Changing States 3 5 C Changing States Vocabulary

Changing States 3. 5 C

Changing States Vocabulary matter gas conductor heat freezing liquid insulator solid evaporating temperature melting

Matter is anything that has mass and takes up space.

Solids have a definite shape and volume. n Solids have mass. n Solids take up space. n

Liquids have a definite volume but no definite shape. n Liquids take the shape of their container. n Liquids have mass. n Liquids take up space. n

Gasses have no definite shape or volume. n Gasses spread out to fill the entire space given. n Gasses have weight. n Gasses take up space. n
States of Matter What does it take for matter to move from one state to another?
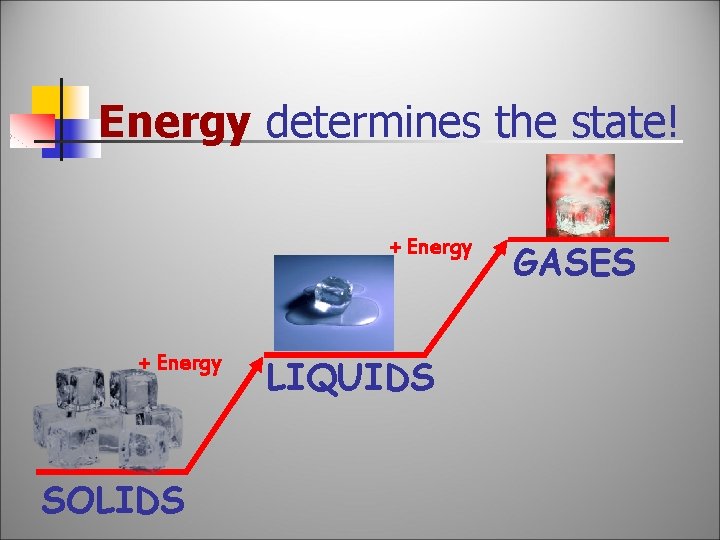
Energy determines the state! + Energy SOLIDS LIQUIDS GASES

Add or Subtract Energy. . . When energy is added, particles move faster! When energy is removed, particles move slower!

• Measure of the movement of molecules (tiny particles) in matter • Measured using a thermometer or temperature probe • Scientists use the Celsius scale to measure temperature

What will happen if heat is added? Why?
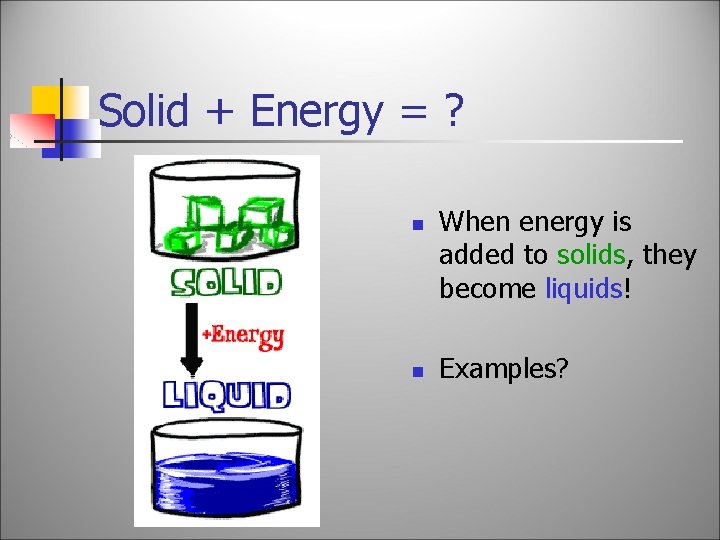
Solid + Energy = ? n n When energy is added to solids, they become liquids! Examples?

Liquid + Energy = ? n n When energy is added to liquids, they become gasses! What examples can you think of?

Changing States n There are several names for matter changing states: n State change n Phase change n Physical change

Ice changes to water—water changes to ice, frozen water is still water

Water changes to steam, a gas, when it is heated to its boiling point, water vapor condenses to form a liquid

Changes in State (phase changes) 1. Melting - solid to liquid 2. Freezing – liquid to solid

Changes in State (phase changes) 3. Evaporating – liquid to gas 4. Condensing - gas to liquid

So, did we get something new? n Ice cream and melted ice cream? n Chocolate and melted chocolate? n Ice, water, and water vapor? n Steel and molten steel?
- Slides: 19