Challenges for Blood Donor Confirmatory Testing Algorithms Susan

Challenges for Blood Donor Confirmatory Testing Algorithms Susan L. Stramer, Ph. D. American Red Cross, for HIV Diagnostics: New Developments and Challenges Meeting, Orlando, FL; Feb 28 -March 1, 2005 E 0506225 A 1

Background u HIV-1 algorithms utilize immunoassays for confirmation of repeat reactive blood donors – Most commonly used: Western Blot (WB) • Contains electrophoresed whole HIV lysate – HIV-1 Immunofluorescence (also uses viral lysate) • Not widely used in blood centers due to difficulty in interpretation – HIV-2 confirmation also required for HIV-1 WB ind/neg samples • Licensed HIV-2 EIA => Research based HIV-2 WB (CA) – 6 or fewer HIV-2 confirmed pos blood donors in the US reported since 1992 – No confirmation for HIV-1 type O E 0506225 A 2

Background u Issues with HIV confirmatory assays – Poor performance • Unreadable, uninterpretable or invalid results generated • Misclassification of donors – False positive and false negative results • High rates of indeterminate results (neither pos or neg) in healthy individuals – Donor deferral, anxiety, and use of complicated donor reentry algorithms, when available – Mixed message, “You are healthy and not HIV infected, but you cannot donate blood” E 0506225 A 3

Background u Issues with HIV confirmatory assays, cont’d – High costs – Inconsistent availability – No new products for “antibody confirmation” on the market for HIV-1 or HIV-2 – Screening tests ongoing improvements since 1985 • Costs associated with new products extremely high u Can alternate algorithms be validated? – HIV RNA (NAT), use of high/low screening S/CO ratios, dual EIA algorithm E 0506225 A 4

Can Alternate Algorithms be Implemented u Kits will require labeling for intended use – 21 CFR 610. 40 Subpart E • “You must further test each donation, including autologous donations, found to be reactive by a screening test …whenever a supplemental (additional, more specific) test has been approved for such use by FDA…. ” – FDA’s current thinking is that an antibody RR must be confirmed by an antibody-specific assay • Variances have been granted to exempt RNA pos samples from requiring WB – Minipool or single donor RNA neg samples still require WB E 0506225 A 5
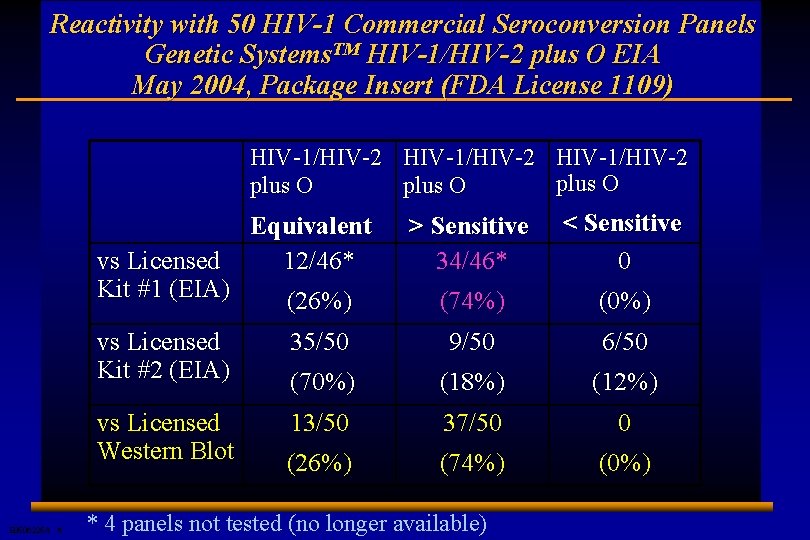
Reactivity with 50 HIV-1 Commercial Seroconversion Panels Genetic Systems. TM HIV-1/HIV-2 plus O EIA May 2004, Package Insert (FDA License 1109) HIV-1/HIV-2 plus O > Sensitive 34/46* < Sensitive 0 (26%) (74%) (0%) vs Licensed Kit #2 (EIA) 35/50 9/50 6/50 (70%) (18%) (12%) vs Licensed Western Blot 13/50 37/50 0 (26%) (74%) (0%) vs Licensed Kit #1 (EIA) E 0506225 A 6 Equivalent 12/46* * 4 panels not tested (no longer available)

Cambridge Biotech Human Immunodeficiency Virus Type 1 (HIV -1) Western Blot Kit March 16, 2000 BPAC u Any bands present but pattern does not meet criteria for POSITIVE = INDETERMINATE u Non-viral bands have been observed with certain specimens. These bands are not usually accompanied by any of the major viral bands of diagnostic significance (p 24, gp 41/120/160). The non-viral bands appear to be cell related with the most common in the molecular weight range of 70 K, 51 -55 K (possible HLA-DR) and 43 K (possible HLA-ABC). E 0506225 A 7
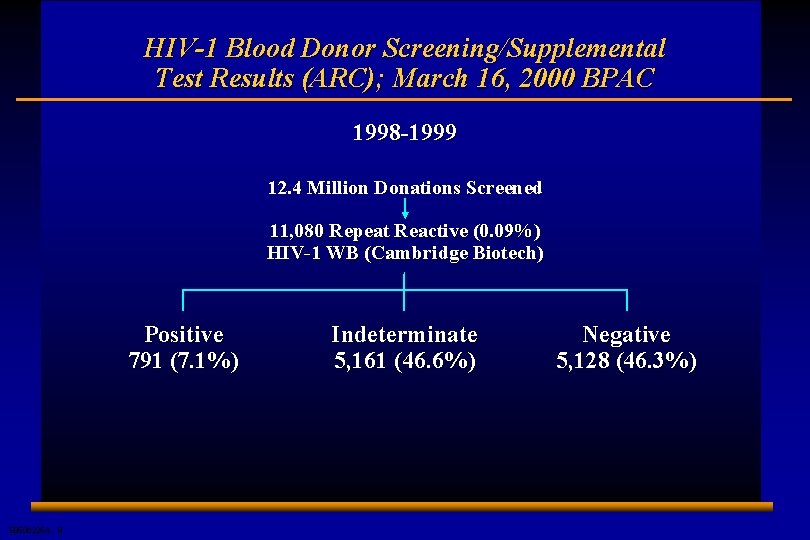
HIV-1 Blood Donor Screening/Supplemental Test Results (ARC); March 16, 2000 BPAC 1998 -1999 12. 4 Million Donations Screened 11, 080 Repeat Reactive (0. 09%) HIV-1 WB (Cambridge Biotech) Positive 791 (7. 1%) E 0506225 A 8 Indeterminate 5, 161 (46. 6%) Negative 5, 128 (46. 3%)
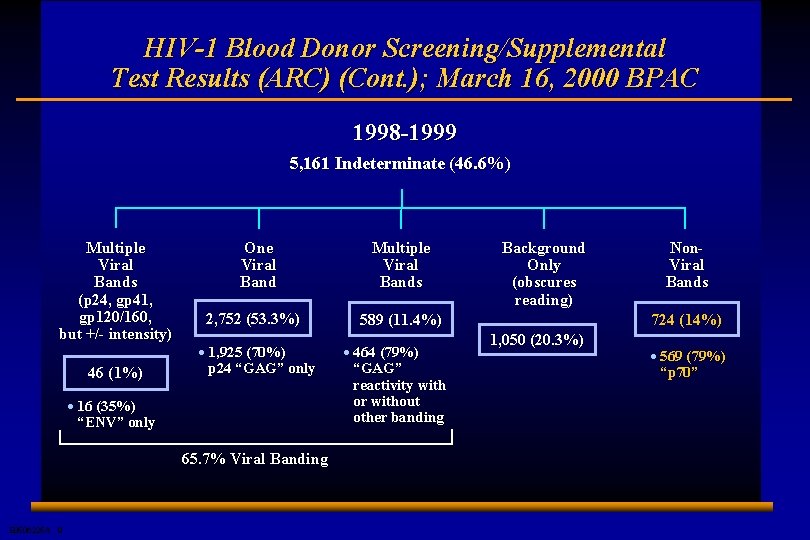
HIV-1 Blood Donor Screening/Supplemental Test Results (ARC) (Cont. ); March 16, 2000 BPAC 1998 -1999 5, 161 Indeterminate (46. 6%) Multiple Viral Bands (p 24, gp 41, gp 120/160, but +/- intensity) 46 (1%) One Viral Band 2, 752 (53. 3%) · 1, 925 (70%) p 24 “GAG” only · 16 (35%) “ENV” only 65. 7% Viral Banding E 0506225 A 9 Multiple Viral Bands Background Only (obscures reading) 589 (11. 4%) · 464 (79%) “GAG” reactivity with or without other banding Non. Viral Bands 724 (14%) 1, 050 (20. 3%) · 569 (79%) “p 70”
Calypte Biotech HIV-1 Western Blot gp 160 gp 120 p 66 p 55/51 gp 41 p 31 p 24 p 17 E 0506225 A 10
Calypte HIV-1 Western Blot gp 160 gp 120 p 66 p 55/51 gp 41 p 31 p 24 p 17 E 0506225 A 11
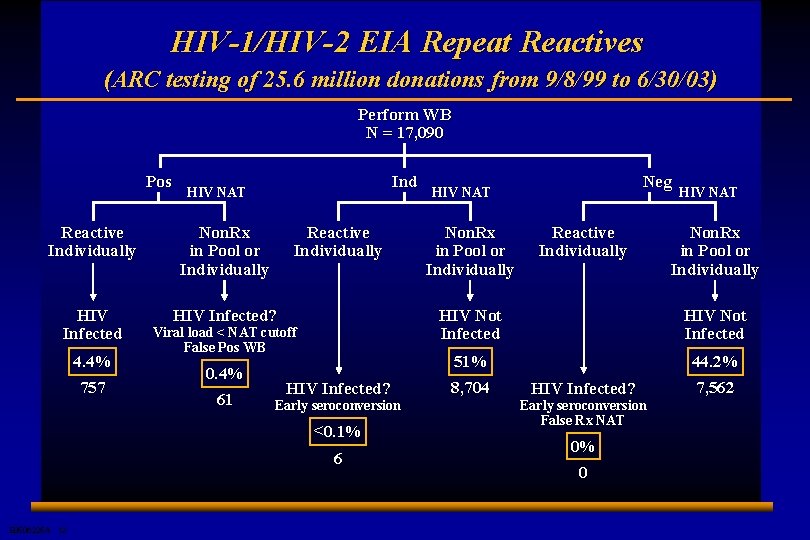
HIV-1/HIV-2 EIA Repeat Reactives (ARC testing of 25. 6 million donations from 9/8/99 to 6/30/03) Perform WB N = 17, 090 Pos Ind HIV NAT Reactive Individually Non. Rx in Pool or Individually HIV Infected? 4. 4% 757 Reactive Individually Viral load < NAT cutoff False Pos WB 0. 4% 61 HIV Infected? Early seroconversion <0. 1% 6 E 0506225 A 12 Neg HIV NAT Non. Rx in Pool or Individually Reactive Individually HIV NAT Non. Rx in Pool or Individually HIV Not Infected 51% 44. 2% 8, 704 HIV Infected? Early seroconversion False Rx NAT 0% 0 7, 562

HIV-1/HIV-2 EIA Repeat Reactives (ARC testing of 25. 6 million donations from 9/8/99 to 6/30/03) Review HIV-1 NAT Results N = 17, 090 Pool or Individual Unit NAT Non. Rx Individual Unit NAT Rx 96% 4% 16, 327 763 Perform WB No Further Testing HIV Infected (16 dtns ® HIV-1/HCV TMA) Pos 0. 4% Ind 53. 3% Neg 46. 3% 61 8, 704 7, 562 (d. HIV TMA) 99. 2% PPV 757 WB Pos 92. 5% Sensitivity E 0506225 A 13
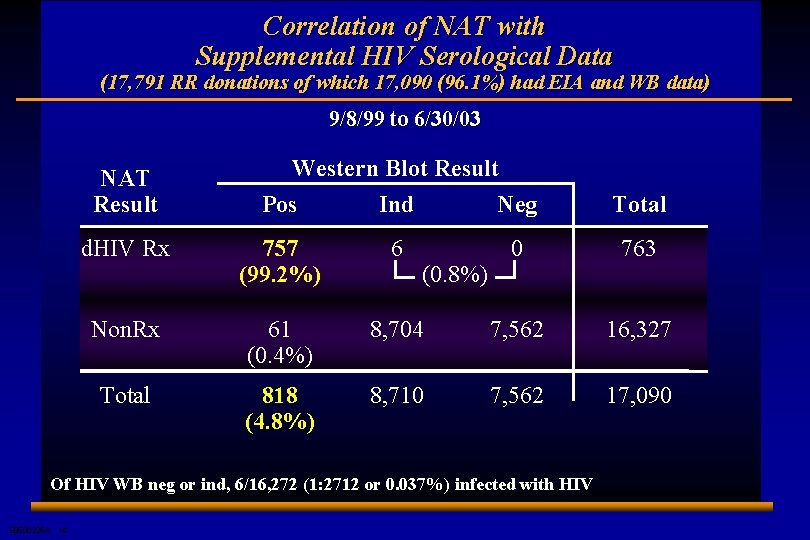
Correlation of NAT with Supplemental HIV Serological Data (17, 791 RR donations of which 17, 090 (96. 1%) had EIA and WB data) 9/8/99 to 6/30/03 NAT Result Western Blot Result Pos Ind Neg Total d. HIV Rx 757 (99. 2%) 6 0 763 Non. Rx 61 (0. 4%) 8, 704 7, 562 16, 327 Total 818 (4. 8%) 8, 710 7, 562 17, 090 (0. 8%) Of HIV WB neg or ind, 6/16, 272 (1: 2712 or 0. 037%) infected with HIV E 0506225 A 14
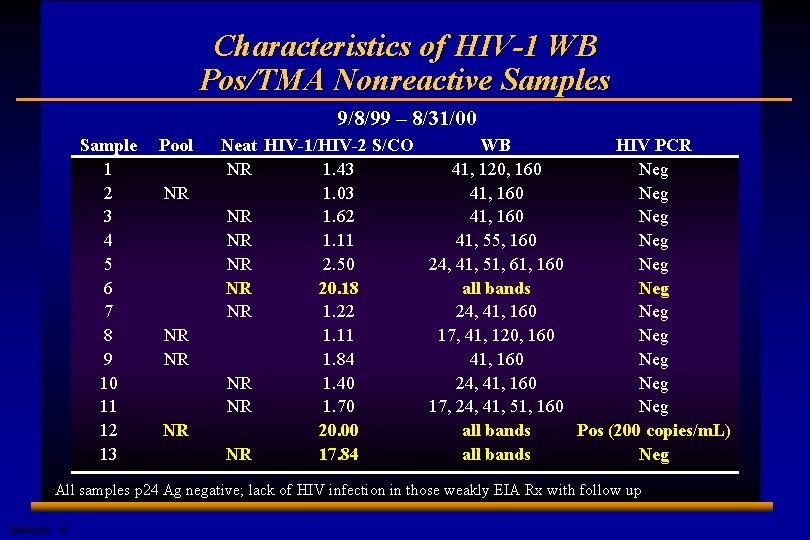
Characteristics of HIV-1 WB Pos/TMA Nonreactive Samples 9/8/99 – 8/31/00 Sample 1 2 3 4 5 6 7 8 9 10 11 12 13 Pool NR NR Neat HIV-1/HIV-2 S/CO WB HIV PCR NR 1. 43 41, 120, 160 Neg 1. 03 41, 160 Neg NR 1. 62 41, 160 Neg NR 1. 11 41, 55, 160 Neg NR 2. 50 24, 41, 51, 61, 160 Neg NR 20. 18 all bands Neg NR 1. 22 24, 41, 160 Neg 1. 11 17, 41, 120, 160 Neg 1. 84 41, 160 Neg NR 1. 40 24, 41, 160 Neg NR 1. 70 17, 24, 41, 51, 160 Neg 20. 00 all bands Pos (200 copies/m. L) NR 17. 84 all bands Neg All samples p 24 Ag negative; lack of HIV infection in those weakly EIA Rx with follow up E 0506225 A 15
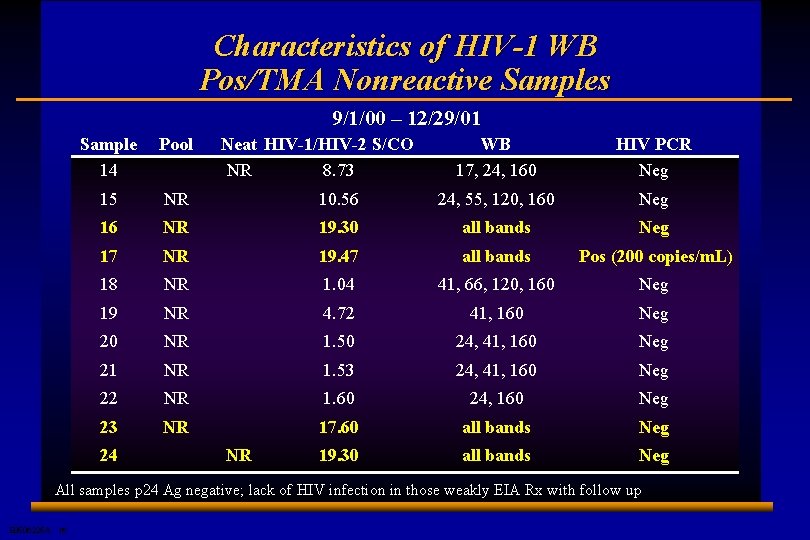
Characteristics of HIV-1 WB Pos/TMA Nonreactive Samples 9/1/00 – 12/29/01 Sample 14 Pool 15 NR 16 WB 17, 24, 160 HIV PCR Neg 10. 56 24, 55, 120, 160 Neg NR 19. 30 all bands Neg 17 NR 19. 47 all bands Pos (200 copies/m. L) 18 NR 1. 04 41, 66, 120, 160 Neg 19 NR 4. 72 41, 160 Neg 20 NR 1. 50 24, 41, 160 Neg 21 NR 1. 53 24, 41, 160 Neg 22 NR 1. 60 24, 160 Neg 23 NR 17. 60 all bands Neg 19. 30 all bands Neg 24 Neat HIV-1/HIV-2 S/CO NR 8. 73 NR All samples p 24 Ag negative; lack of HIV infection in those weakly EIA Rx with follow up E 0506225 A 16
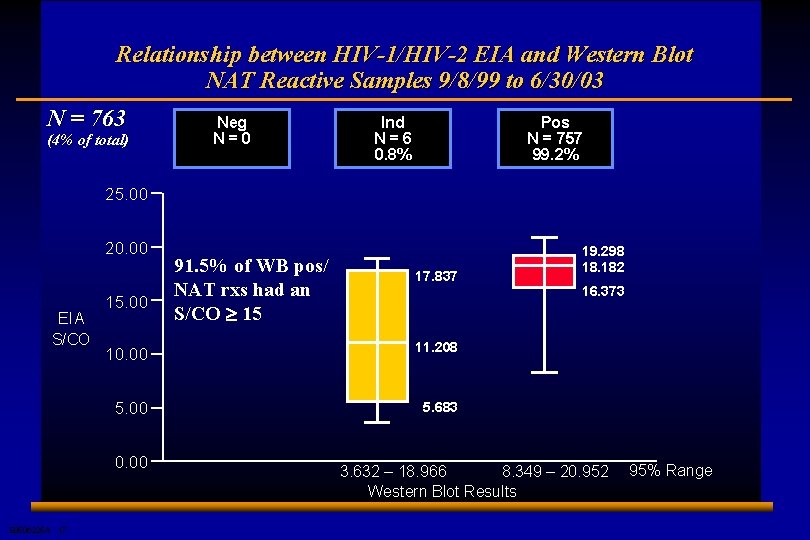
Relationship between HIV-1/HIV-2 EIA and Western Blot NAT Reactive Samples 9/8/99 to 6/30/03 N = 763 (4% of total) Neg N=0 Ind N=6 0. 8% Pos N = 757 99. 2% 25. 00 20. 00 EIA S/CO 15. 00 17. 837 10. 00 11. 208 5. 00 5. 683 0. 00 E 0506225 A 17 91. 5% of WB pos/ NAT rxs had an S/CO ³ 15 19. 298 18. 182 16. 373 3. 632 – 18. 966 8. 349 – 20. 952 Western Blot Results 95% Range
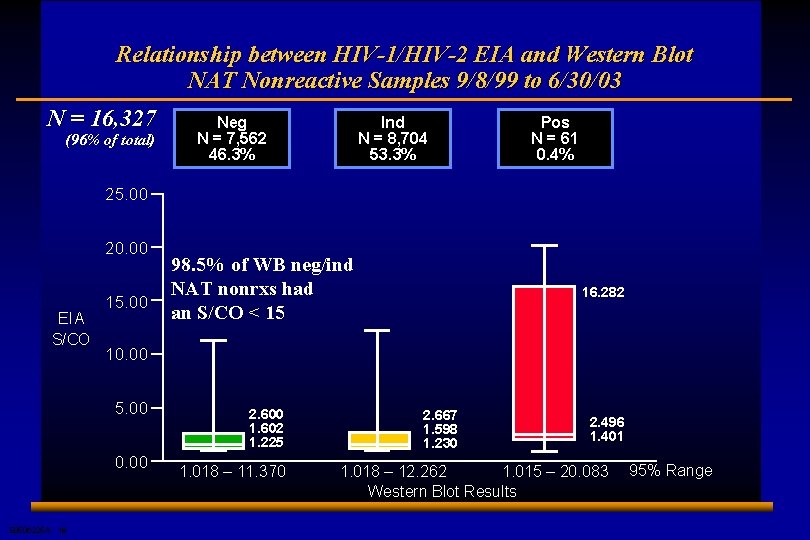
Relationship between HIV-1/HIV-2 EIA and Western Blot NAT Nonreactive Samples 9/8/99 to 6/30/03 N = 16, 327 (96% of total) Neg N = 7, 562 46. 3% Ind N = 8, 704 53. 3% Pos N = 61 0. 4% 25. 00 20. 00 EIA S/CO 15. 00 16. 282 10. 00 5. 00 0. 00 E 0506225 A 18 98. 5% of WB neg/ind NAT nonrxs had an S/CO < 15 2. 600 1. 602 1. 225 1. 018 – 11. 370 2. 667 1. 598 1. 230 2. 496 1. 401 1. 018 – 12. 262 1. 015 – 20. 083 Western Blot Results 95% Range
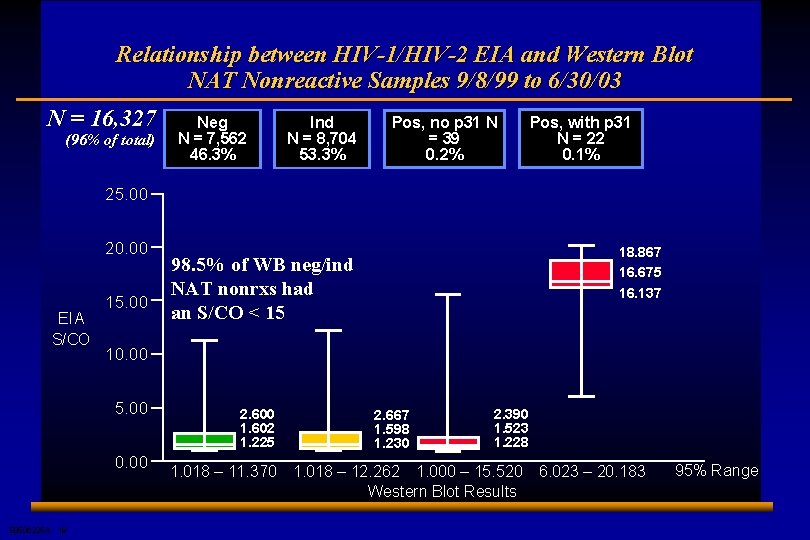
Relationship between HIV-1/HIV-2 EIA and Western Blot NAT Nonreactive Samples 9/8/99 to 6/30/03 N = 16, 327 (96% of total) Neg N = 7, 562 46. 3% Ind N = 8, 704 53. 3% Pos, no p 31 N = 39 0. 2% Pos, with p 31 N = 22 0. 1% 25. 00 20. 00 EIA S/CO 15. 00 10. 00 5. 00 0. 00 E 0506225 A 19 18. 867 16. 675 16. 137 98. 5% of WB neg/ind NAT nonrxs had an S/CO < 15 2. 600 1. 602 1. 225 1. 018 – 11. 370 2. 667 1. 598 1. 230 2. 390 1. 523 1. 228 1. 018 – 12. 262 1. 000 – 15. 520 Western Blot Results 6. 023 – 20. 183 95% Range

Dual EIA Algorithm u Feasibility based on the concept: – If two assays with comparable sensitivity are composed of differing rare reagents and have a different format, the false positive populations should have limited cross over; the more unique the tests, the greater the separation of false positive populations u Used successfully for HTLV to eliminate >60% of repeat reactives requiring further testing by expensive, complicated, error prone and unavailable/unlicensed tests E 0506225 A 20

HIV Dual EIA Algorithm Qualification u Qualified in both directions based on the two FDA licensed HIV-1/HIV-2 EIAs: – Abbott (EIA-1) ® Genetic Systems (EIA-2) (ARC) – Genetic Systems (EIA-1) ® Abbott (EIA-2) (BSL) u All HIV EIA repeat reactive samples from 1/1/00 -3/31/02 having adequate volume for additional testing were evaluated (N=7884); allogeneic donors u Western Blot and NAT (TMA pools of 16) test-of-record data were used for analysis u All 2 nd EIA testing was performed centrally at Blood Systems Laboratories (BSL) E 0506225 A 21
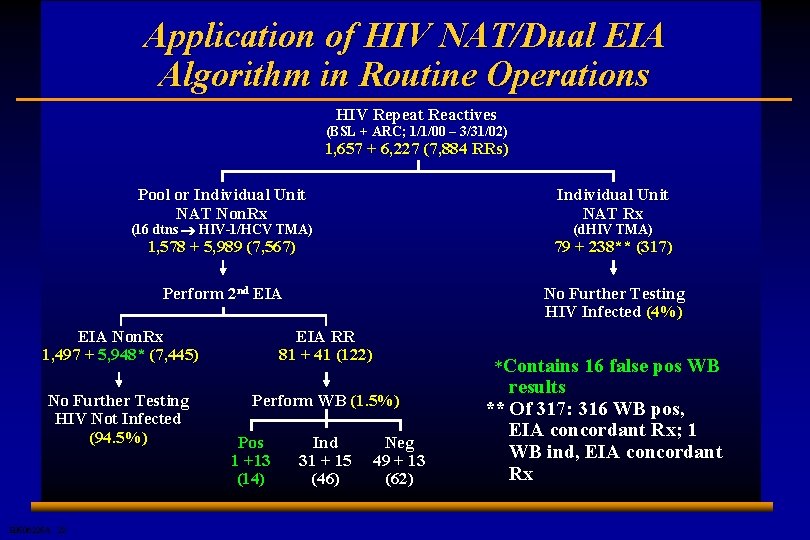
Application of HIV NAT/Dual EIA Algorithm in Routine Operations HIV Repeat Reactives (BSL + ARC; 1/1/00 – 3/31/02) 1, 657 + 6, 227 (7, 884 RRs) Pool or Individual Unit NAT Non. Rx Individual Unit NAT Rx 1, 578 + 5, 989 (7, 567) 79 + 238** (317) Perform 2 nd EIA No Further Testing HIV Infected (4%) (16 dtns ® HIV-1/HCV TMA) (d. HIV TMA) EIA Non. Rx 1, 497 + 5, 948* (7, 445) EIA RR 81 + 41 (122) No Further Testing HIV Not Infected (94. 5%) Perform WB (1. 5%) E 0506225 A 22 Pos 1 +13 (14) Ind 31 + 15 (46) Neg 49 + 13 (62) *Contains 16 false pos WB results ** Of 317: 316 WB pos, EIA concordant Rx; 1 WB ind, EIA concordant Rx
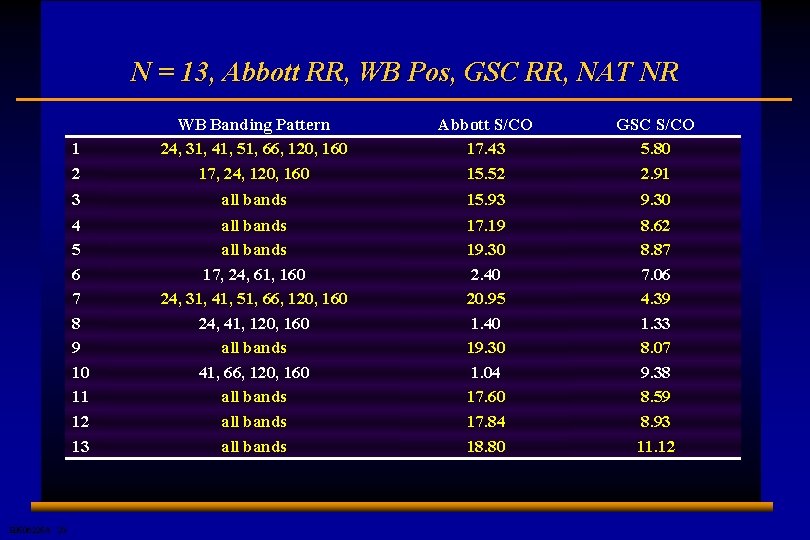
N = 13, Abbott RR, WB Pos, GSC RR, NAT NR E 0506225 A 23 1 2 WB Banding Pattern 24, 31, 41, 51, 66, 120, 160 17, 24, 120, 160 Abbott S/CO 17. 43 15. 52 GSC S/CO 5. 80 2. 91 3 all bands 15. 93 9. 30 4 5 6 7 8 9 10 11 12 13 all bands 17, 24, 61, 160 24, 31, 41, 51, 66, 120, 160 24, 41, 120, 160 all bands 41, 66, 120, 160 all bands 17. 19 19. 30 2. 40 20. 95 1. 40 19. 30 1. 04 17. 60 17. 84 18. 80 8. 62 8. 87 7. 06 4. 39 1. 33 8. 07 9. 38 8. 59 8. 93 11. 12
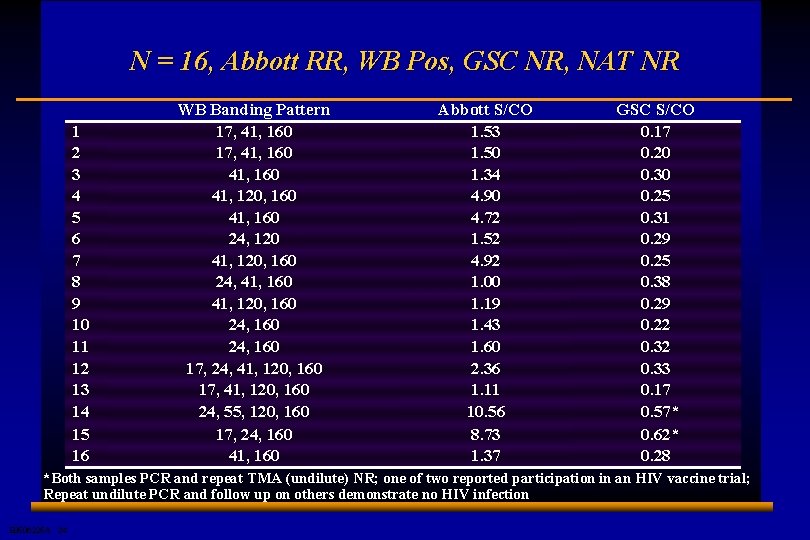
N = 16, Abbott RR, WB Pos, GSC NR, NAT NR 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 WB Banding Pattern 17, 41, 160 41, 120, 160 41, 160 24, 120 41, 120, 160 24, 41, 160 41, 120, 160 24, 160 17, 24, 41, 120, 160 17, 41, 120, 160 24, 55, 120, 160 17, 24, 160 41, 160 Abbott S/CO 1. 53 1. 50 1. 34 4. 90 4. 72 1. 52 4. 92 1. 00 1. 19 1. 43 1. 60 2. 36 1. 11 10. 56 8. 73 1. 37 GSC S/CO 0. 17 0. 20 0. 30 0. 25 0. 31 0. 29 0. 25 0. 38 0. 29 0. 22 0. 33 0. 17 0. 57* 0. 62* 0. 28 *Both samples PCR and repeat TMA (undilute) NR; one of two reported participation in an HIV vaccine trial; Repeat undilute PCR and follow up on others demonstrate no HIV infection E 0506225 A 24

HIV Dual EIA Algorithm Findings/Conclusions u Feasibility of NAT combined with the dual EIA algorithm for HIV confirmation was demonstrated – – Sensitivity = 100% (317/317); 95% CI 98. 84 -100% Specificity = 98. 4% (7, 445/7, 567); 95% CI 98. 11 -98. 68% No. WBs eliminated = 98. 5% (7, 762/7, 884) No. indeterminate interpretations eliminated = 98. 6% (3, 388/3, 434) u Majority of WB false pos, selected by the use of one particular EIA, were eliminated u Changes in EIAs to more sensitive/specific versions should not require extensive validations u WB shown to be of little if any value!!! E 0506225 A 25
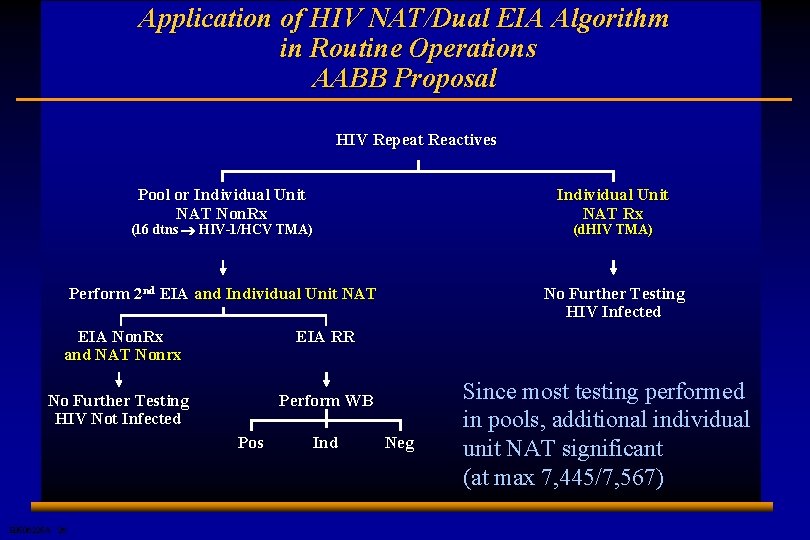
Application of HIV NAT/Dual EIA Algorithm in Routine Operations AABB Proposal HIV Repeat Reactives Pool or Individual Unit NAT Non. Rx Individual Unit NAT Rx Perform 2 nd EIA and Individual Unit NAT No Further Testing HIV Infected (16 dtns ® HIV-1/HCV TMA) EIA Non. Rx and NAT Nonrx EIA RR No Further Testing HIV Not Infected Perform WB Pos E 0506225 A 26 Ind (d. HIV TMA) Neg Since most testing performed in pools, additional individual unit NAT significant (at max 7, 445/7, 567)
- Slides: 26