Changes in Matter Law of Mass Conservation Mass












































- Slides: 71

Changes in Matter

Law of Mass Conservation � Mass before change = mass after change

Types of changes � Physical changes � Chemical � Nuclear changes

Physical Change � Does not change the fundamental properties of object ◦ Ex. Ripping paper in half, both pieces are still paper ◦ Clothing wearing out, it’s all still cloth � substance still melts at same temperature, is still the same “stuff” � Physical changes can easily be undone

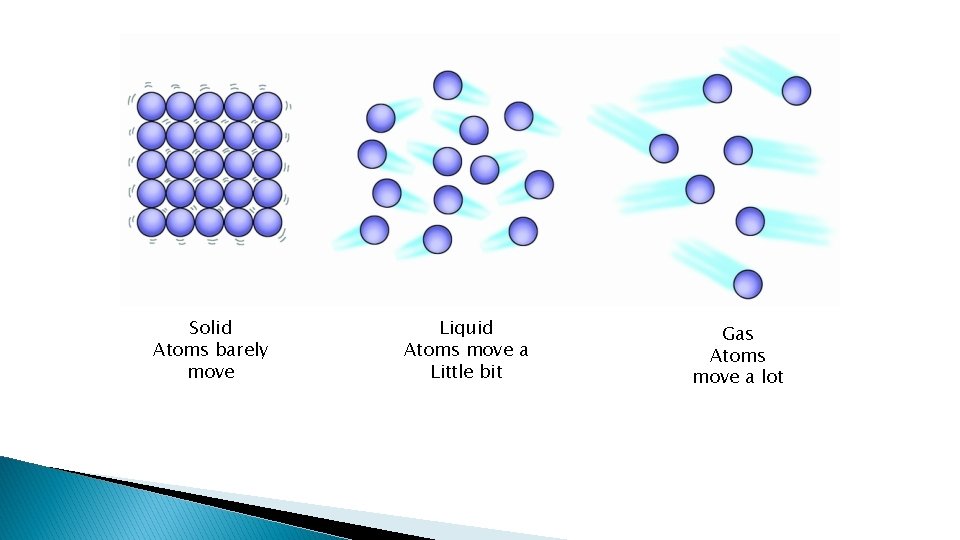
Physical changes cont. � All phase changes are physical changes

Solid Atoms barely move Liquid Atoms move a Little bit Gas Atoms move a lot

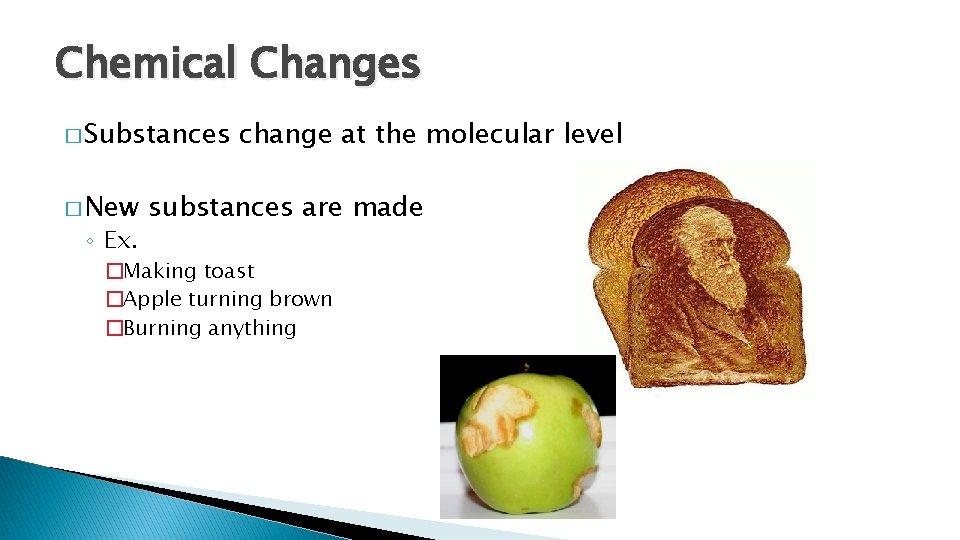
Chemical Changes � Substances � New ◦ Ex. change at the molecular level substances are made �Making toast �Apple turning brown �Burning anything

Chemical changes cont � There 1. are four signs that a chemical reaction has occurred Colour change (extreme, blue to red)

2. Gas is evolved (created)

3. Solid is formed (when there wasn’t one at start)

4. Heat is absorbed or released/Light released

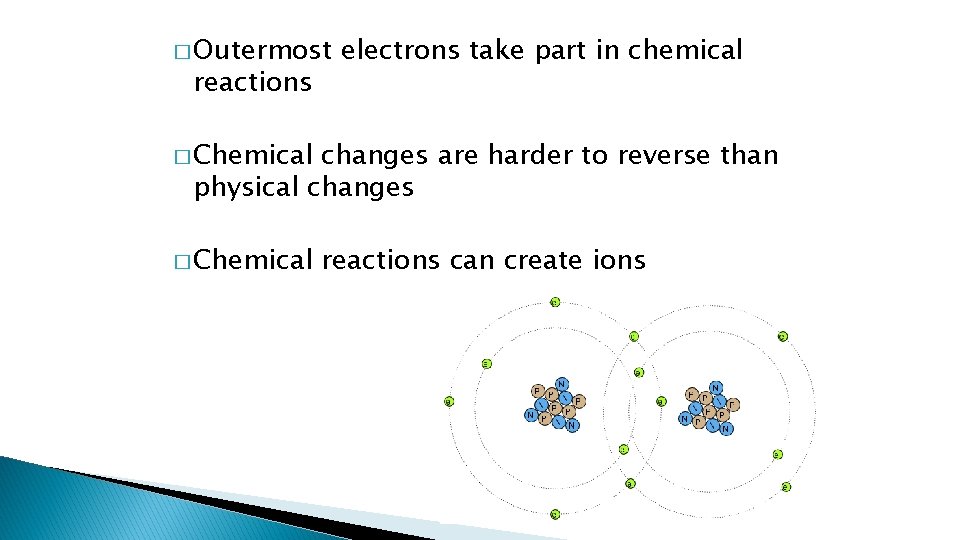
� Outermost reactions electrons take part in chemical � Chemical changes are harder to reverse than physical changes � Chemical reactions can create ions

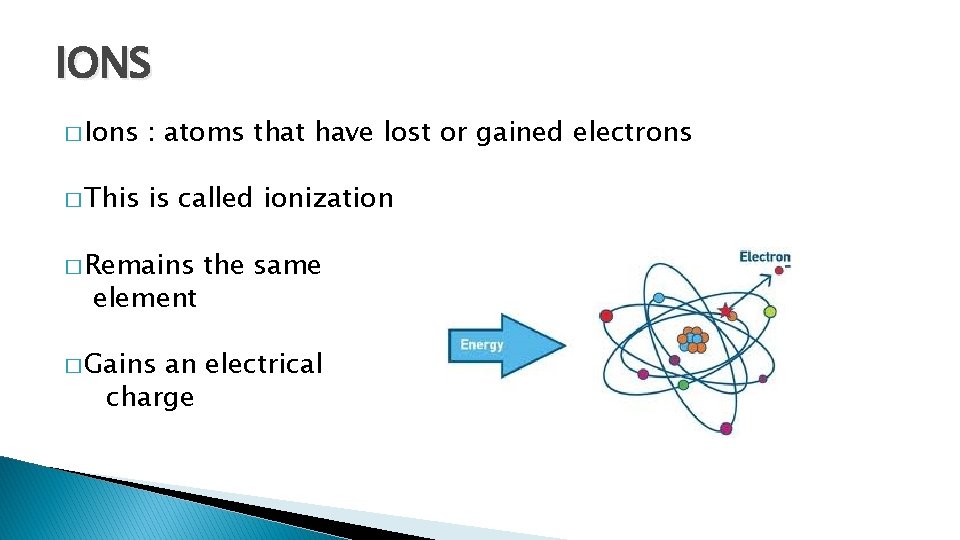
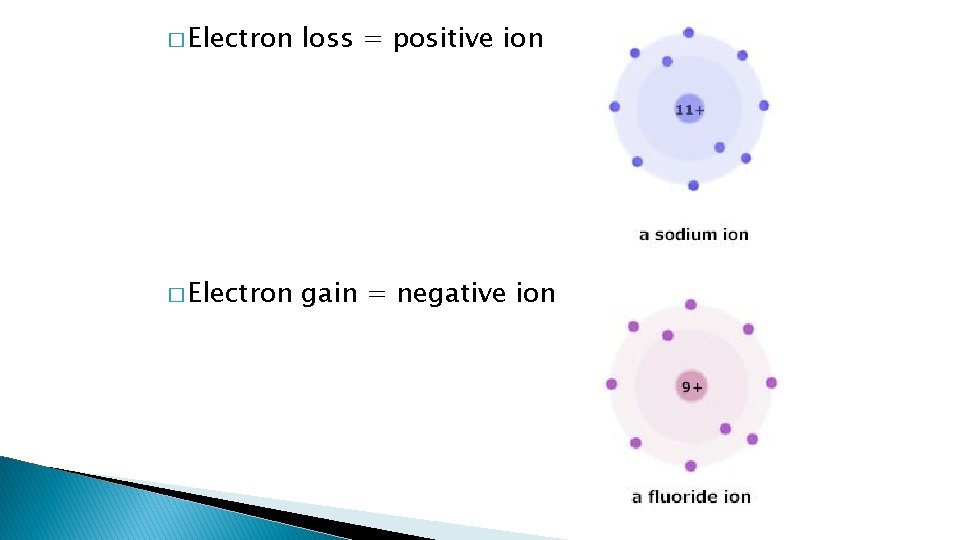
IONS � Ions : atoms that have lost or gained electrons � This is called ionization � Remains element � Gains the same an electrical charge

� Electron loss = positive ion � Electron gain = negative ion



� Question 4. 6 on pg 4. 12

Nuclear Changes � Occur in the nucleus of atoms � Radioactivity is a spontaneous nuclear change called disintegration � actinides and Po, At and Rn undergo nuclear changes

Wilhelm Rontgen

Henri Becquerel

Pierre and Marie Curie Irene Curie and Frederic Joliot Curie

� The curies noticed that when radium disintegrates, it emits different types of radiation.

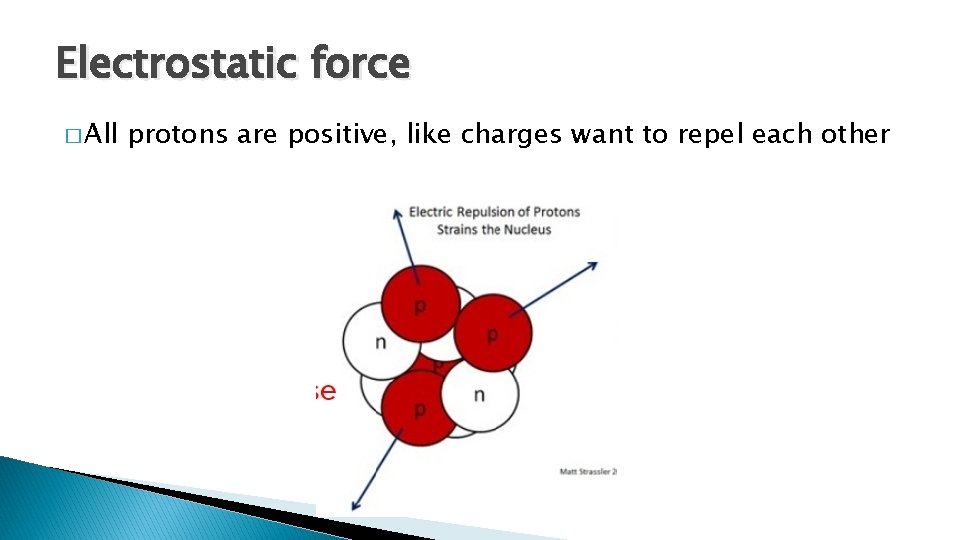
Nuclear stability � Stability of the nucleus depends on the nuclear forces that act between protons and neutrons. � There are two forces acting in the nucleus 1. Electrostatic (force repelling) 2. Nuclear force (force holding it together) � In order for a nucleus the be stable, both forces must be the same.

Electrostatic force � All protons are positive, like charges want to repel each other

Nuclear force � Protons attract neutrons because of strong nuclear forces

Nuclear stability � Nuclei with too many protons or neutrons are unstable. � Should have a ratio of 1: 1 or 1. 5 : 1 ◦ If an atom is unstable, it will try to become stable by splitting into two smaller atoms. ( fission)



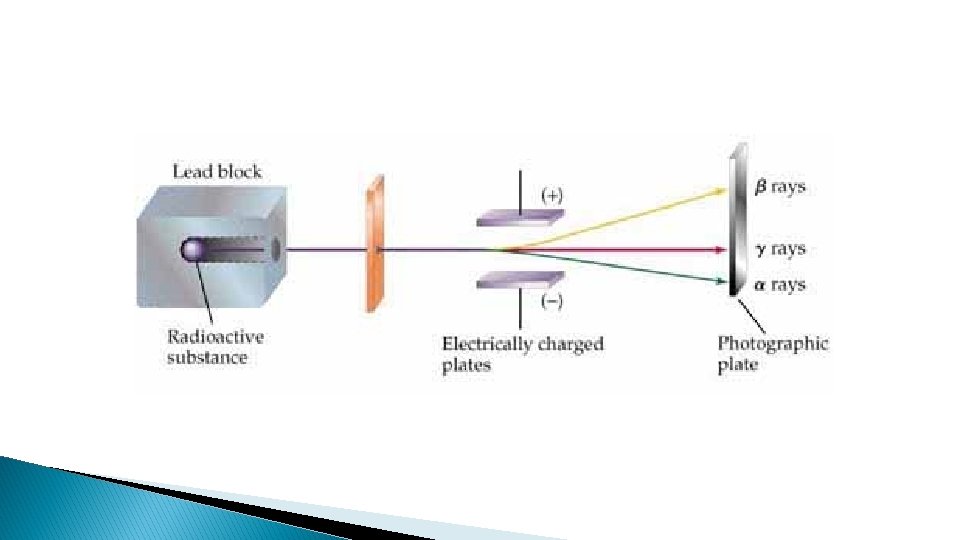
Natural Radioactivity ( table 4. 2 on page 4. 21) Ray Nature speed Electric charge Alpha (α) Helium nucleus (particle) 40000 km/s Positive +2 Beta (β) Electron (particle) Speed of light Negative -1 Gamma (γ) Light wave Speed of light Penetrating Power Low Paper stops it Low 2. 5 cm wood stops it Very high zero 1 m Concrete or thick lead plate stops it

X-rays � Not emitted by radioactive materials � Are light waves � Have no charge � Less penetrating than gamma rays (stopped by 1 inch lead)

� Questions 4. 12, 4. 14, 4. 15, 4. 16 on pg. 4. 22

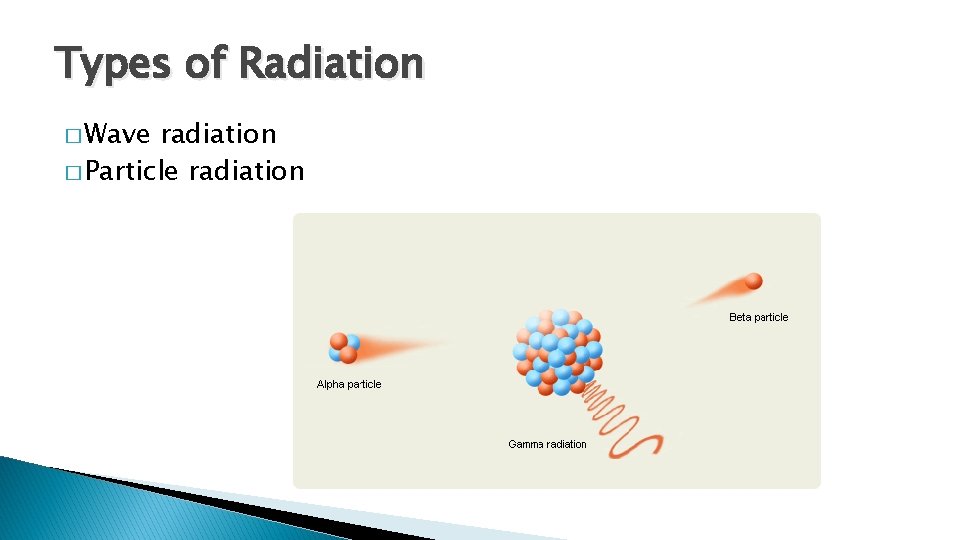
Types of Radiation � Wave radiation � Particle radiation

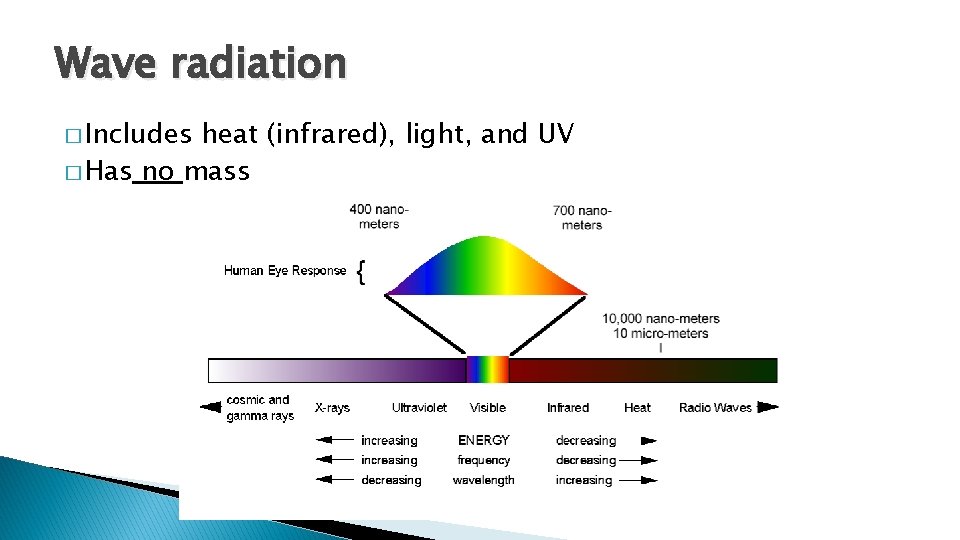
Wave radiation � Includes heat (infrared), light, and UV � Has no mass

Particle Radiation � Have mass

Ionizing Radiation � All types of radiation ionize � They remove an electron.

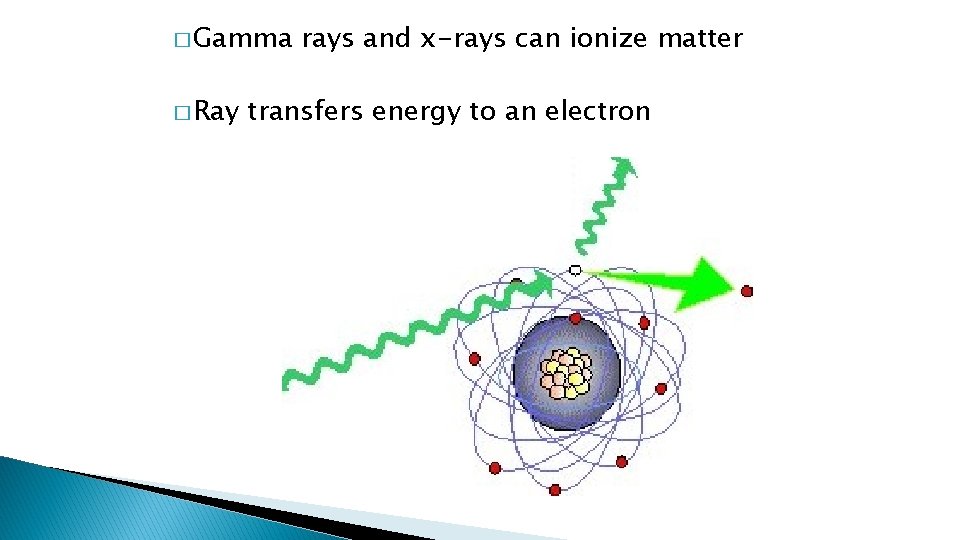
� Gamma � Ray rays and x-rays can ionize matter transfers energy to an electron

� Particle radiation hits electrons knocking it off an atom

� Questions 4. 18, 4. 19, 4. 20 on pg. 4. 27

Radioactive Decay � Unstable atoms breaking down

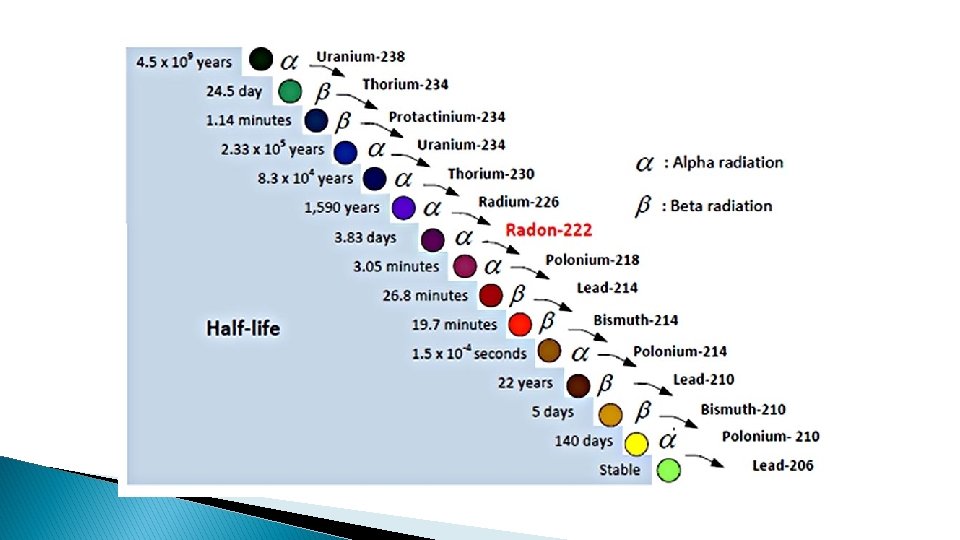
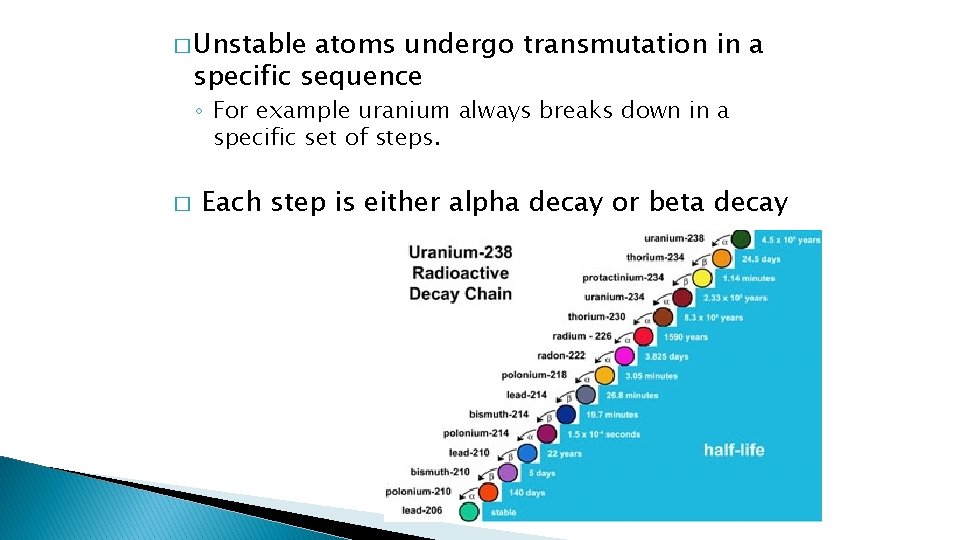
� Unstable atoms undergo transmutation in a specific sequence ◦ For example uranium always breaks down in a specific set of steps. � Each step is either alpha decay or beta decay

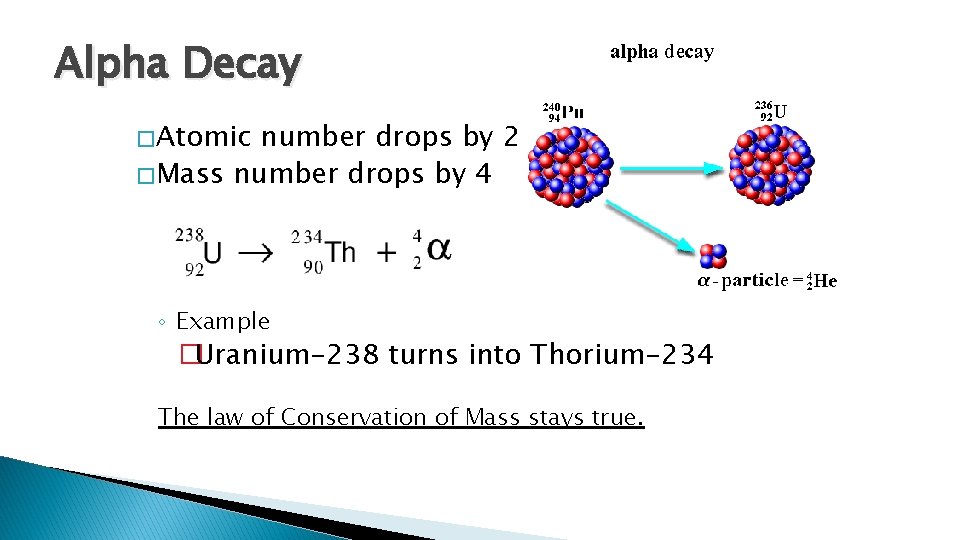
Alpha Decay � Atomic number drops by 2 � Mass number drops by 4 ◦ Example �Uranium-238 turns into Thorium-234 The law of Conservation of Mass stays true.

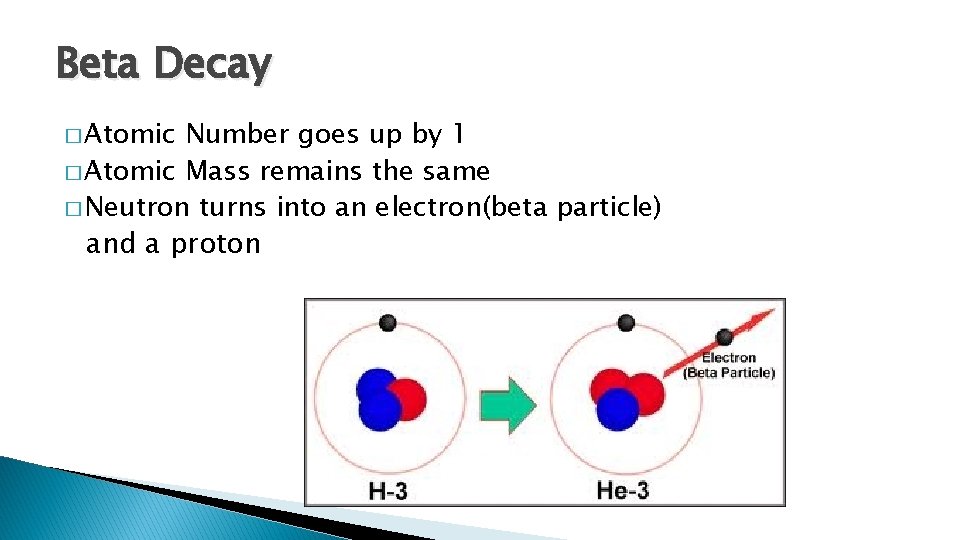
Beta Decay � Atomic Number goes up by 1 � Atomic Mass remains the same � Neutron turns into an electron(beta particle) and a proton

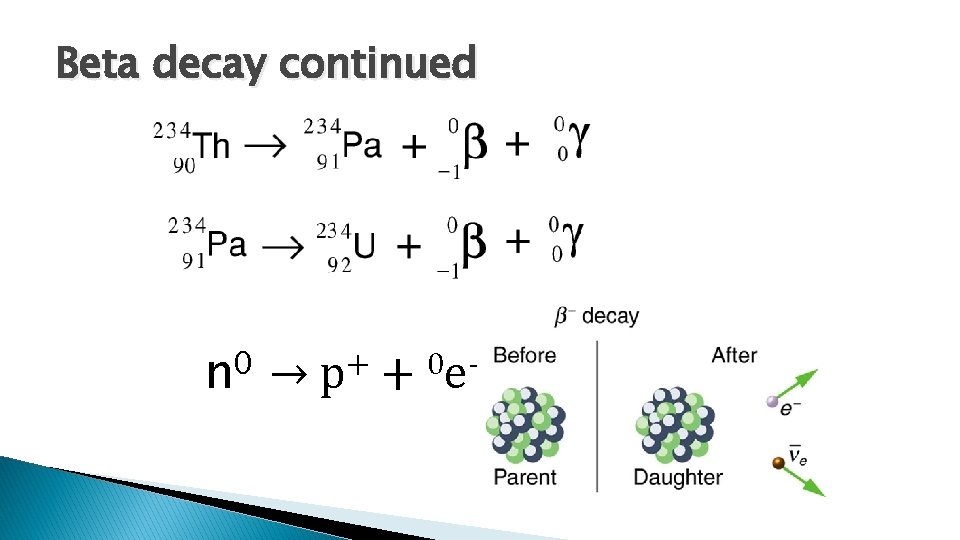
Beta decay continued 0 n → + p + 0 e -


Half-Life � Time it takes for half the atoms to decay � Specific to each radioactive element � the more radioactive an element the shorter the half-life

Artificial radiation � Occurs when the radioactive isotope was created in a lab

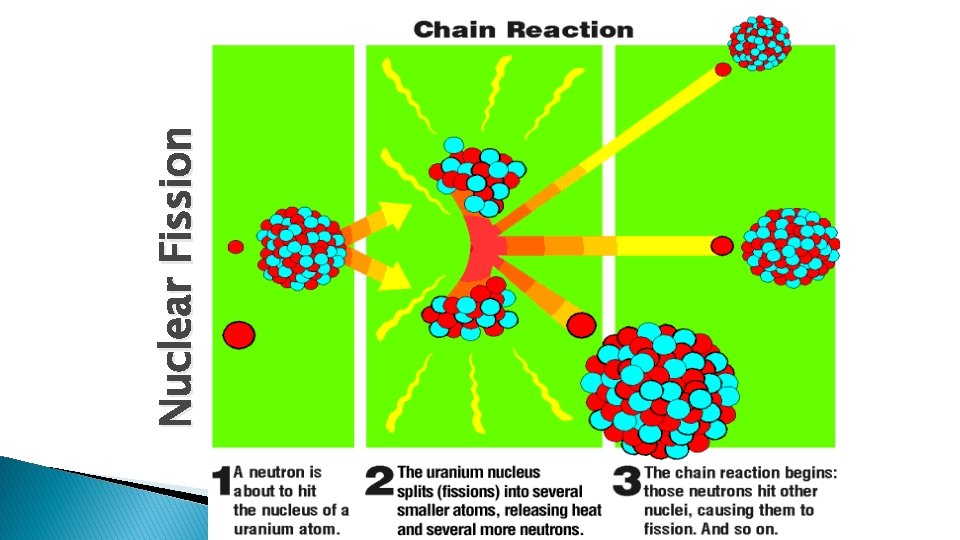
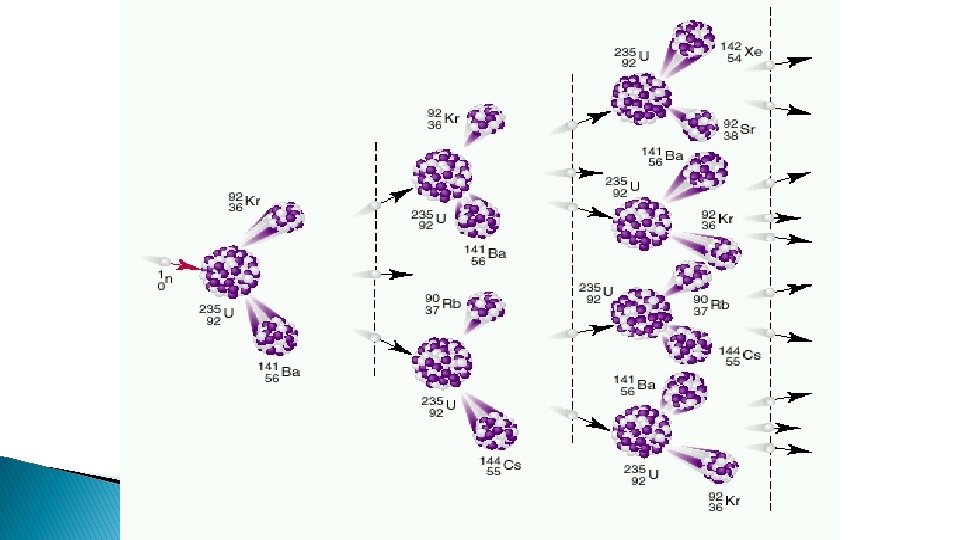
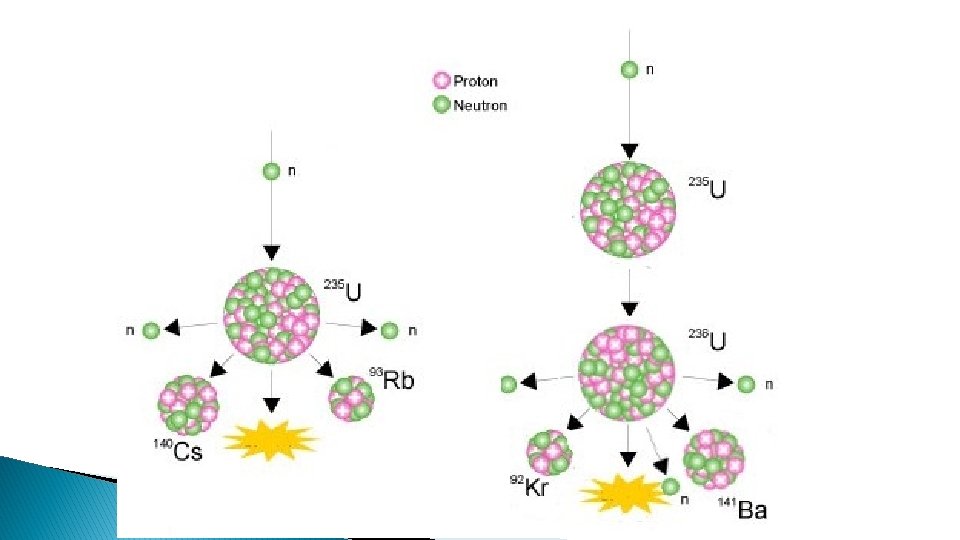
Nuclear Fission � Splits the nucleus � Can be done by bombarding the nucleus with particles usually neutrons

Nuclear Fission



� Only a few isotopes are fissionable ◦ E. g. Uranium-235, not Uranium 238 � chain reactions release radiation and energy

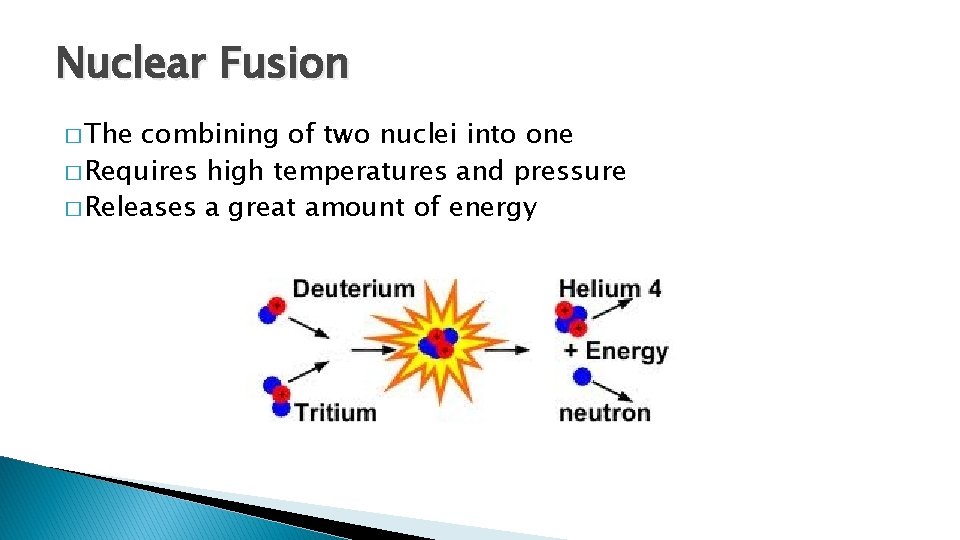
Nuclear Fusion � The combining of two nuclei into one � Requires high temperatures and pressure � Releases a great amount of energy

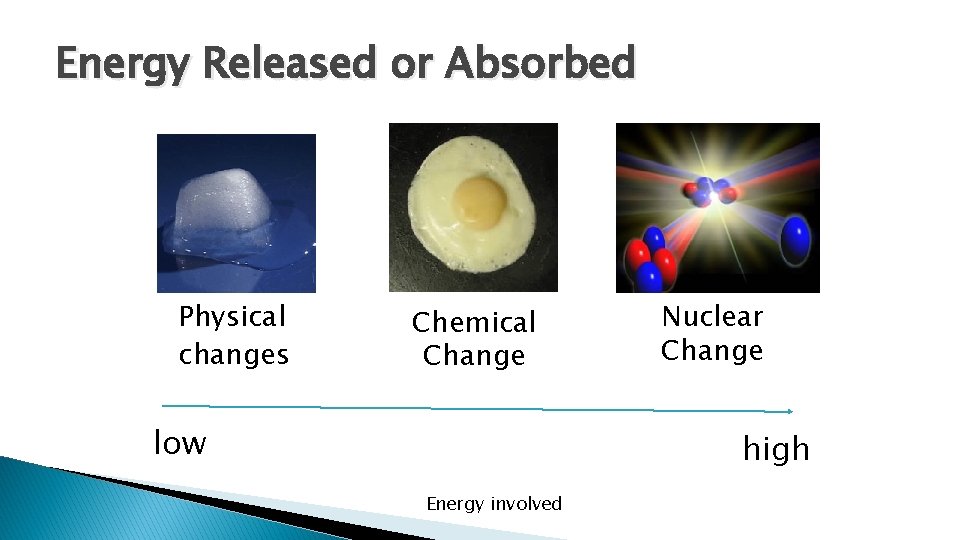
Energy Released or Absorbed Physical changes Chemical Change low Nuclear Change high Energy involved

Applications of radiation � Radiation therapy � Tracers for tumours � Food irradiation � Radiometric dating � Energy production

Medical Applications of atomic science �Radioisotopes = radioactive isotope ◦ Used to locate and destroy tumours

◦ Cobalt-60 most widely used to treat cancer �It releases gamma rays which are directed at the tumour, killing it. �Alternatively, a small dose can be implanted in the patient next to the tumor.

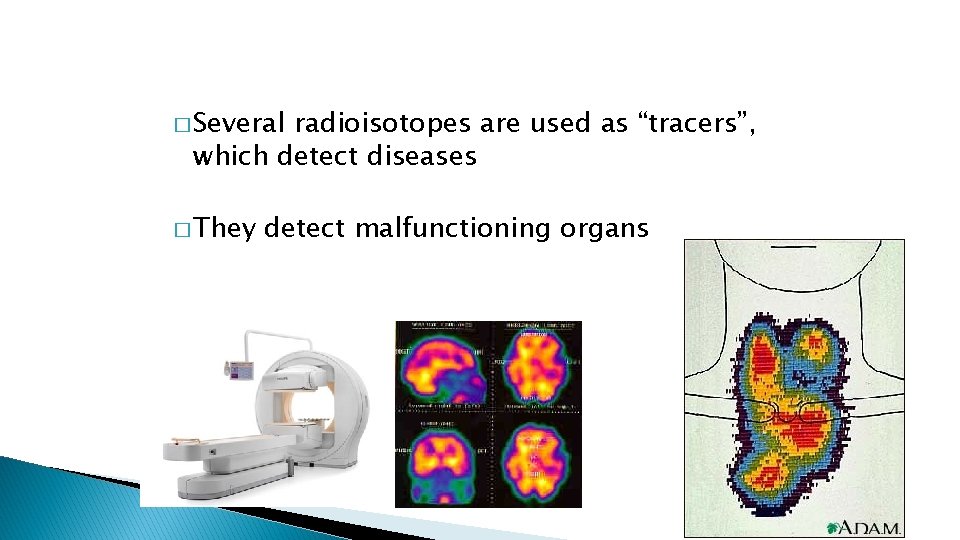
� Several radioisotopes are used as “tracers”, which detect diseases � They detect malfunctioning organs

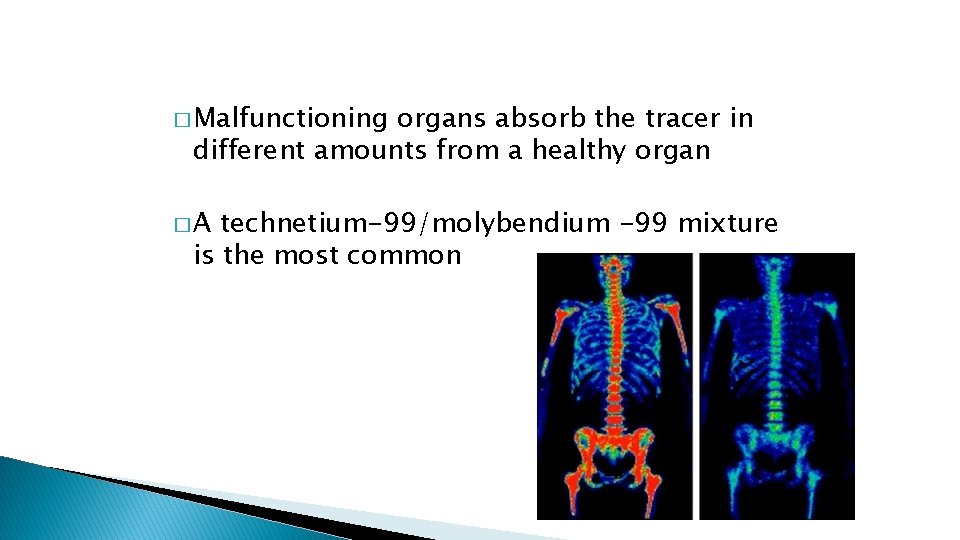
� Malfunctioning organs absorb the tracer in different amounts from a healthy organ �A technetium-99/molybendium -99 mixture is the most common

Irradiation � Expose � Gamma longer � Also food to gamma rays kill all bacteria on food, so food keeps used to sterilize surgical tools

Irradiation continued � Food � It does NOT become radioactive might change the nutritional value of food

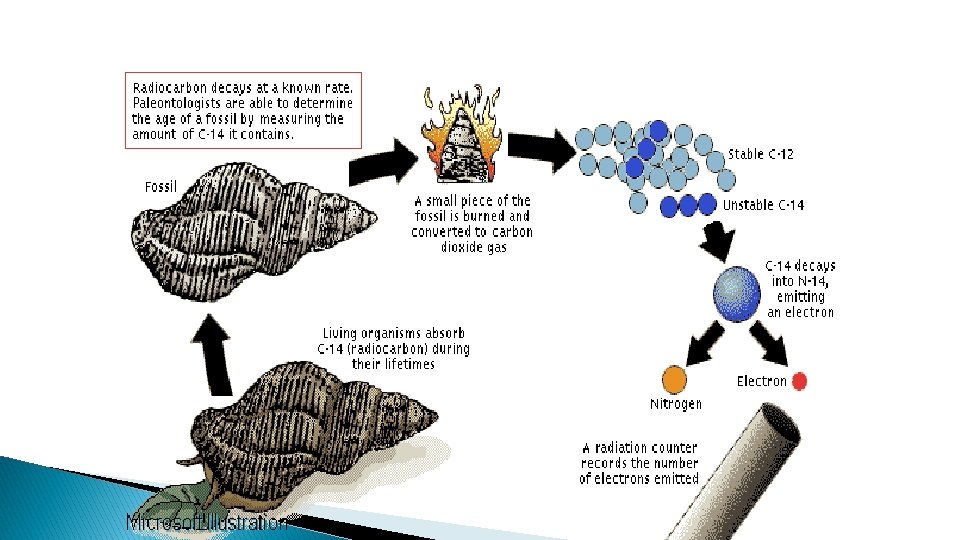
Carbon dating (with precision of 200 years. � Carbon-14 is found in all living things � Once something is dead, no more carbon-14 is taken up (eaten). � Carbon-14 � Can starts to decay into carbon-12 compare ratio of carbon-12 to carbon-14 to tell how long something has been dead using half-life.


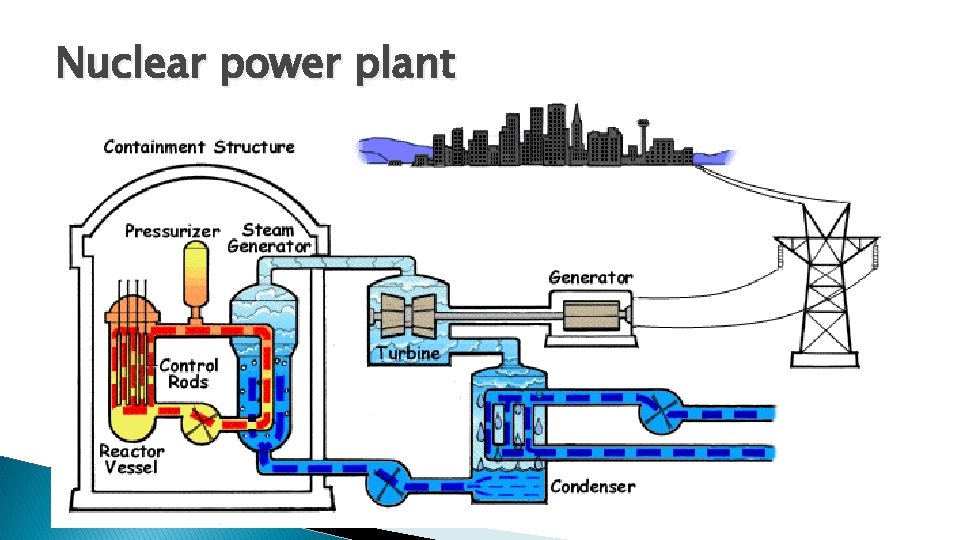
Energy production � 3 types of nuclear energy 1. Thermal power plant(fission) 2. Fusion reactor 3. Radioisotope thermoelectric generators.

Nuclear power plant


Nuclear Vessels � Submarines, ships, space ships � Vessels carry miniature power stations � Vessels have to refuel very rarely

Fusion & Plasma � The sun is undergoing fusion, creating our heat and light � Plasma happens at 3000°C � At this temperature, even gas breaks apart, becoming PLASMA � Plasma is made up of electrically charged ions and electrons moving fast

Fusion reactors – the Good � Releases fission � Use more 3 times more energy than deuterium and tritium to make helium � No radioactive waste � No chain reaction � Abundant fuel

Fusion reactors – The Bad � None operational for commercial purposes � A few experimental generators � Requires extremely high temperatures, melts all known substances

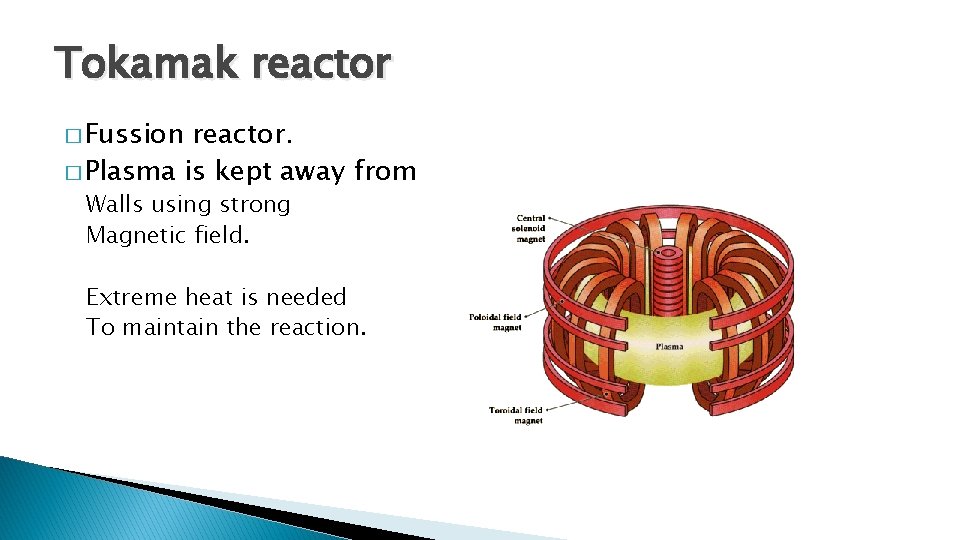
Tokamak reactor � Fussion reactor. � Plasma is kept away from Walls using strong Magnetic field. Extreme heat is needed To maintain the reaction.

Radioisotope thermoelectric generators �A battery source for spacecraft. � Often contains plutonium 238 which generates heat during decay. � A thermocouple generates heat into electricity.