CHANGES OF STATES OF MATTER Phase Changes Matter









- Slides: 18

CHANGES OF STATES OF MATTER Phase Changes

Matter = has mass & takes up space (has volume) Volume = amount of space an object takes up

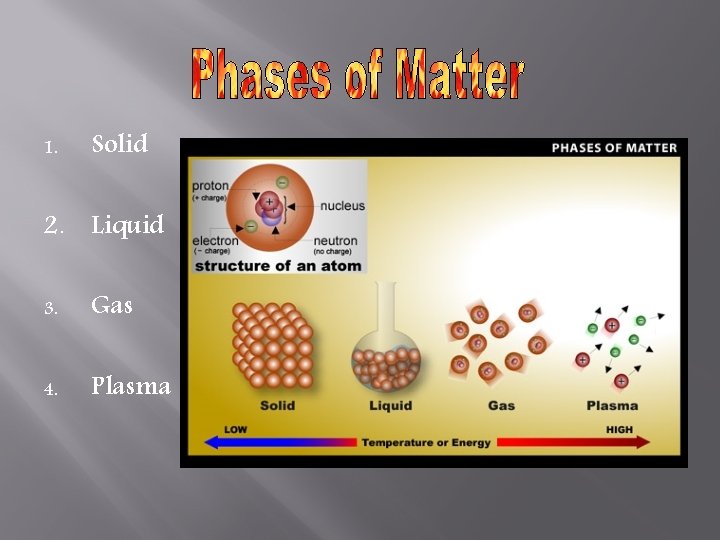
1. Solid 2. Liquid 3. Gas 4. Plasma

Solid Definite shape Definite volume *Molecules have little energy *Molecules packed together

Liquid Indefinite shape Takes shape of container Definite volume *molecules have medium energy *bump into each other & push each other apart

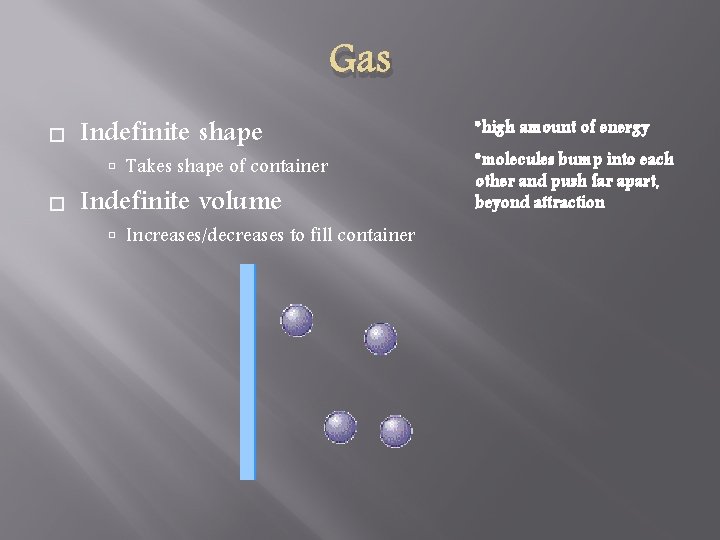
Gas Indefinite shape Takes shape of container Indefinite volume Increases/decreases to fill container *high amount of energy *molecules bump into each other and push far apart, beyond attraction


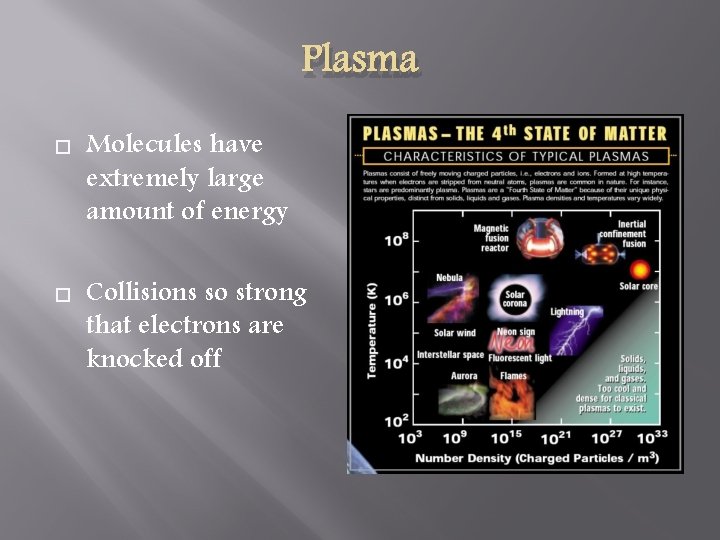
Plasma Molecules have extremely large amount of energy Collisions so strong that electrons are knocked off


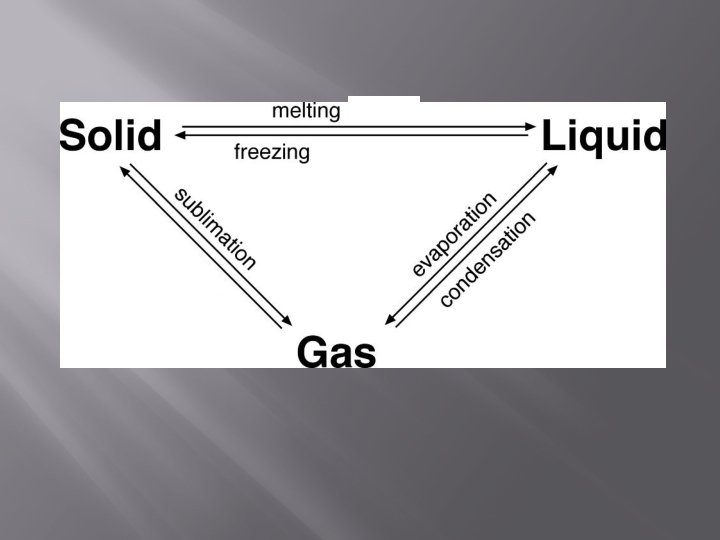
at a certain temperature: *melting point = solid liquid *freezing point = liquid solid *boiling point = liquid gas

How does the physical state of matter change? All Matter has thermal energy Increase thermal energy – molecules move faster Decrease thermal energy – molecules move slower

Melting Increased Thermal Energy Solid Liquid At it’s melting point, the particles of a solid substance are vibrating (moving) so fast that they break free from their fixed positions

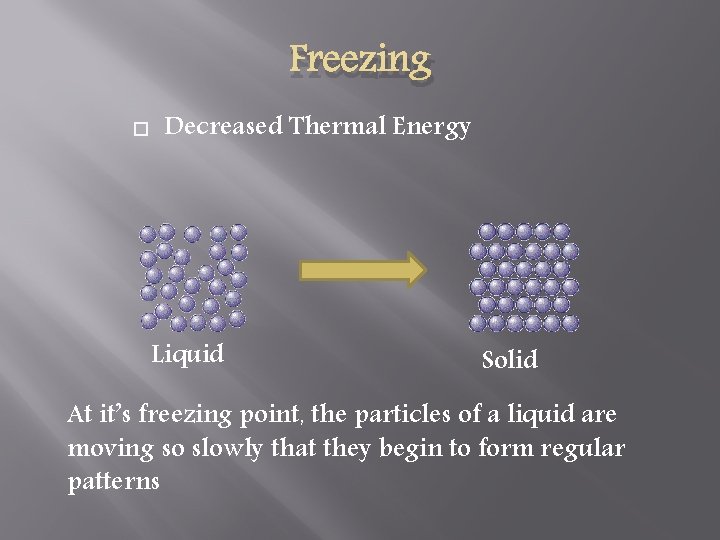
Freezing Decreased Thermal Energy Liquid Solid At it’s freezing point, the particles of a liquid are moving so slowly that they begin to form regular patterns

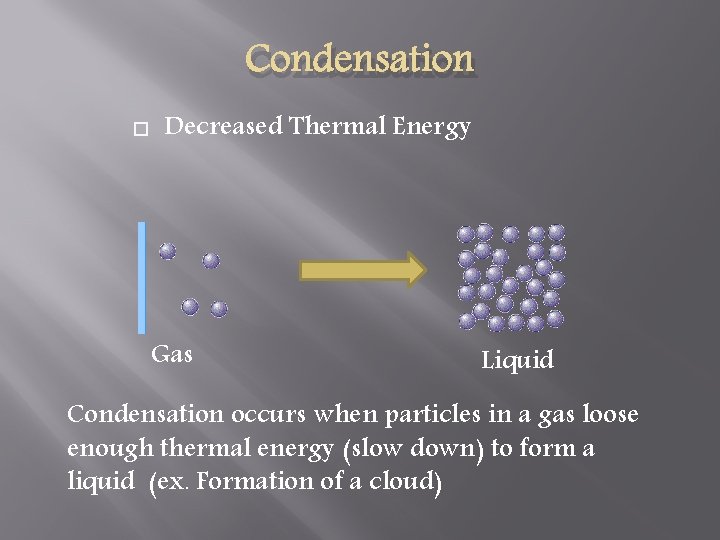
Condensation Decreased Thermal Energy Gas Liquid Condensation occurs when particles in a gas loose enough thermal energy (slow down) to form a liquid (ex. Formation of a cloud)

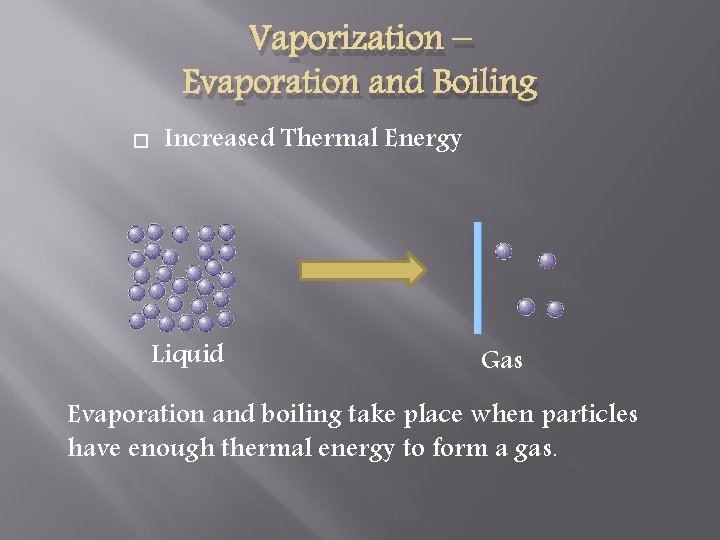
Vaporization – Evaporation and Boiling Increased Thermal Energy Liquid Gas Evaporation and boiling take place when particles have enough thermal energy to form a gas.

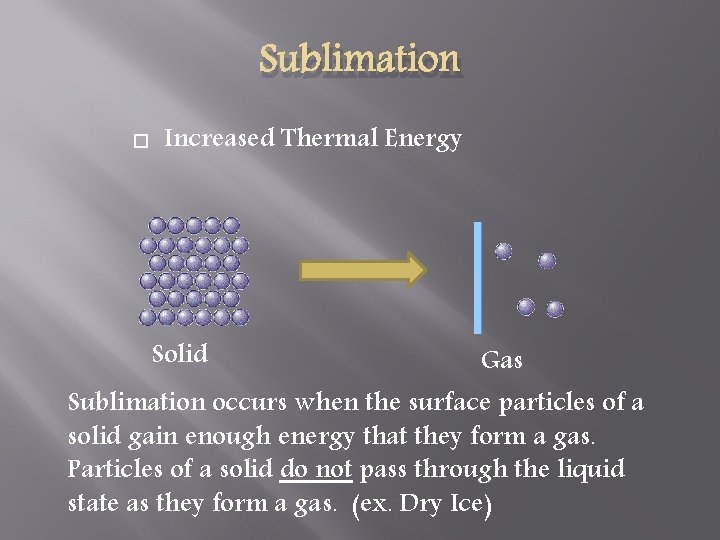
Sublimation Increased Thermal Energy Solid Gas Sublimation occurs when the surface particles of a solid gain enough energy that they form a gas. Particles of a solid do not pass through the liquid state as they form a gas. (ex. Dry Ice)

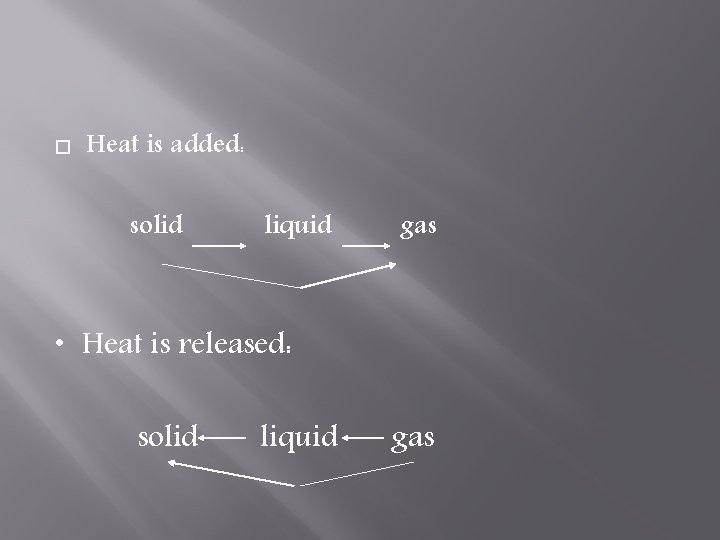
Heat is added: solid liquid gas • Heat is released: solid liquid gas
