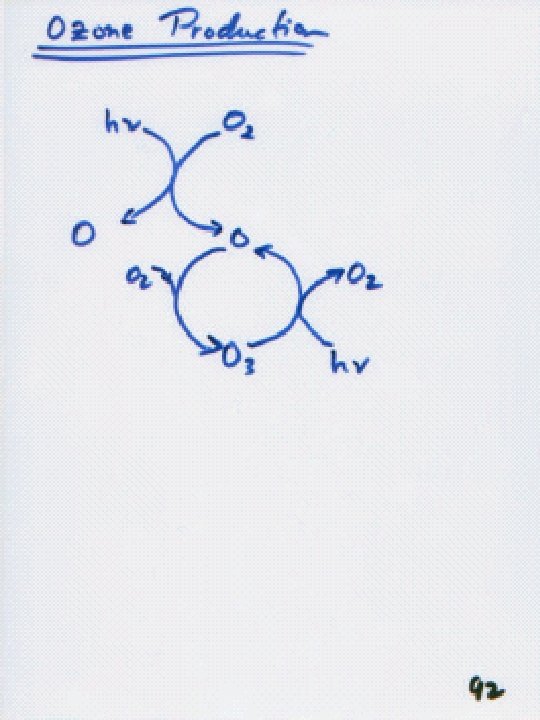
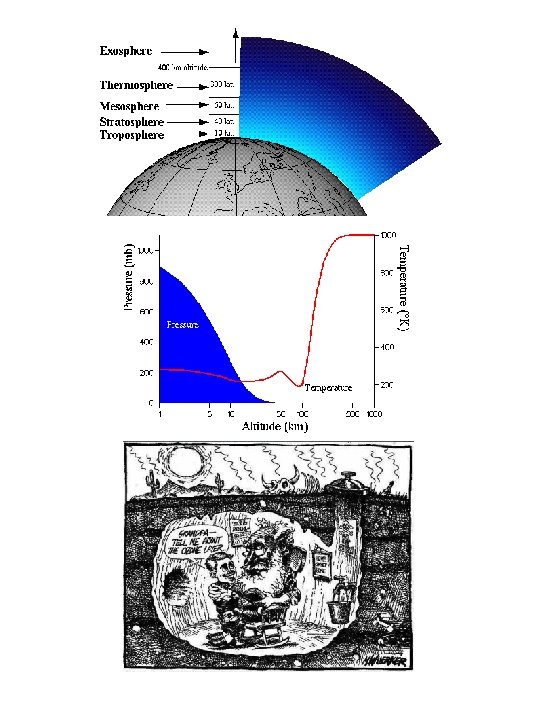
Chapman Mechanism 1930 Sidney Chapman O 2 h





![In lower stratosphere (~15 -25 km), [O] is relatively low: UV-C absorbed by ozone. In lower stratosphere (~15 -25 km), [O] is relatively low: UV-C absorbed by ozone.](https://slidetodoc.com/presentation_image_h/114b230060b4aa8d11926696f0dd31dd/image-13.jpg)






- Slides: 22

Chapman Mechanism (~1930, Sidney Chapman) O 2 + h O + O ( <242. 4 nm) O + O 2 + M O 3 + M O 3 + h O + O 2 ( <280 nm) O + O 3 2 O 2 O + M O 2 + M







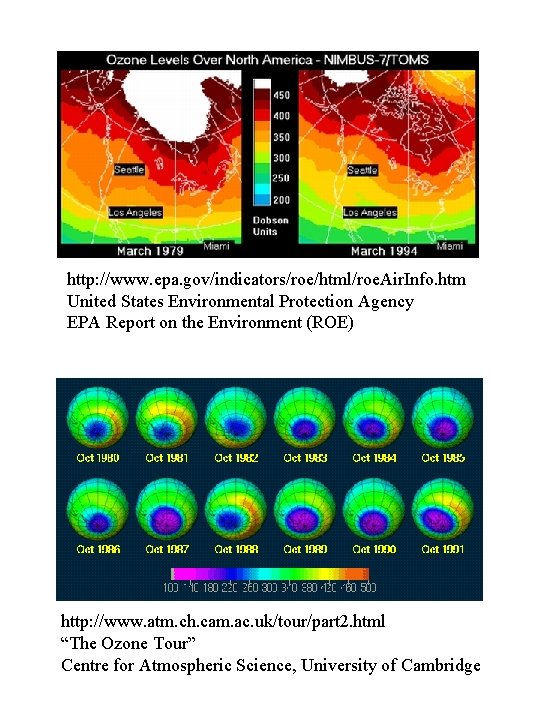
http: //www. epa. gov/indicators/roe/html/roe. Air. Info. htm United States Environmental Protection Agency EPA Report on the Environment (ROE) http: //www. atm. ch. cam. ac. uk/tour/part 2. html “The Ozone Tour” Centre for Atmospheric Science, University of Cambridge


“The Ozone Tour” -- http: //www. atm. ch. cam. ac. uk/tour/part 3. html Centre for Atmospheric Science, University of Cambridge Nobel Prize 1995 (1/3 each) Paul J. Crutzen Mario J. Molina F. Sherwood Rowland The Netherlands Max-Planck Institut Mainz, Germany USA M. I. T. Cambridge, Mass. USA University of California Irvine, California


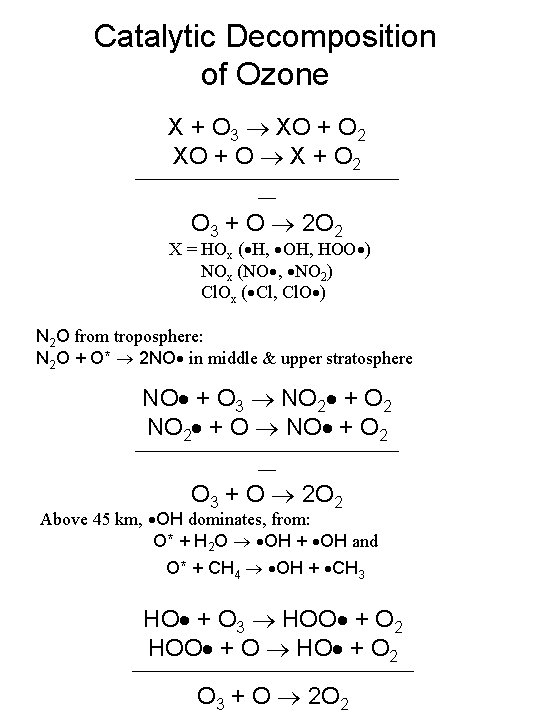
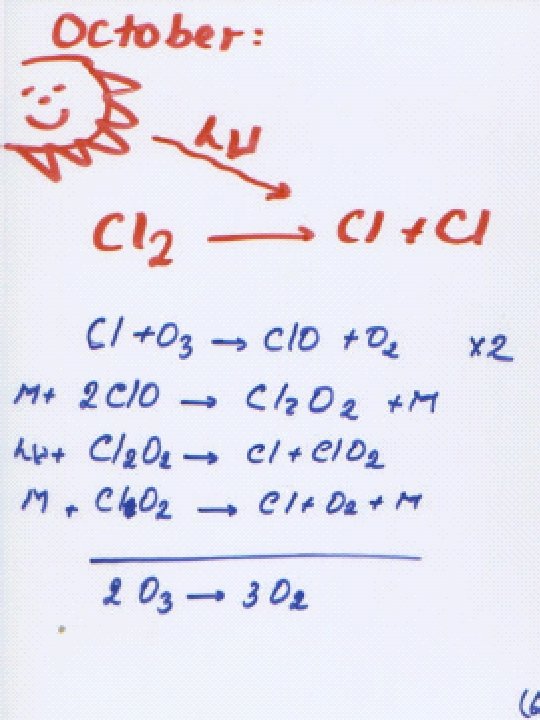
Catalytic Decomposition of Ozone X + O 3 XO + O 2 XO + O X + O 2 _______________ __ O 3 + O 2 O 2 X = HOx ( H, OH, HOO ) NOx (NO , NO 2) Cl. Ox ( Cl, Cl. O ) N 2 O from troposphere: N 2 O + O* 2 NO in middle & upper stratosphere NO + O 3 NO 2 + O 2 NO 2 + O NO + O 2 _______________ __ O 3 + O 2 O 2 Above 45 km, OH dominates, from: O* + H 2 O OH + OH and O* + CH 4 OH + CH 3 HO + O 3 HOO + O 2 HOO + O HO + O 2 ________________ O 3 + O 2 O 2
![In lower stratosphere 15 25 km O is relatively low UVC absorbed by ozone In lower stratosphere (~15 -25 km), [O] is relatively low: UV-C absorbed by ozone.](https://slidetodoc.com/presentation_image_h/114b230060b4aa8d11926696f0dd31dd/image-13.jpg)
In lower stratosphere (~15 -25 km), [O] is relatively low: UV-C absorbed by ozone. [O 2] is high (so most O quickly reacts with it). Therefore, the dominant ozone loss mechanism is: X + O 3 XO + O 2 X + O 3 X O + O 2 XO + X O X + O 2 ________________ 2 O 3 3 O 2 Reaction goes by: XO + X O [XOOX ] X + O 2 Rate of O 3 production depends on [O 2], [O 3], h (UV-C) Destruction is more complex, but depends on [X], UV-B. If something changes, generally [O 3] increases or decreases until it reaches a steady state. Self-healing: [O 3] , UV-C , more O 3 forms below. Next: Atomic Cl and Br as X.

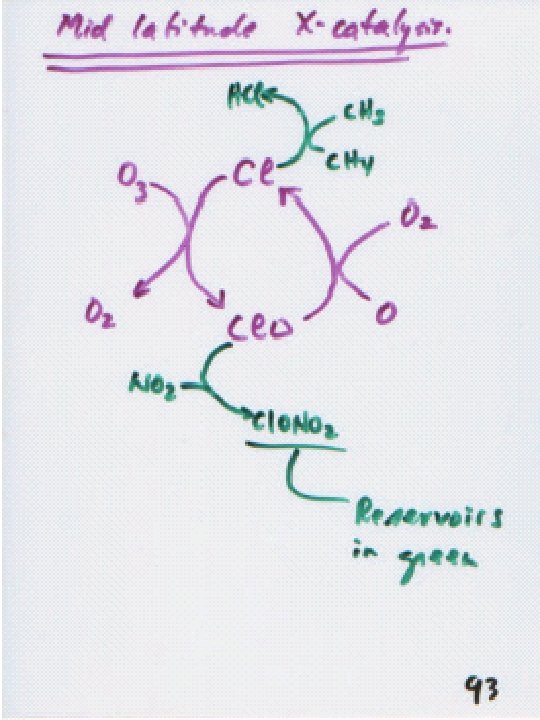
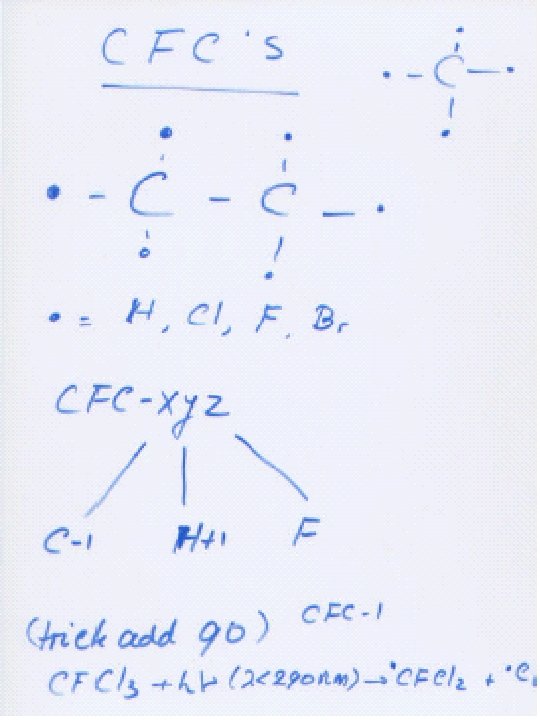
Atomic Cl and Br as X: Cl can destroy tens of thousands of O 3 molecules each, but is mainly in inactive forms (HCl, Cl. ONO 2) in stratosphere. Cl. O + NO 2 Cl. ONO 2 Cl + CH 4 HCl + CH 3 does not operate as an X catalyst, since it combines with O 2 to give CO 2.



On crystals: Cl. ONO 2 g + HCls Cl 2 g + HNO 3 aq Cl 2 + h 2 Cl or: Cl. ONO 2 g + Hs. Oaq HOClaq + HNO 3 aq HClg H+aq + Cl-aq Cl -aq + HOClaq Cl 2 g + OH-aq Crystals bind NO 2 that would normally deactivate Cl , removing it to the troposphere (denitrification). Conditions in the Arctic are similar to those in the Antarctic, but not as severe, because the temperature is not as low there as in the Antarctic.




