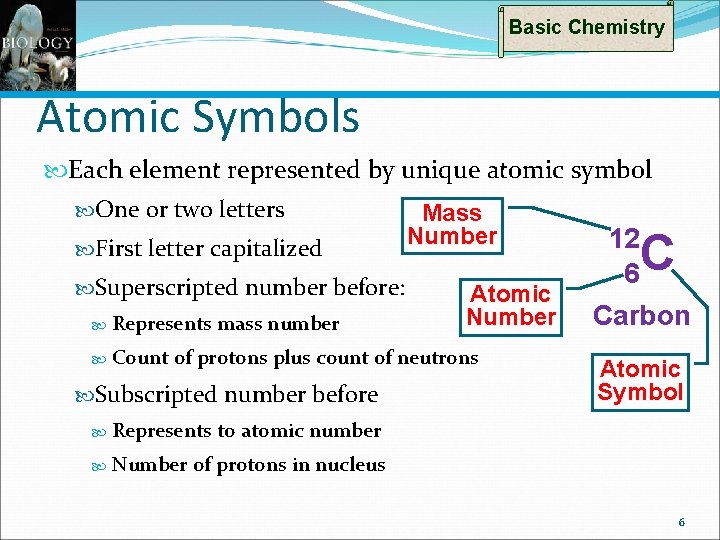
Basic Chemistry Atomic Symbols Each element represented by unique atomic symbol One or two letters First letter capitalized Superscripted number before: Represents Count mass number Mass Number Atomic Number of protons plus count of neutrons Subscripted number before Represents Number 12 6 C Carbon Atomic Symbol to atomic number of protons in nucleus 6
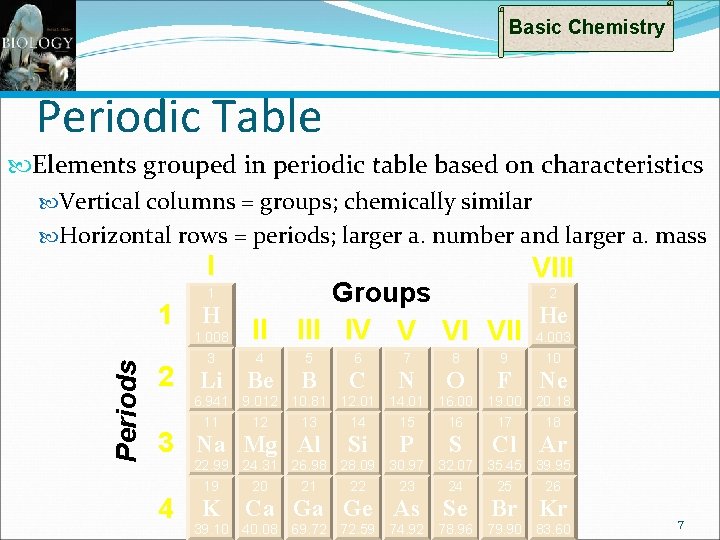
Basic Chemistry Periodic Table Elements grouped in periodic table based on characteristics Vertical columns = groups; chemically similar Horizontal rows = periods; larger a. number and larger a. mass I VIII 1 Periods 1 H 1. 008 3 II 4 2 Groups He III IV V VI VII 4. 003 5 2 Li Be B 6 7 8 9 10 C N O F Ne 6. 941 9. 012 10. 81 12. 01 14. 01 16. 00 19. 00 20. 18 11 12 13 14 15 16 17 18 3 Na Mg Al Si P S Cl Ar 22. 99 24. 31 26. 98 28. 09 30. 97 32. 07 35. 45 39. 95 19 20 21 22 23 24 25 26 4 K Ca Ga Ge As Se Br Kr 39. 10 40. 08 69. 72 72. 59 74. 92 78. 96 79. 90 83. 60 7

Basic Chemistry Isotopes Atoms of the same element with a differing numbers of neutrons 12 6 13 6 C C Carbon 13 Carbon 12 Some isotopes spontaneously decay 14 6 C Carbon 14 Radioactive Give Can off energy in the form of rays and subatomic particles be used as tracers Mutagenic – Can cause cancer 8
for Basic Chemistry Low Level Radiation 9
High Level Radiation Basic Chemistry 10

Basic Chemistry Electron Shells, Orbitals and Energy Levels Atoms normally have as many electrons as protons Opposite charges balance leaving atom neutral Electrons are attracted to the positive nucleus Revolve around nucleus in orbitals Can be pushed into higher orbitals with energy Release that energy when they fall back to lower orbital Different energy levels referred to as electron shells 11

Basic Chemistry The Octet Rule for Distribution of Electrons Bohr models show electron shells as concentric circles around nucleus Each shell has one or more electron orbitals Innermost shell has two electrons Others have 8 or multiples thereof Atoms with fewer than 8 electrons in outermost shell are chemically reactive If 3 or less – Tendency to donate electrons If 5 or more – Tendency to receive electrons 12
Atoms Basic Chemistry 13
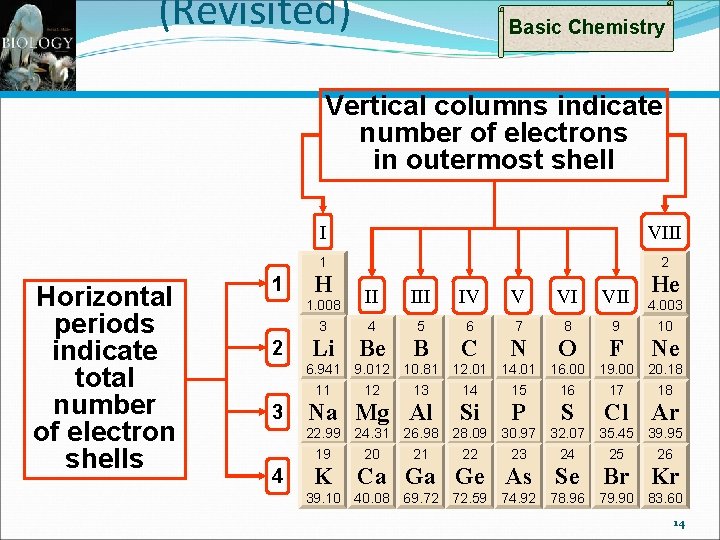
(Revisited) Basic Chemistry Vertical columns indicate number of electrons in outermost shell Horizontal periods indicate total number of electron shells 1 I VIII 1 2 H 1. 008 3 2 Li He II IV V VI VII 4 5 6 7 8 9 4. 003 10 Be B C N O F Ne 6. 941 9. 012 10. 81 12. 01 14. 01 16. 00 19. 00 20. 18 11 12 13 14 15 16 17 18 3 Na Mg Al Si P S Cl Ar 22. 99 24. 31 26. 98 28. 09 30. 97 32. 07 35. 45 39. 95 19 20 21 22 23 24 25 26 4 K Ca Ga Ge As Se Br Kr 39. 10 40. 08 69. 72 72. 59 74. 92 78. 96 79. 90 83. 60 14

Basic Chemistry Elements and Compounds Molecule - Two or more atoms bonded together If all atoms in molecule are of the same element Material O 2, is still an element H 2, N 2, etc. If at least one atom is from a different element Material CO 2, formed is a compound H 2 O, C 6 H 12 O 6, etc. Characteristics elements dramatically different from constituent 15

Basic Chemistry Chemical Bonding Bonds between atoms are caused by electrons in outermost shells The process of bond formation is called a reaction The intensity of simple reactions can be predicted by the periodic table If two elements are horizontally close in the table, they usually react mildly If they are horizontally far apart, they usually react vigorously 16

Types of Bonds: Ionic Bonding Basic Chemistry Ionic Bonds Octet rule: Atoms If “want” 8 electrons in outer shell have < 4 outers, desire to donate them If have > 4 outers, desire to receive more Consider two elements from opposite ends of periodic table Element from right side: Has 7 electrons in outer shell “Desperately wants” one more (7+1=8) Element Has from left side: only 1 electron in outer shell “Desperately wants” to donate it (1 -1=0=8) 17

Types of Bonds: Ionic Bond Example Basic Chemistry Sodium: From left end Has 1 outer electron Chlorine: From right end of table Has 7 outer electrons In reaction, Na completely gives up its outer electron to Cl Na now a positive ion and Cl a negative ion Dissimilar charges now bind ions together Forms sodium chloride An ionic compound Na. Cl table salt) 18
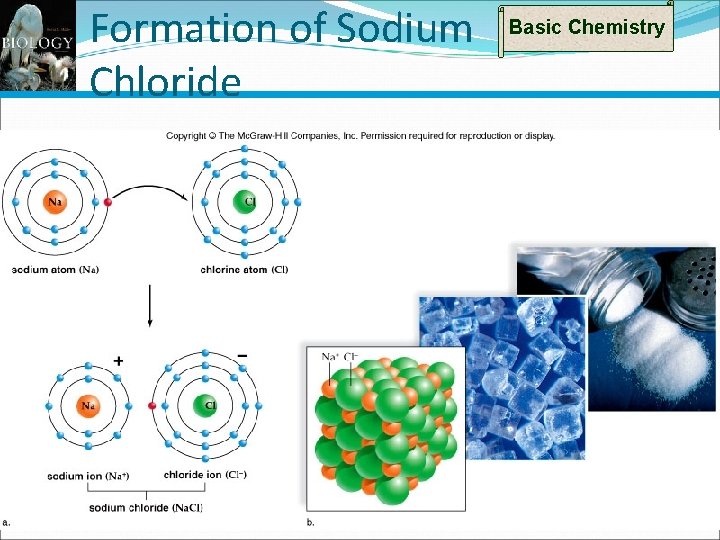
Formation of Sodium Chloride Basic Chemistry 19

Types of Bonds: Covalent Bonds Basic Chemistry When atoms are horizontally closer together in the periodic table The electrons are not permanently transferred from one atom to the other like in Na. Cl A pair of electrons from the outer shell will “time share” with one atom and then the other This also causes the atoms to remain together Known as covalent bonding Sometimes two pair of electrons are shared between atoms – a double covalent bond 20

Basic Chemistry Covalently Bonded Molecules 21

Basic Chemistry Nonpolar Covalent Bonds Consider two elements that are equidistant from the edges of the periodic table Atoms will have about equal affinity for electrons One will “want” (with a specific intensity) to donate electron(s) The other will “want, ” with the same intensity, to receive electron(s) When bonded covalently: The bond electrons will spend about equal time with both atoms Such covalent bonds are said to be nonpolar 22

Basic Chemistry Polar Covalent Bonds Consider two elements with one much closer to the edge of the table than the other Atoms will have unequal affinity for electrons One will “want” (with a specific intensity) to donate or receive electron(s) The other will “want” (with a different intensity) to donate or receive electron(s) When bonded covalently: The bond electrons will spend more time with one atom than the other The atom that gets the most time with the electrons will be slightly negative The other will be slightly positive Such covalent bonds are said to be polar 23

Types of Bonds: Hydrogen Bonds Basic Chemistry Water (H 2 O or H–O–H) is a polar molecule Electrons spend more time with O than H’s become slightly +, O slightly – When polar molecules are dissolved in water The H’s of water molecules are attracted to the negative parts of the solute molecules Results in a weak bond – the hydrogen bond Easily broken, but many together can be quite strong 24
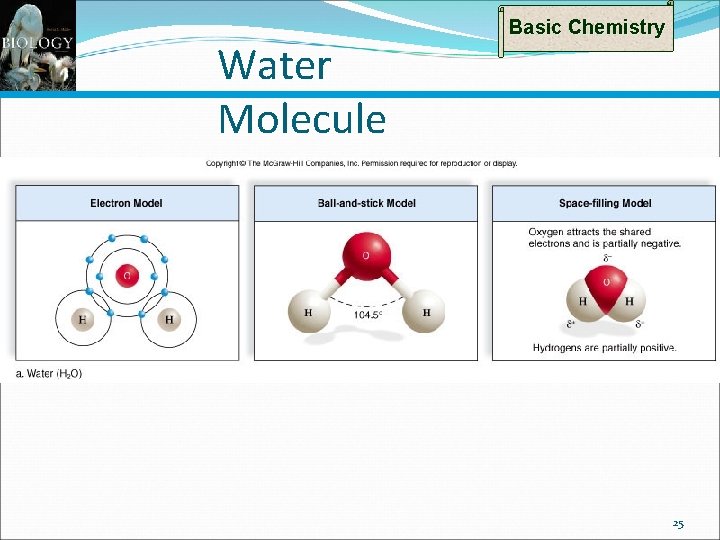
Water Molecule Basic Chemistry 25
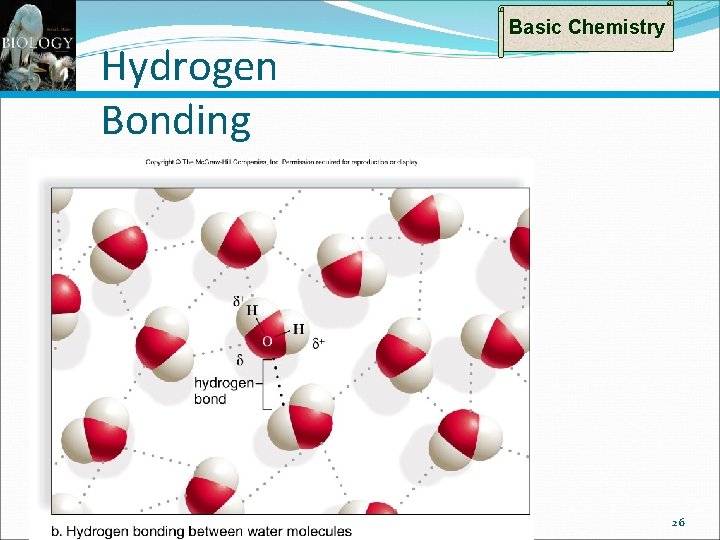
Hydrogen Bonding Basic Chemistry 26

Basic Chemistry The Chemistry of Water: Heat Capacity Water has a high heat capacity Temperature = rate of vibration of molecules Apply heat to liquid Molecules bounce faster Increases temperature But, when heat applied to water Hydrogen bonds restrain bouncing Temperature rises more slowly per unit heat Water at a given temp. has more heat than most liquids Thermal inertia – resistance to temperature change More heat required to raise water one degree than most other liquids (1 calorie per gram) Also, more heat is extracted/released when lowering water one degree than most other liquids 27

Properties of Water: Heat of Vaporization Basic Chemistry High heat of vaporization To raise water from 98 to 99 ºC; ~1 calorie To raise water from 99 to 100 ºC; ~1 calorie However, large numbers of hydrogen bonds must be broken to evaporate water To raise water from 100 to 101 ºC; ~540 calories! This is why sweating (and panting) cools Evaporative cooling is best when humidity is low because evaporation occurs rapidly Evaporative cooling works poorest when humidity is high because evaporation occurs slowly 28
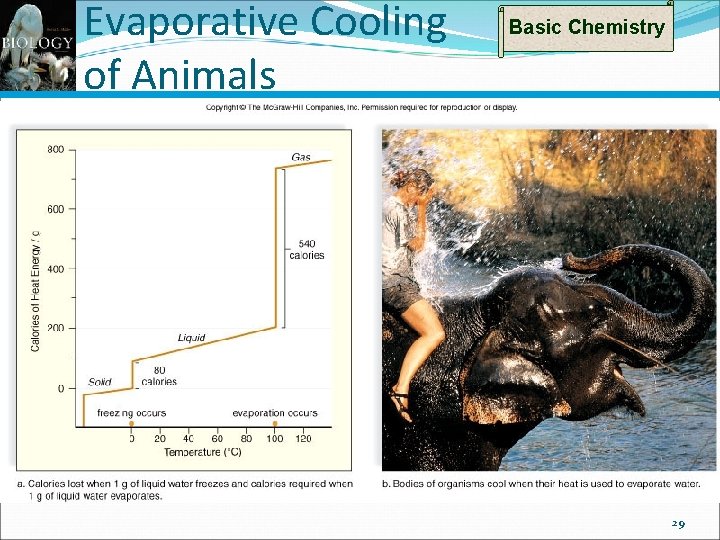
Evaporative Cooling of Animals Basic Chemistry 29

Properties of Water: Heat of Fusion Basic Chemistry Heat of fusion (melting) To raise ice from -2 to -1 ºC; ~1 calorie To raise water from -1 to 0 ºC; ~1 calorie To raise water from 0 to 1 ºC; ~80 calories! This is why ice at 0 ºC keeps stuff cold MUCH longer than water at 1 ºC This is why ice is used for cooling NOT because ice is cold But because it absorbs so much heat before it will warm by one degree 30
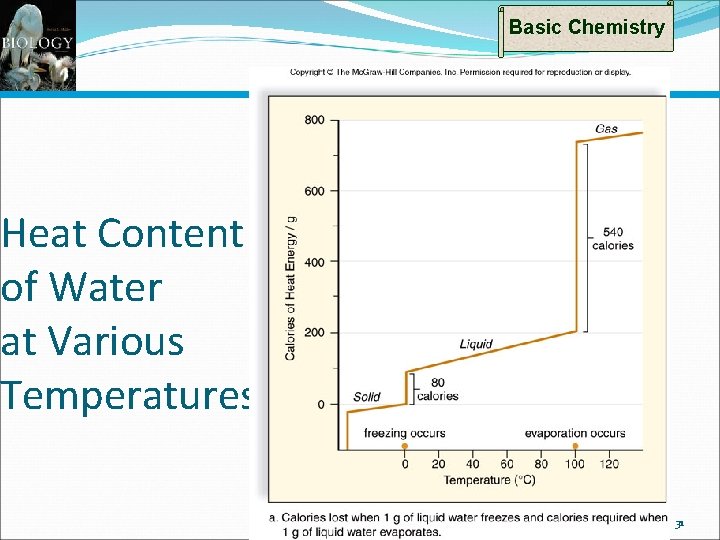
Basic Chemistry Heat Content of Water at Various Temperatures 31

Properties of Water: Water as a Solvent Basic Chemistry Solutions consist of: A solvent (the most abundant part) and A solute (less abundant part) that is dissolved in the solvent Polar compounds readily dissolve; hydrophilic Nonpolar compounds dissolve only slightly; hydrophobic Ionic compounds dissociate in water Na+ Attracted to negative (O) end of H 2 O Each Na+ completely surrounded by H 2 O Cl Attracted to positive (H 2) end of H 2 O Each Cl- completely surrounded by H 2 O 32

Basic Chemistry Properties of Water: Uniqueness of Ice Frozen water less dense than liquid water Otherwise, oceans and deep lakes would fill with ice from the bottom up Ice acts as an insulator on top of a frozen body of water Melting ice draws heat from the environment 33
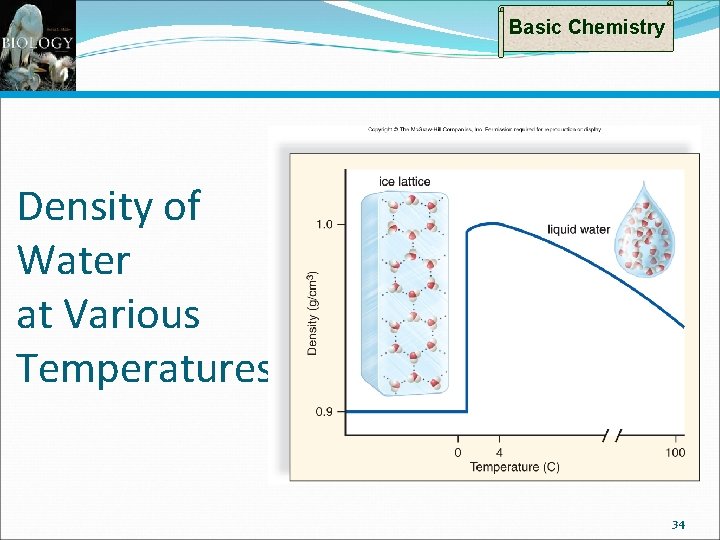
Basic Chemistry Density of Water at Various Temperatures 34

Basic Chemistry Stratification of a pond in winter 35

Basic Chemistry Water as a Transport Medium 36

Properties of Water: Cohesion & Adhesion Basic Chemistry Cohesive and Adhesive Cohesion – Hydrogen bonds hold water molecules tightly together Adhesion – Hydrogen bonds for between water and other polar materials Allow water be drawn many meters up a tree in a tubular vessel High Surface Tension Water molecules at surface hold more tightly than below surface Amounts to an invisible “skin” on water surface Allows small nonpolar objects (like water strider) to sit on top of water 37

p. H of Water: Acids Basic Chemistry Acids Dissociate in water and release hydrogen ions (H+) Sour to taste Hydrochloric acid (stomach acid) is a gas with symbol HCl In water, it dissociates into H+ and Cl- Dissociation a strong acid of HCl is almost total, therefore it is 38

p. H of Water: Bases Basic Chemistry Bases: Either take up hydrogen ions (H+) or release hydroxide ions (OH-) Bitter to taste Sodium hydroxide (drain cleaner) is a solid with symbol Na. OH In water, it dissociates into Na+ and OH- Dissociation of Na. OH is almost total, therefore it is a strong base 39

Basic Chemistry p. H Scale p. H scale used to indicate acidity and alkalinity of a solution. Values range from 0 -14 0 to <7 = Acidic 7 = Neutral >7 to 14 = Basic (or alkaline) Logarithmic Scale Each unit change in p. H represents a change of 10 X p. H of 4 is 10 X as acidic as p. H of 5 p. H of 10 is 100 X more basic than p. H of 8 40
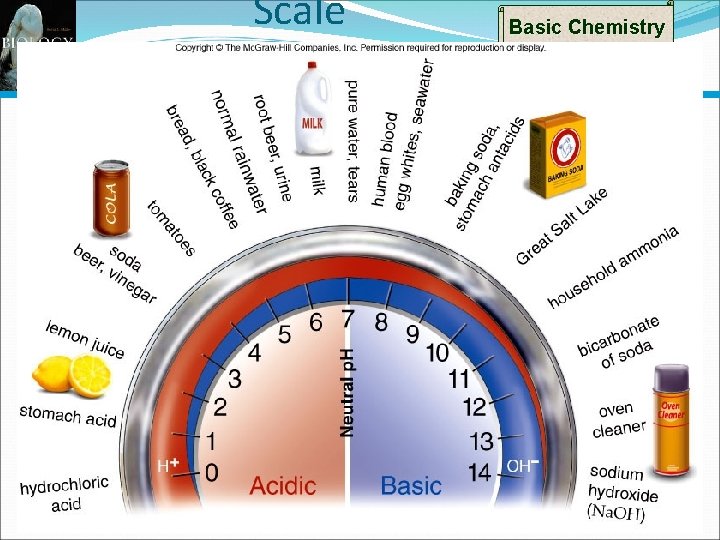
Scale Basic Chemistry 41

Basic Chemistry Buffers and p. H When H+ is added to pure water at p. H 7, p. H goes down and water becomes acidic When OH- is added to pure water at p. H 7, p. H goes up and water becomes alkaline Buffers are solutes in water that resist change in p. H When H+ is added, buffer may absorb, or counter by adding OH- When OH- is added, buffer may absorb, or counter by adding H+ 42

Basic Chemistry Buffers in Biology Health of organisms requires maintaining p. H of body fluids within narrow limits Human blood normally 7. 4 (slightly alkaline) Many foods and metabolic processes add or subtract H + or OH- ions Reducing blood p. H to 7. 0 results in acidosis Increasing Both blood p. H to 7. 8 results in alkalosis life threatening situations Bicarbonate ion (-HCO 3) in blood buffers p. H to 7. 4 43

Basic Chemistry Review Chemical Elements Atoms Isotopes Molecules and Compounds Chemical Bonding Ionic and Covalent Hydrogen Properties of Water Acids and Bases 44

Ending Slide Chapter 02 Basic Chemistry
- Slides: 45