Challenges for International Standardization and Traceability HORMONES Lothar

Challenges for International Standardization and Traceability - HORMONES _______________________ Lothar Siekmann Institute of Clinical Biochemisstry, University of Bonn Reference Institute of Bioanalysis, DGKL http: //www. dgkl-rfb. de
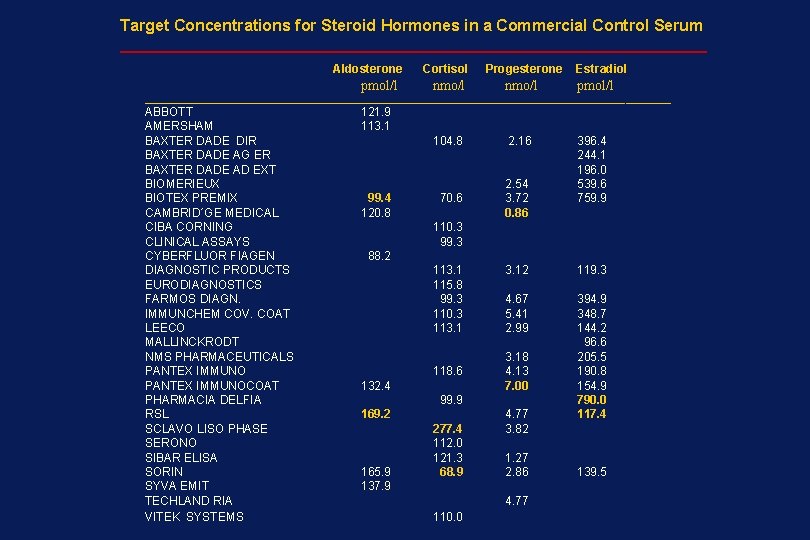
Target Concentrations for Steroid Hormones in a Commercial Control Serum _________________________________ Aldosterone pmol/l Cortisol Progesterone nmo/l Estradiol pmol/l _____________________________________________________ ABBOTT AMERSHAM BAXTER DADE DIR BAXTER DADE AG ER BAXTER DADE AD EXT BIOMERIEUX BIOTEX PREMIX CAMBRID´GE MEDICAL CIBA CORNING CLINICAL ASSAYS CYBERFLUOR FIAGEN DIAGNOSTIC PRODUCTS EURODIAGNOSTICS FARMOS DIAGN. IMMUNCHEM COV. COAT LEECO MALLINCKRODT NMS PHARMACEUTICALS PANTEX IMMUNOCOAT PHARMACIA DELFIA RSL SCLAVO LISO PHASE SERONO SIBAR ELISA SORIN SYVA EMIT TECHLAND RIA VITEK SYSTEMS 121. 9 113. 1 99. 4 120. 8 104. 8 2. 16 70. 6 2. 54 3. 72 0. 86 396. 4 244. 1 196. 0 539. 6 759. 9 110. 3 99. 3 88. 2 113. 1 115. 8 99. 3 110. 3 113. 1 118. 6 132. 4 3. 12 119. 3 4. 67 5. 41 2. 99 394. 9 348. 7 144. 2 96. 6 205. 5 190. 8 154. 9 790. 0 117. 4 3. 18 4. 13 7. 00 99. 9 169. 2 165. 9 137. 9 277. 4 112. 0 121. 3 68. 9 4. 77 3. 82 1. 27 2. 86 4. 77 110. 0 139. 5

In Vitro Diagnostica Directive European Union _____________________ “. . the traceability of values assigned to calibrators and control materials must be assured through available reference measurement procedures and/or reference materials of higher order. . . ”

Measurement Traceability ____________________ property of the result of a measurement or the value of a standard whereby it can be related to stated references, usually national or international standards, through an unbroken chain of comparisons all having stated uncertainties
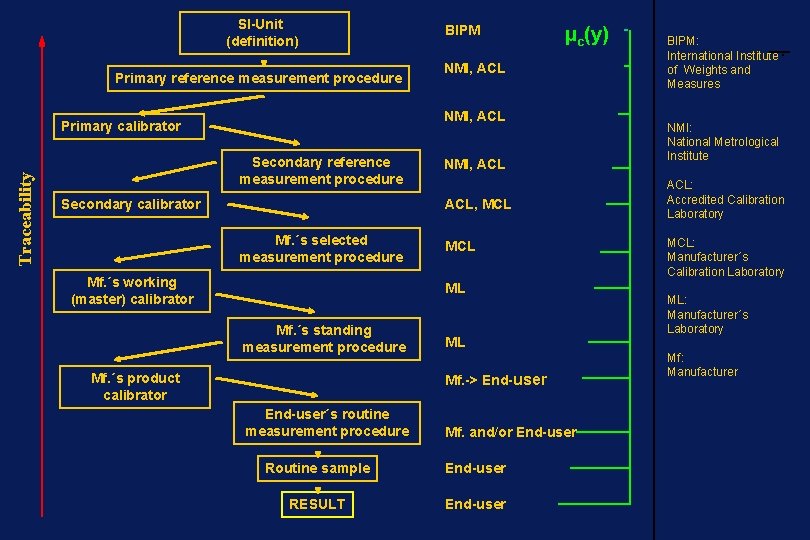
SI-Unit (definition) Primary reference measurement procedure µc(y) NMI, ACL Primary calibrator Traceability BIPM Secondary reference measurement procedure Secondary calibrator NMI, ACL, MCL Mf. ´s selected measurement procedure Mf. ´s working (master) calibrator MCL ML Mf. ´s standing measurement procedure Mf. ´s product calibrator ML Mf. -> End-user´s routine measurement procedure Mf. and/or End-user Routine sample End-user RESULT End-user BIPM: International Institute of Weights and Measures NMI: National Metrological Institute ACL: Accredited Calibration Laboratory MCL: Manufacturer´s Calibration Laboratory ML: Manufacturer´s Laboratory Mf: Manufacturer

Reference Systems Laboratory Medicine _______________________ • Reference Materials • • Reference Measurement Procedures Reference Laboratories
![MEASUREMENT OF ESTRADIOL-17ß BY ISOTOPE DILUTION MASS SPECTROMETRY ______________________________ SERUM Addition of [14 C]Estradiol-17ß MEASUREMENT OF ESTRADIOL-17ß BY ISOTOPE DILUTION MASS SPECTROMETRY ______________________________ SERUM Addition of [14 C]Estradiol-17ß](http://slidetodoc.com/presentation_image_h/984bebe128ae7bd93e4191c4d0da4439/image-8.jpg)
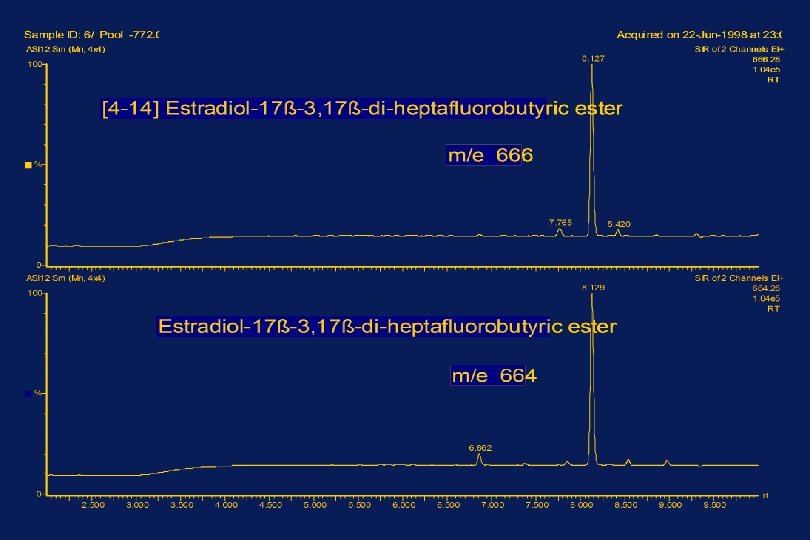
MEASUREMENT OF ESTRADIOL-17ß BY ISOTOPE DILUTION MASS SPECTROMETRY ______________________________ SERUM Addition of [14 C]Estradiol-17ß Extraction with Dichloromethane Column chromatography on Sephadex LH-20 Formation of the Heptafluorobutyric ester Selected Ion Recording at m/z 664 and 666 Calculation of the Estradiol-17ß Concentration from Isotope Ratios in Samples and Standard Mixtures
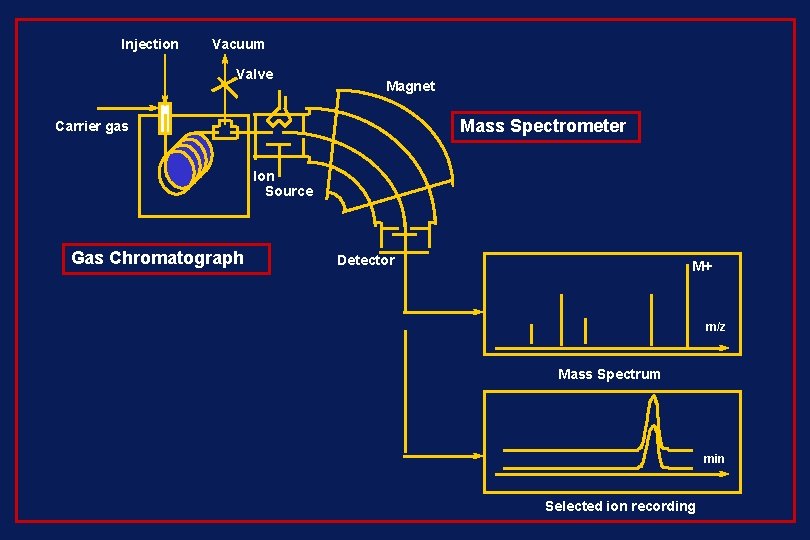
Injection Vacuum Valve Magnet Mass Spectrometer Carrier gas Ion Source Gas Chromatograph Detector M+ m/z Mass Spectrum min Selected ion recording


Implementation of the Traceability Concept ______________________ by: - Diagnostic Kit Manufacturers - Organisers of External Quality Assessment Schemes

DGKL Reference Procedures • • Electrolytes: Calcium Chloride Lithium Magnesium Potassium Sodium • Metabolites and Substrates: Cholesterol Creatinine Glucose Total Glycerol Uric Acid Urea Bilirubine Lactate • • Drugs: Theophylline Digoxin Digitoxin Enzmes; AST ALT CK Hormones: Aldosterone Cortisol Estradiol-17ß Estriol Progesterone 17 -Hydroxy-progesterone Testosterone Thyroxine GGT AMYLASE • Total Protein
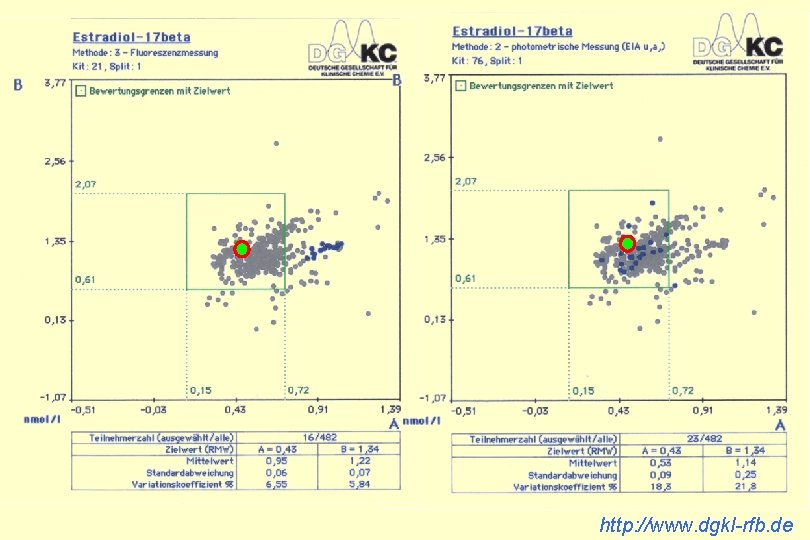
http: //www. dgkl-rfb. de
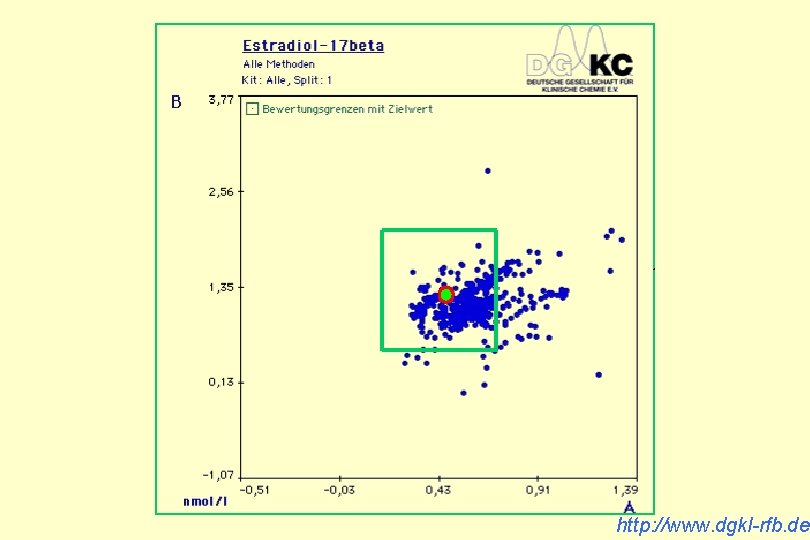
http: //www. dgkl-rfb. de

http: //www. dgkl-rfb. de
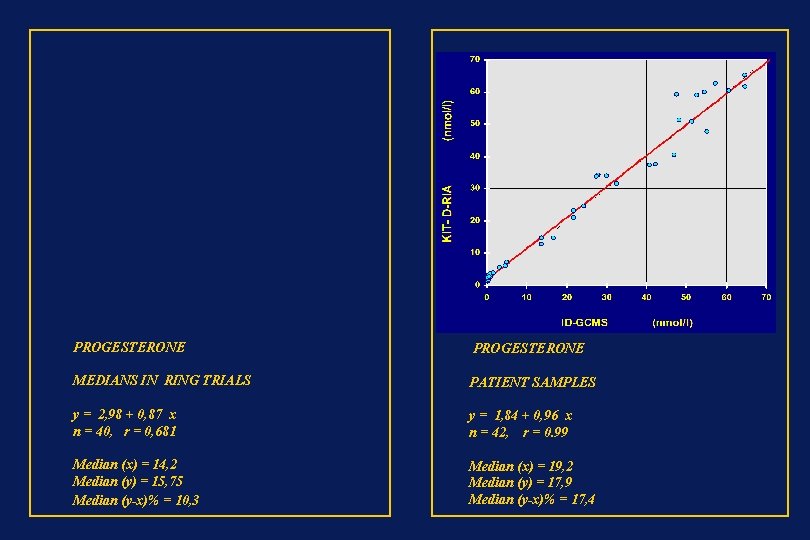
PROGESTERONE MEDIANS IN RING TRIALS PATIENT SAMPLES y = 2, 98 + 0, 87 x n = 40, r = 0, 681 y = 1, 84 + 0, 96 x n = 42, r = 0. 99 Median (x) = 14, 2 Median (y) = 15, 75 Median (y-x)% = 10, 3 Median (x) = 19, 2 Median (y) = 17, 9 Median (y-x)% = 17, 4
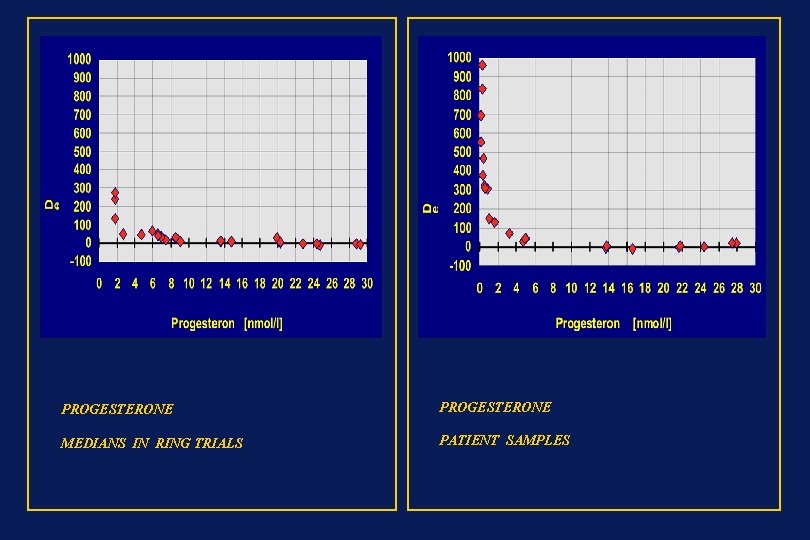
PROGESTERONE MEDIANS IN RING TRIALS PATIENT SAMPLES
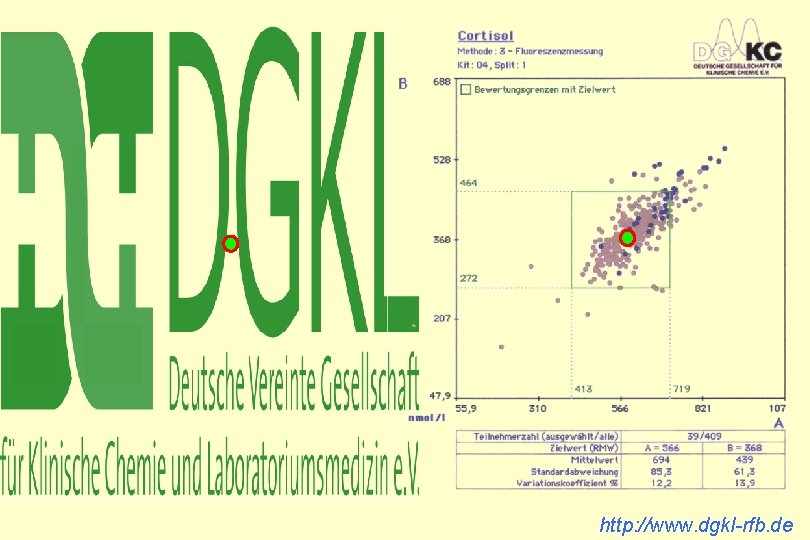
http: //www. dgkl-rfb. de
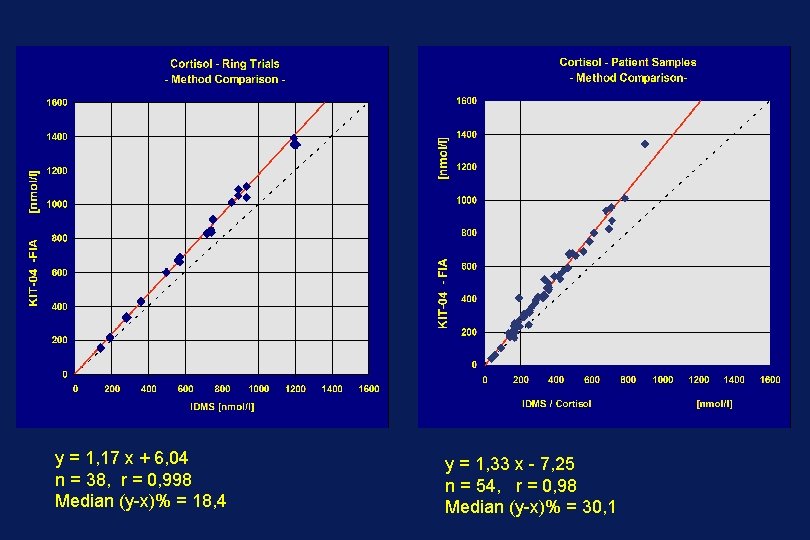
y = 1, 17 x + 6, 04 n = 38, r = 0, 998 Median (y-x)% = 18, 4 y = 1, 33 x - 7, 25 n = 54, r = 0, 98 Median (y-x)% = 30, 1
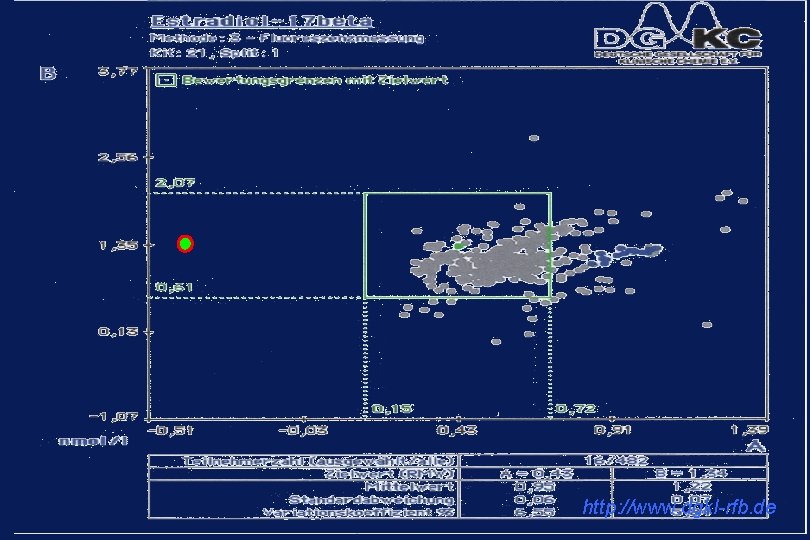
http: //www. dgkl-rfb. de
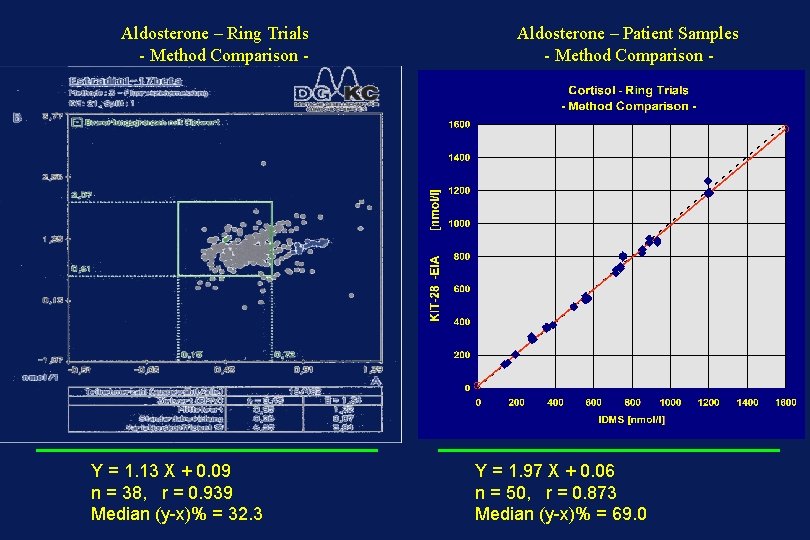
Aldosterone – Ring Trials - Method Comparison - Y = 1. 13 X + 0. 09 n = 38, r = 0. 939 Median (y-x)% = 32. 3 Aldosterone – Patient Samples - Method Comparison - Y = 1. 97 X + 0. 06 n = 50, r = 0. 873 Median (y-x)% = 69. 0
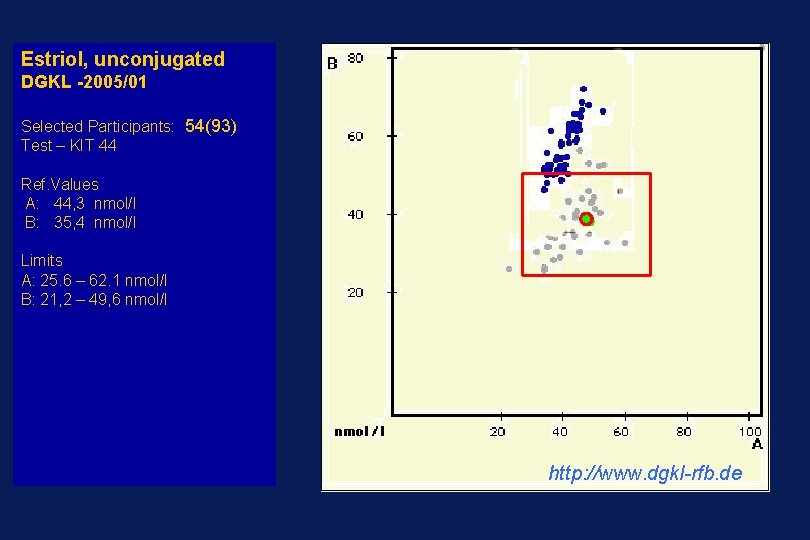
Estriol, unconjugated DGKL -2005/01 Selected Participants: 54(93) Test – KIT 44 Ref. Values A: 44, 3 nmol/l B: 35, 4 nmol/l Limits A: 25. 6 – 62. 1 nmol/l B: 21, 2 – 49, 6 nmol/l http: //www. dgkl-rfb. de
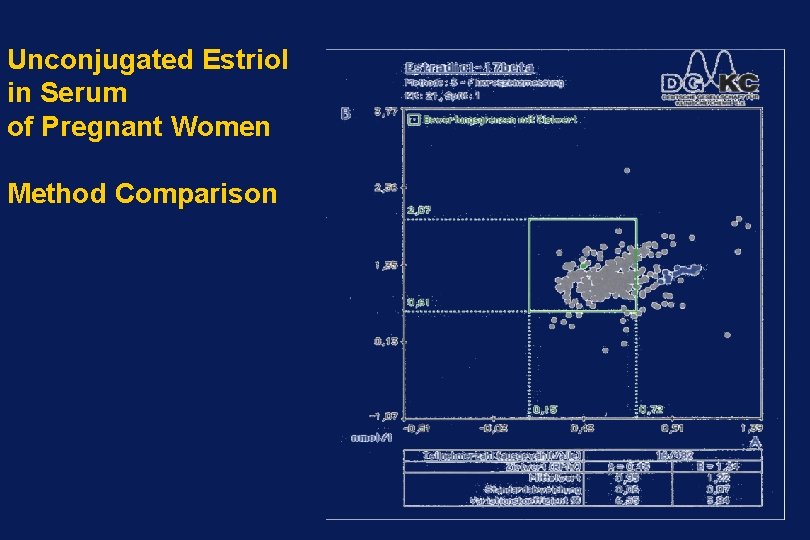
Unconjugated Estriol in Serum of Pregnant Women Method Comparison
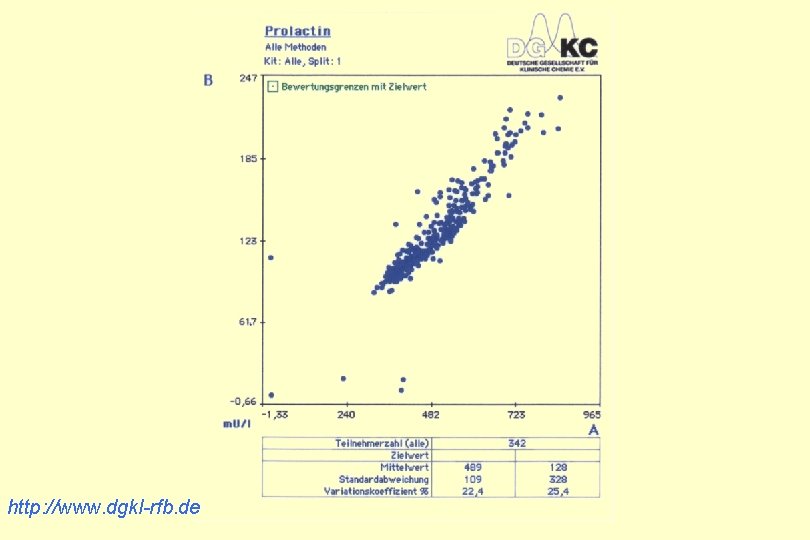
http: //www. dgkl-rfb. de
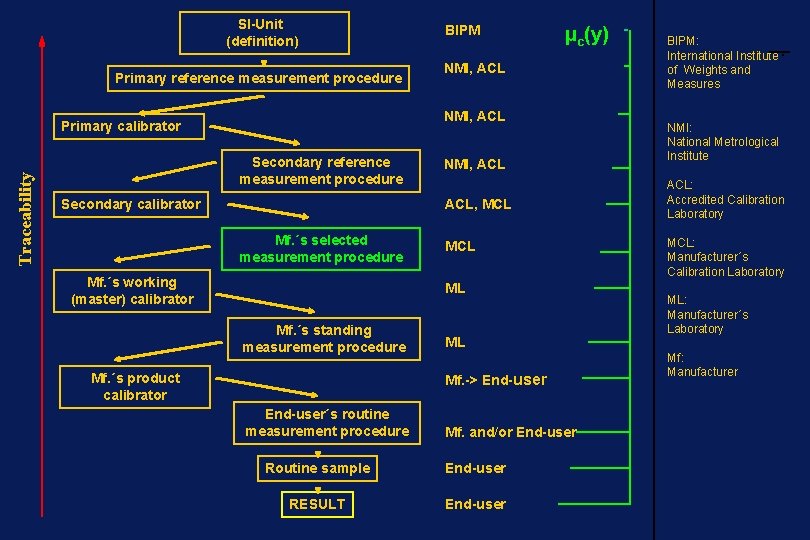
SI-Unit (definition) Primary reference measurement procedure µc(y) NMI, ACL Primary calibrator Traceability BIPM Secondary reference measurement procedure Secondary calibrator NMI, ACL, MCL Mf. ´s selected measurement procedure Mf. ´s working (master) calibrator MCL ML Mf. ´s standing measurement procedure Mf. ´s product calibrator ML Mf. -> End-user´s routine measurement procedure Mf. and/or End-user Routine sample End-user RESULT End-user BIPM: International Institute of Weights and Measures NMI: National Metrological Institute ACL: Accredited Calibration Laboratory MCL: Manufacturer´s Calibration Laboratory ML: Manufacturer´s Laboratory Mf: Manufacturer
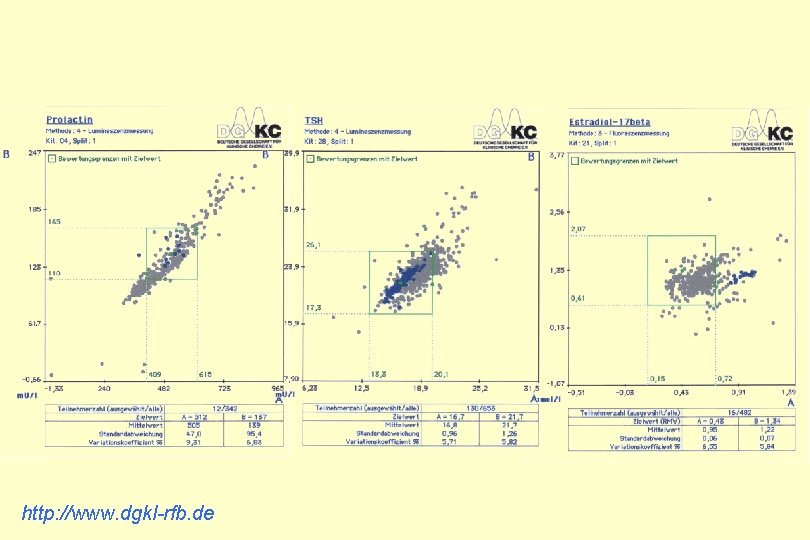
http: //www. dgkl-rfb. de
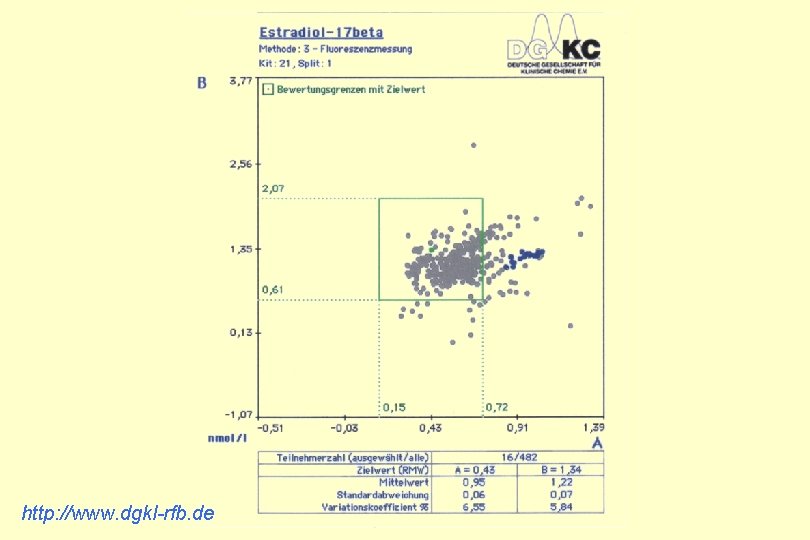
http: //www. dgkl-rfb. de
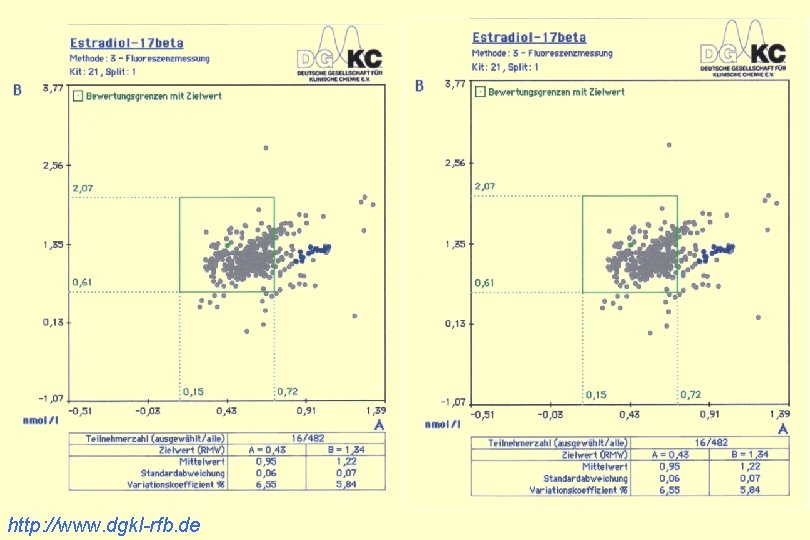
http: //www. dgkl-rfb. de
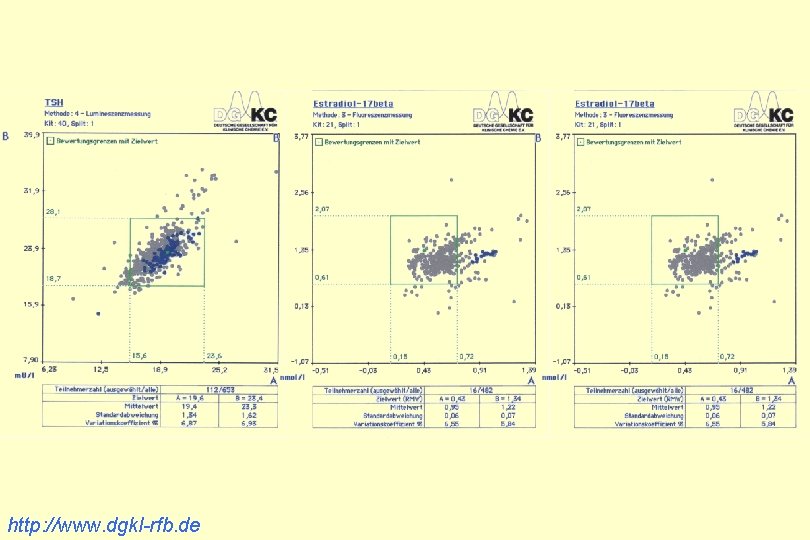
http: //www. dgkl-rfb. de

Strategy to establish Reference Systems for non-SI Traceable Quantities ______________________ • Definition of the Analyte • Development of a Reference Procedure • Development of a Reference Material

Definition of Proteo-Hormone Analytes _____________________ • Decision on the sub-unit to be measured according to clinical relevance • Decision on the epitope(s) to be detected by mono- or poli-clonal antibodies • Decision on the glycosidic structure to be measured


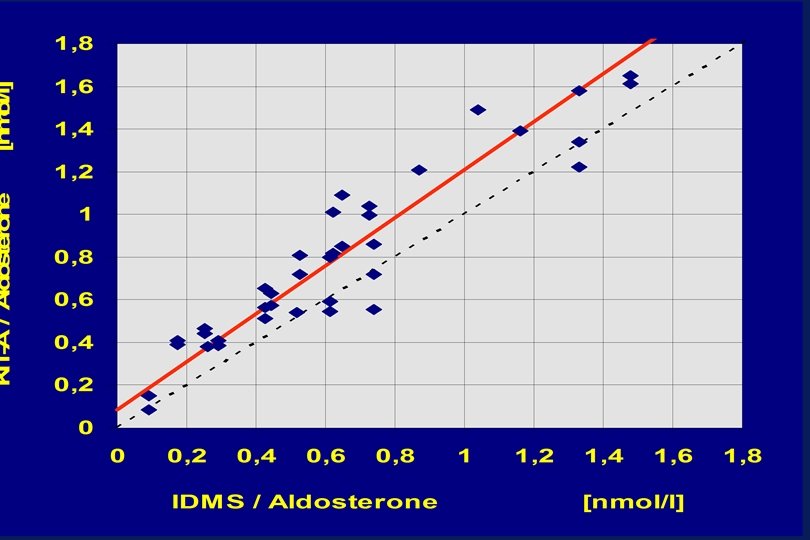
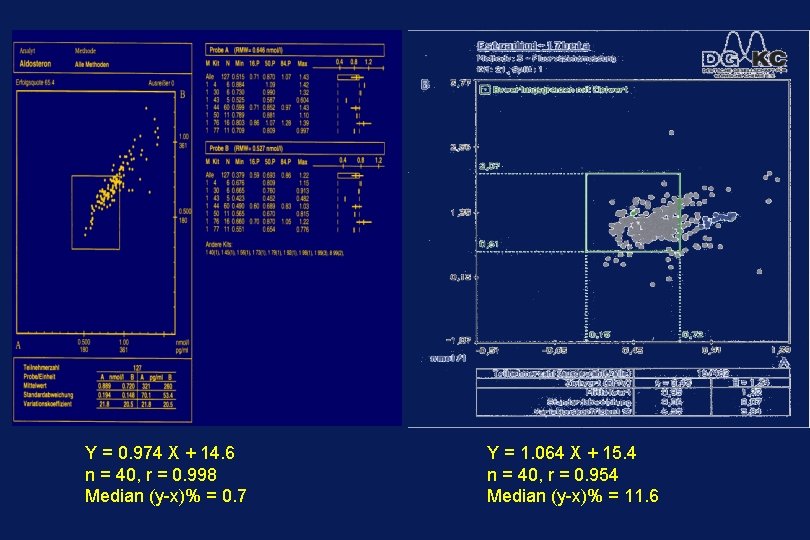
Y = 0. 974 X + 14. 6 n = 40, r = 0. 998 Median (y-x)% = 0. 7 Y = 1. 064 X + 15. 4 n = 40, r = 0. 954 Median (y-x)% = 11. 6
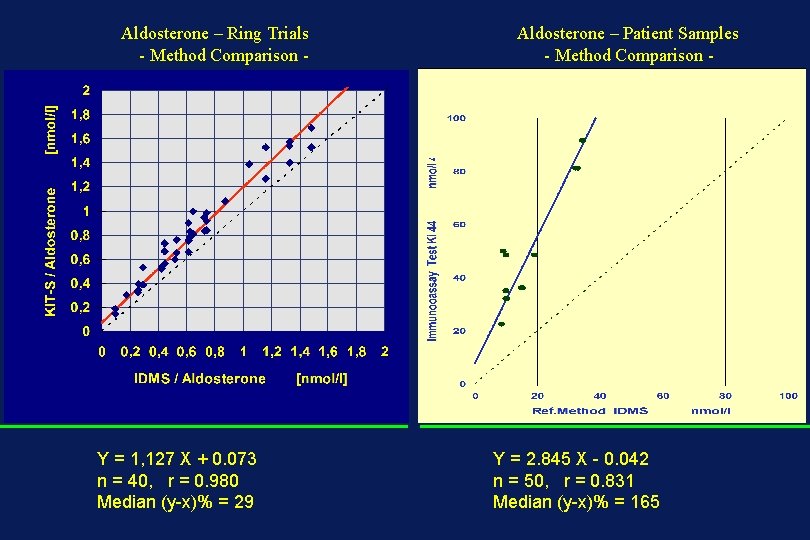
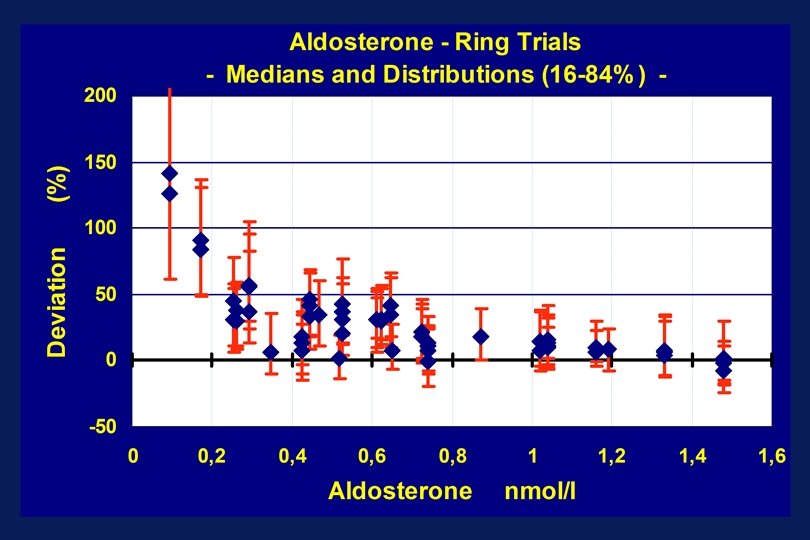
Aldosterone – Ring Trials - Method Comparison - Y = 1, 127 X + 0. 073 n = 40, r = 0. 980 Median (y-x)% = 29 Aldosterone – Patient Samples - Method Comparison - Y = 2. 845 X - 0. 042 n = 50, r = 0. 831 Median (y-x)% = 165
- Slides: 39