CHALLENGES FACED IN THE DEVELOPMENT OF BIOSIMILARS Dr

CHALLENGES FACED IN THE DEVELOPMENT OF BIOSIMILARS Dr. G. Hima Bindu MD; PG dip. diabetology Asst. Professor Dept. of Pharmacology Rajiv Gandhi Institute of Medical Sciences Ongole drgujjarla@gmail. com

Biologics Differences Manufacturing process Challenges in the development 28. 10. 2014 2
INTRODUCTION • Biologics derived from living cells • Currently used in PBlood conditions : leuco/neutro/pancytopenia PCancer: colon, breast, non Hodgkins lymphoma PImmune system disorders: RA, psoriasis PNeurological: multiple sclerosis 28. 10. 2014 • World wide, nearly 200 biologics 3

Need for biosimilars • Increasing demand for biologics Patent expiry Takes more time and cost to develop • Alternate version of innovator biopharmceuticals – Similar biotherapeutic products : WHO 28. 10. 2014 4

WHO: “A biotherapeutic product similar in terms of quality, safety and efficacy to an already licensed reference biotherapeutic product. ” Biosimilars are similar……. …. Not Identical Similarity must be shown on the basis of the analytical, non clinical and clinical data 28. 10. 2014 5

DIFFERENCES 28. 10. 2014 6
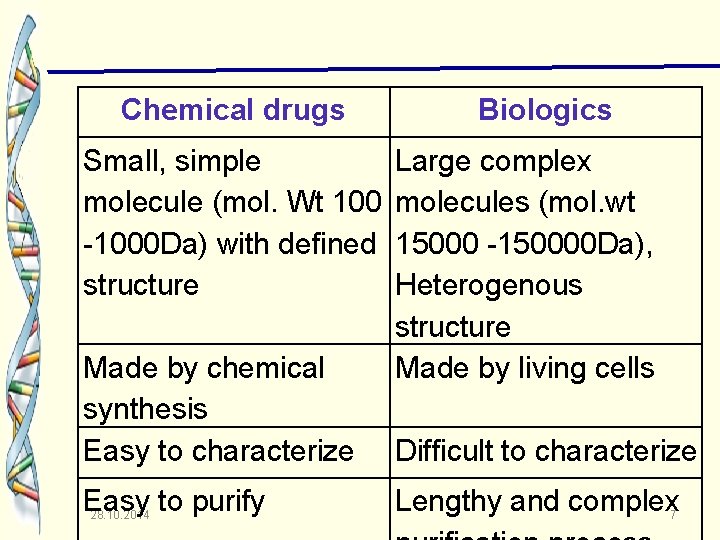
Chemical drugs Small, simple molecule (mol. Wt 100 -1000 Da) with defined structure Made by chemical synthesis Easy to characterize Easy to purify 28. 10. 2014 Biologics Large complex molecules (mol. wt 15000 -150000 Da), Heterogenous structure Made by living cells Difficult to characterize Lengthy and complex 7
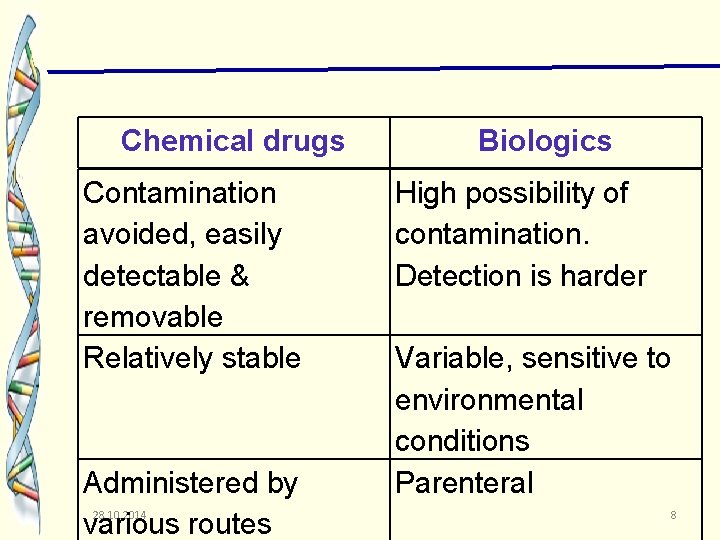
Chemical drugs Contamination avoided, easily detectable & removable Relatively stable Administered by various routes 28. 10. 2014 Biologics High possibility of contamination. Detection is harder Variable, sensitive to environmental conditions Parenteral 8

MANUFACTURING PROCESS 28. 10. 2014 9

Manufacturing Process ü Complex process ü Developed through sequential process ü Systematically engineered to match the reference product 28. 10. 2014 10
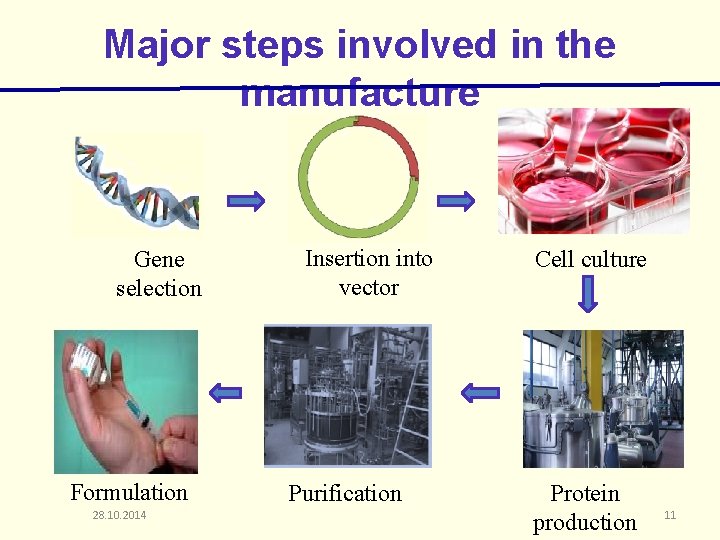
Major steps involved in the manufacture Gene selection Formulation 28. 10. 2014 Insertion into vector Purification Cell culture Protein production 11

CHALLENGES 28. 10. 2014 12

ü Cell line generation ü Protein production ü Protein purification 28. 10. 2014 13
Cell Line Generation • Microbial cells, cell lines of human or animal origin or tissues derived from animals or plants are used. • Bacteria & yeast cell lines require minimal growth media conditions and are fast growing. Traditional cell lines are prone to host cell protein contamination. 28. 10. 2014 14

Cont…. • Chinese Hamster ovary (CHO) cells are preferred mammalian cell line • If the host cell line used for production of reference product is known same cell line is used. The expression system can have a significant effect on the types and extent of translational and post translational 28. 10. 2014 15

Cont…. • Every manufacturer uses unique cell line and proprietary process. ü So it is difficult to produce biosimilars that are identical to the original drug. 28. 10. 2014 16

Protein Production • Structure of protein is heterogeneous exhibit complex three dimensional conformations. The structure – function relationships are very sensitive as modifications of may affect safety, purity and/ or potency. PELISA and size exclusion chromatography are used 28. 10. 2014 17

Cont…. The function of a protein can be altered by üReplication errors in the DNA encoding protein sequence üAmino acid misincorporation during translation üPost translational modifications 28. 10. 2014 18
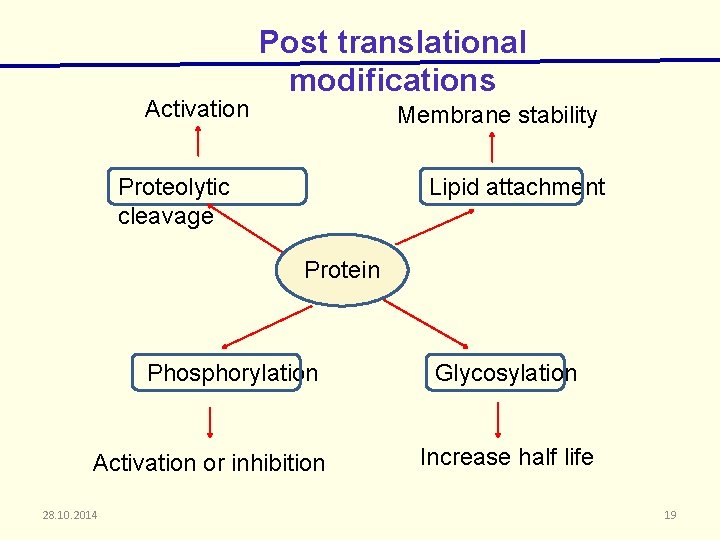
Activation Post translational modifications Membrane stability Proteolytic cleavage Lipid attachment Protein Phosphorylation Activation or inhibition 28. 10. 2014 Glycosylation Increase half life 19

Cont…. Changes in protein modification may reduce biological activity These post translational modifications are affected by the cell line. Glycosylation is sensitive to cell growth conditions Pchanges in culture p. H P the availability of precursors and nutrients 28. 10. 2014 20

Cont…. It is difficult to characterize glycosylation pattern or the higher order/ 3 D structure. . • Tools to detect: PSDS-PAGE PRP-HPLC PSize exclusion chromatography PMass spectroscopy 28. 10. 2014 21

Protein Purification Oxidation/deamidation or even protein aggregation can occur during the process. Protein molecules can also be degraded and impurities created ü Additives used to stabilize proteins: – human serum albumin (HSA), – polysorbates, 28. 10. 2014 22

Immunogenicity • All therapeutic proteins induce some antibody response. • Various factors influence immunogenicity üpatient related characteristics PProduct related factors 28. 10. 2014 23

Product related factors ü Molecule design ü Cell line ü Deglycosylation ü Exposure of antigenic sites ü Misfolding/ aggregation ü Impurities 28. 10. 2014 24

SUMMARY • The increasing demand for more cost effective treatment and patent expiry of biologics created growing market for biosimilars. • It is difficult to develop biosimilars identical to original drug. üUse of different vector üDiff. Cell expression system üDiff. in growth media, nutrients üDiff. operating methods, reagents, 28. 10. 2014 25

28. 10. 2014 26
- Slides: 26