Challenges and opportunities of metal complexes in medicinal




















- Slides: 49

Challenges and opportunities of metal complexes in medicinal field Dr. A. Jeya Rajendran Asst. Professor, Advanced Materials Research lab Department of Chemistry Loyola College, Chennai

Functional role of inorganic elements

Functional role of d and f-block elements in bio-field

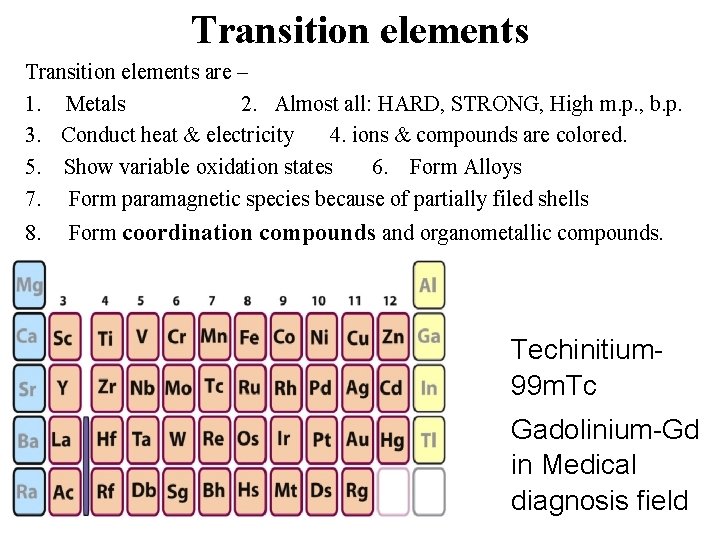
Transition elements are – 1. Metals 2. Almost all: HARD, STRONG, High m. p. , b. p. 3. Conduct heat & electricity 4. ions & compounds are colored. 5. Show variable oxidation states 6. Form Alloys 7. Form paramagnetic species because of partially filed shells 8. Form coordination compounds and organometallic compounds. Techinitium 99 m. Tc Gadolinium-Gd in Medical diagnosis field

Necessity for metal complexes • Biomedical applications- Molecular Imaging Transition metal ions are often highly toxic and are deposited in tissues. Toxic metal ions must be bound in a thermo dynamically and kinetically stable complexes. Kinetic inertness decreases the probability of the complex decomposition. Importance is given to design of new ligands forming metal complexes- with in vivo stability and holding their expected function in treatment and diagnosis

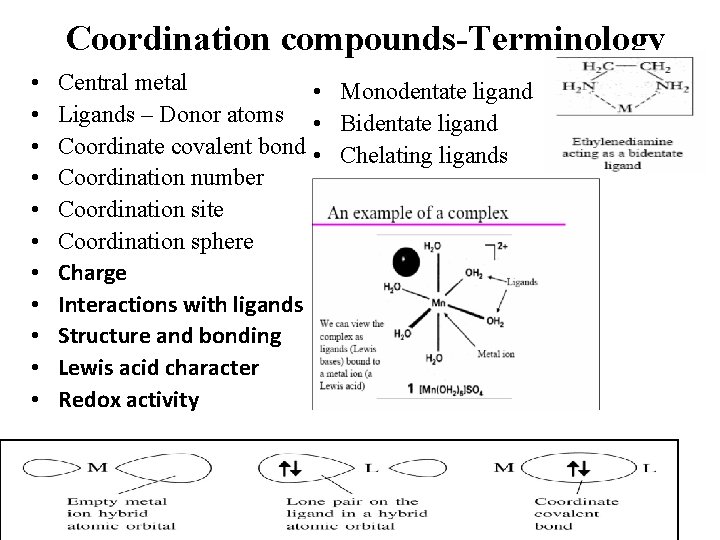
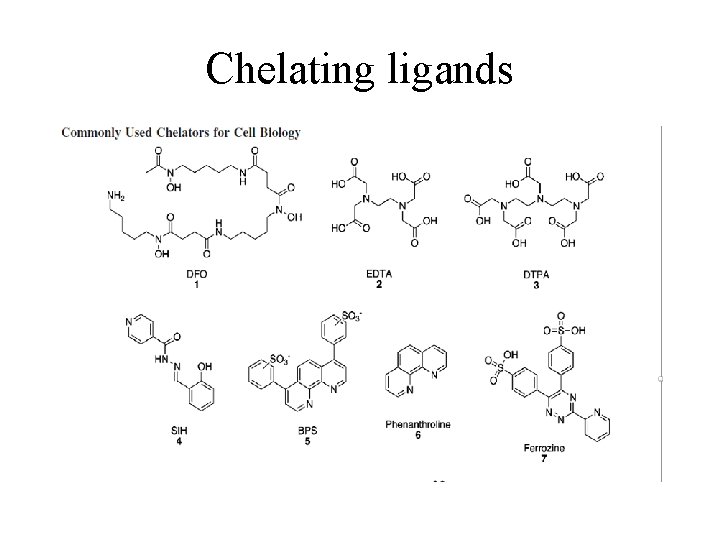
Coordination compounds-Terminology • • • Central metal • Monodentate ligand Ligands – Donor atoms • Bidentate ligand Coordinate covalent bond • Chelating ligands Coordination number Coordination site Coordination sphere Charge Interactions with ligands Structure and bonding Lewis acid character Redox activity

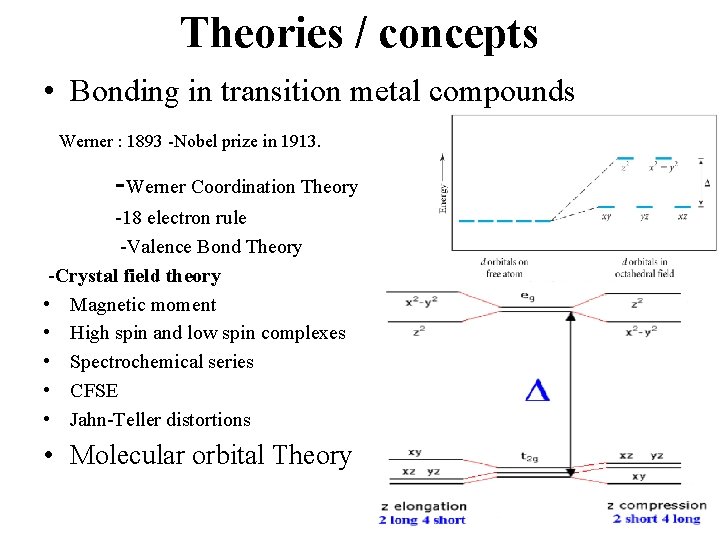
Theories / concepts • Bonding in transition metal compounds Werner : 1893 -Nobel prize in 1913. -Werner Coordination Theory -18 electron rule -Valence Bond Theory -Crystal field theory • Magnetic moment • High spin and low spin complexes • Spectrochemical series • CFSE • Jahn-Teller distortions • Molecular orbital Theory

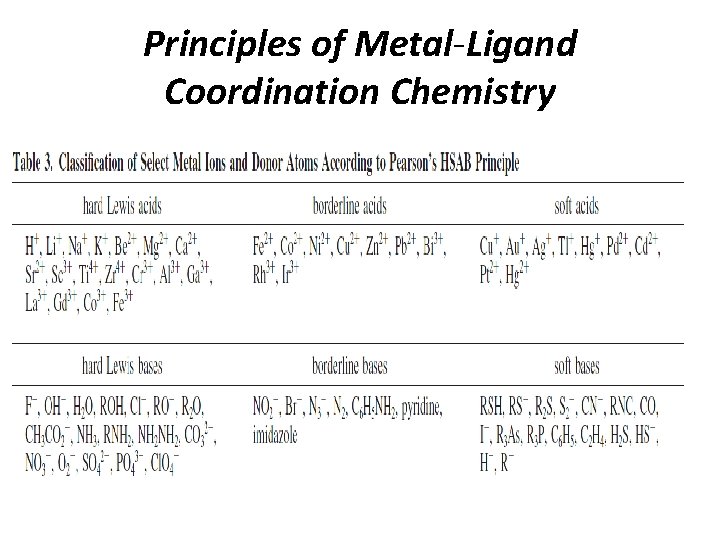
Principles of Metal-Ligand Coordination Chemistry

Chelating ligands

Applications of Metal Complexes • • • • Catalysts – Wilkinson catalyst and Ziegler-Natta catalyst Photo catalysts optical applications dyes and pigments solar cells macrocycles as ligands metal based drugs metal drug interactions MRI contrast agents radiopharmaceuticals photodynamic therapy magnetic materials precursors for semiconductor films and nanoparticles

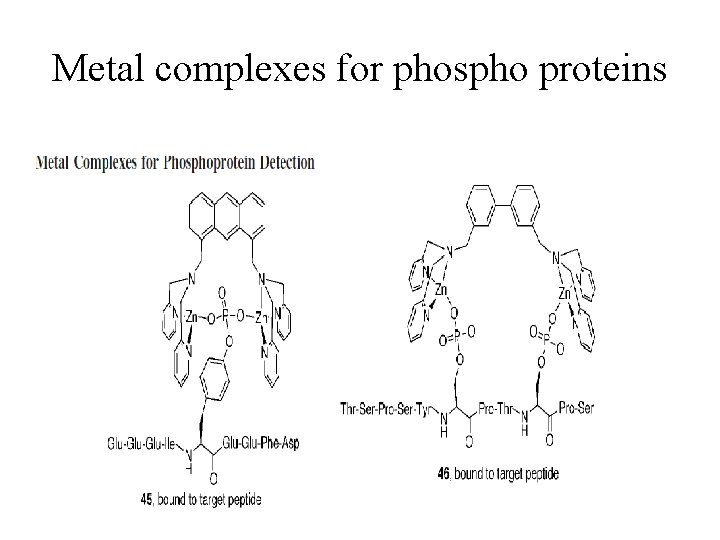
Metal complexes for phospho proteins

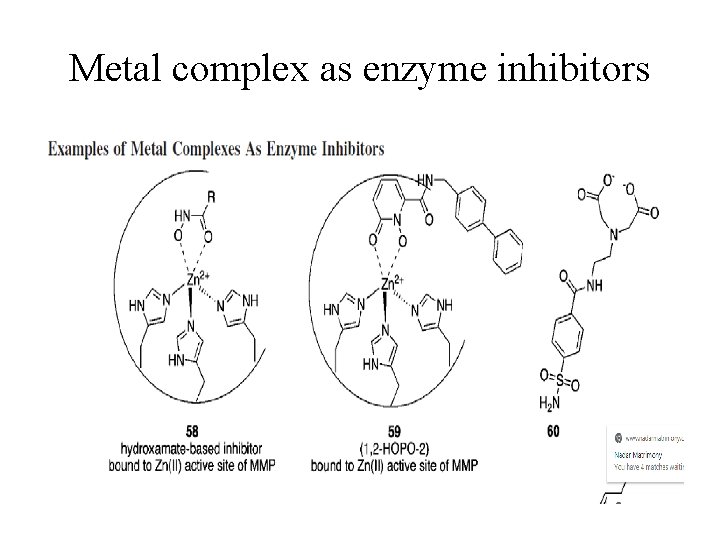
Metal complex as enzyme inhibitors

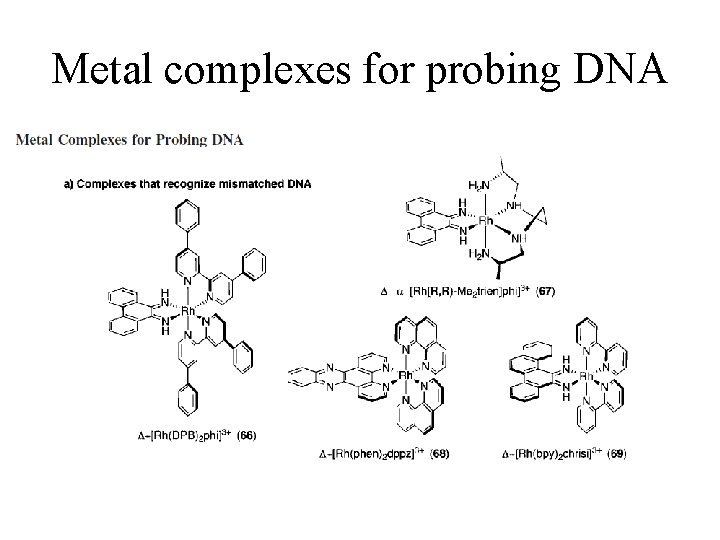
Metal complexes for probing DNA

Molecular Imaging • • Molecular imaging – non-invasive technique a group of imaging techniques used in the medicine and the molecular biology (in the medicine often called CT – Computer Tomography). • MRI (Magnetic Resonance Imaging)- Contrast agents are often applied to reach better contrast of the image • • Radiodiagnostic methods (SPECT – Single. Photon Emission Computer Tomography, PET – Positron Emission Tomography) Optical imaging methods (luminescent and fluorescent markers) and an ultrasound.

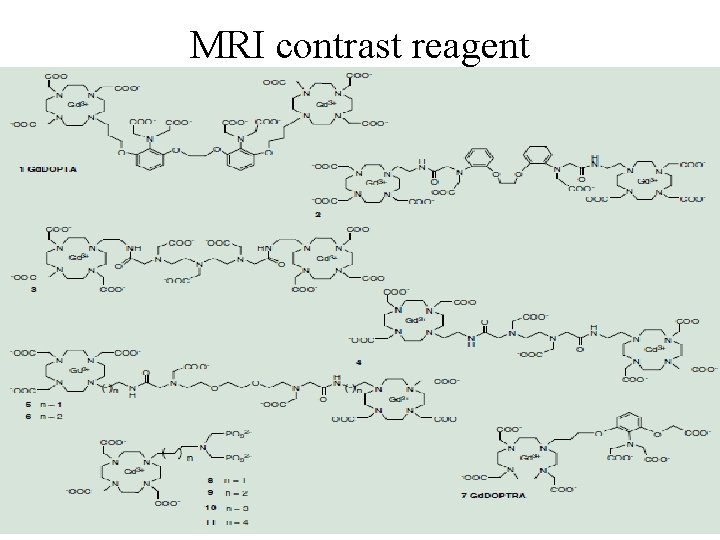
MRI-Design of suitable ligands Most of commercially applied contrast agents are based on gadolinium(III) ion. Presence of paramagnetic compounds dramatically changes the behavior of water molecules Complex of Gd(III) ion must exhibit stability and transfer of a magnetic information between the metal ion and the water molecules of the bulk. MRI is based on changes in behavior of water molecules in an external magnetic field of the scanner and depends on 1. water concentration in the tissue, 2. Concentration of ions and organic compounds in the body fluids 3. p. H 4. Temperature MRI contrast agents - macromolecular carrier. superparamagnetic properties of nanocrystalline iron

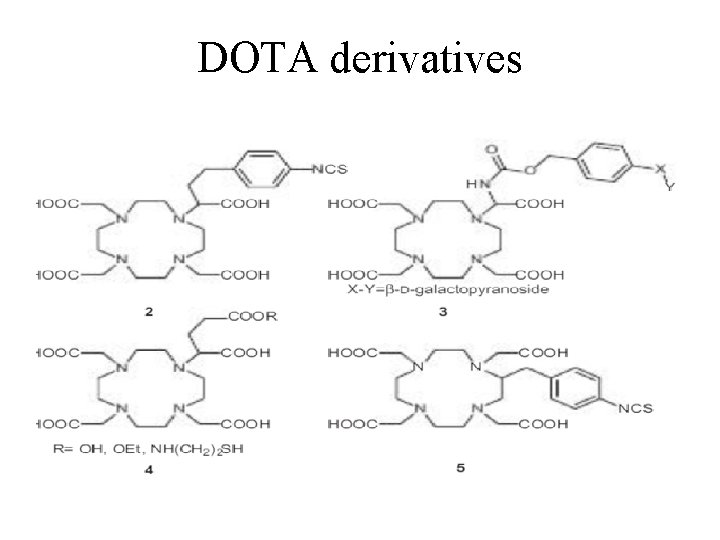
DOTA derivatives

MRI contrast reagent

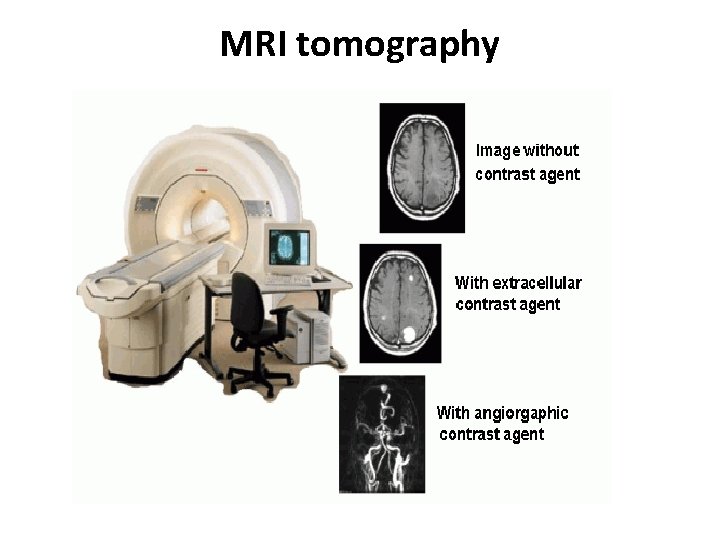
MRI tomography

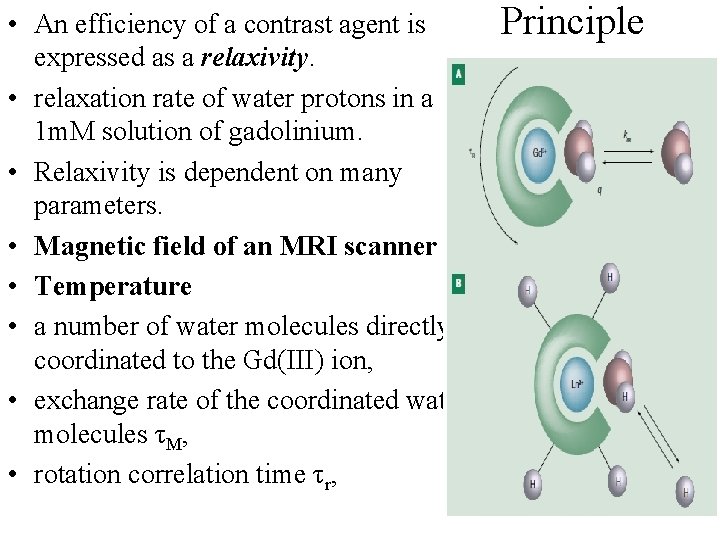
• An efficiency of a contrast agent is expressed as a relaxivity. • relaxation rate of water protons in a 1 m. M solution of gadolinium. • Relaxivity is dependent on many parameters. • Magnetic field of an MRI scanner • Temperature • a number of water molecules directly coordinated to the Gd(III) ion, • exchange rate of the coordinated water molecules τM, • rotation correlation time τr, Principle

Electronic relaxation parameters of the metal ion etc. Coordination number of Gd(III) ion is 8 or 9 in dependence on steric requirements of ligands. In MRI, the ligands containing eight donor groups are used. MRI contrast agents are derived from polyaminocarboxylates DOTA or DTPA The exchange of the water molecule between the coordination sphere and the bulk solvent is main way how the magnetic information is transferred. Absence of a water molecule in the first coordination sphere results in reduction of the magnetic information transfer

Presence of two coordinated water molecules decreases stability of the complex. Flexibility of such a coordination sphere allows substitution of the coordinated water molecules by biogenic ligands (acetates, carbonates, aminoacids, etc. ) resulting again in the decrease of contrast ability. The exchange rate of the coordinated water molecules is one of the most important parameters, which can be chemically tuned through choice of suitable ligand. To reach the maximum efficiency, the residence time (τM) of the water molecule in the coordination sphere should be 10– 20 ns for magnetic fields that used nowadays Another parameter, which can be chemically modified, is size of a molecule and its tumbling rate (rotation corellation time τr). Bigger molecules show better MRI properties. The increase of the rotation correlation time is usually reached by attachment of a low -molecular weight complex to a macromolecular carrier (e. g.

Challenges • The dependency of the relaxivity on the water residence time and on the rotation correlation time shows a mountain-like shape. • The efficiencies of the todayapplied contrast agents are close to the root of the mountain (see arrow in the picture) and we shall climb up.

Dept. of Chemistry Nuclear Medicine • Two broad classification of Nuclear Medicine (i) Therapeutic Nuclear Medicine ―> administered to the patient orally or intravenously, over a period of time they slowly accumulate in the tumor site and the alpha or beta particles emitted by them destroy the tumor cells Treating the diseases. (ii) Diagnostic Nuclear Medicine ―> v known as Nuclear Imaging, Nuclear Scintigraphy, Isotope Scan, Isotope Imaging, Isotope Scintigraphy, Gamma Scan, Gamma Imaging or Gamma Scintigraphy. -the distribution of the radio tracer inside the body is imaged by the equipment called Gamma Camera. Finding the diseases.

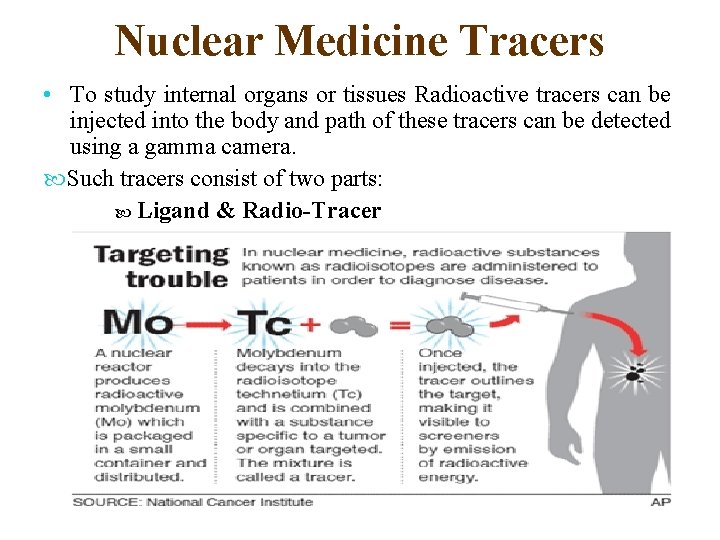
Nuclear Medicine Tracers • To study internal organs or tissues Radioactive tracers can be injected into the body and path of these tracers can be detected using a gamma camera. Such tracers consist of two parts: Ligand & Radio-Tracer

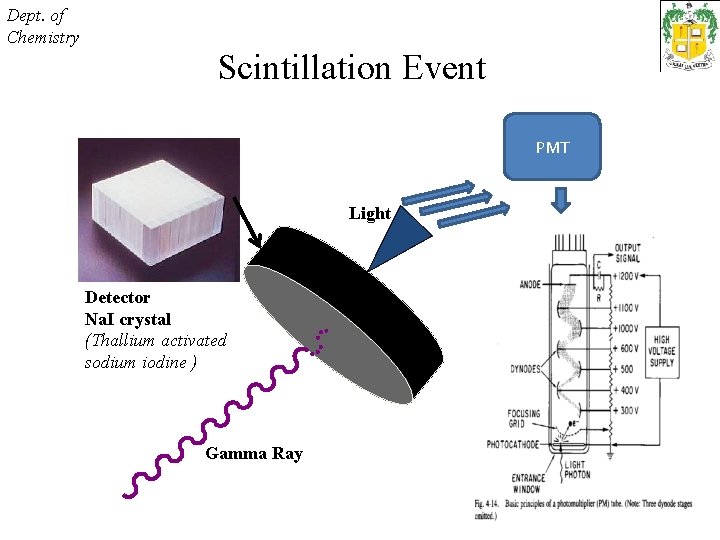
Dept. of Chemistry Nuclear Scan Radionuclide tagged with suitable ligand having specificity for an organ is injected to patients. It is uniformly mixed during blood circulation, slowly extracted from the blood and concentrated in that organ for which it has been studied. The gamma camera detector is focused over the organ of interest. The collimator attached to the detector selects only parallel beam of gamma rays from the organ. A gamma ray that successfully traverses the collimator strikes the Na. I (Tl) crystals, called a scintillator.

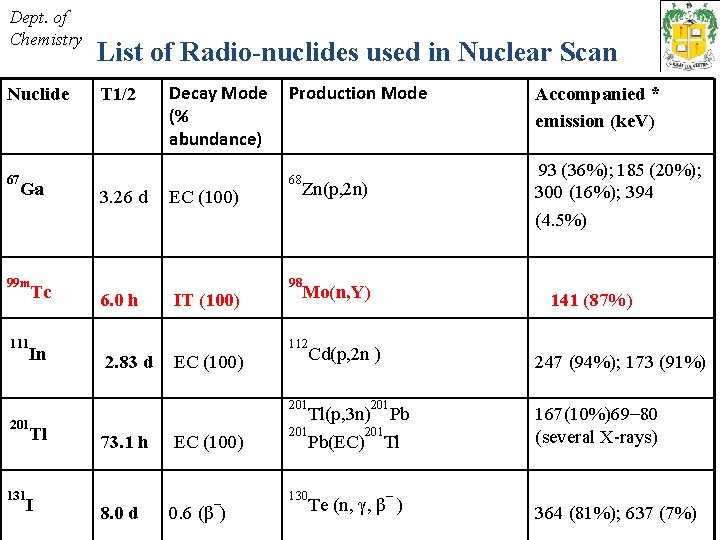
Dept. of Chemistry Nuclide 67 Ga T 1/2 Decay Mode Production Mode (% abundance) 3. 26 d EC (100) Tc 6. 0 h IT (100) In 2. 83 d EC (100) 99 m 111 201 Tl 131 List of Radio-nuclides used in Nuclear Scan I 73. 1 h 8. 0 d EC (100) – 0. 6 (β ) Accompanied * emission (ke. V) 93 (36%); 185 (20%); 300 (16%); 394 (4. 5%) 68 Zn(p, 2 n) 98 141 (87%) 112 247 (94%); 173 (91%) Mo(n, Y) Cd(p, 2 n ) 201 130 – Tl(p, 3 n) Pb 201 Pb(EC) Tl Te (n, γ, β ) 167(10%)69– 80 (several X-rays) 364 (81%); 637 (7%)

Dept. of Chemistry An ideal Radio-Isotopes for Nuclear Scan? • Must be readily available. • Must be pure Gamma emitter. • Must have reasonable short half- life (Few Hours) • Must have medium energy range (75 -364 ke. V). • Must be easily labeled with many ligands. • Cost effective “ 99 m. Tc possesses all these qualities”

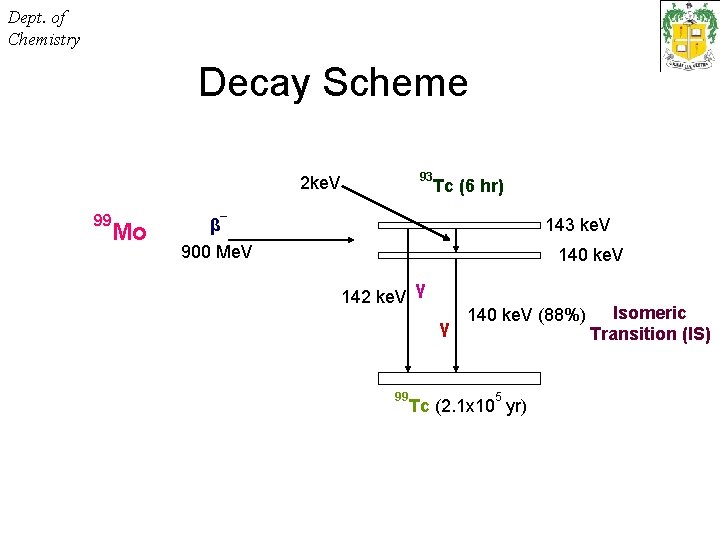
Dept. of Chemistry Decay Scheme 93 2 ke. V 99 Mo Tc (6 hr) – β 900 Me. V 143 ke. V 140 ke. V 142 ke. V γ γ 99 140 ke. V (88%) 5 Tc (2. 1 x 10 yr) Isomeric Transition (IS)

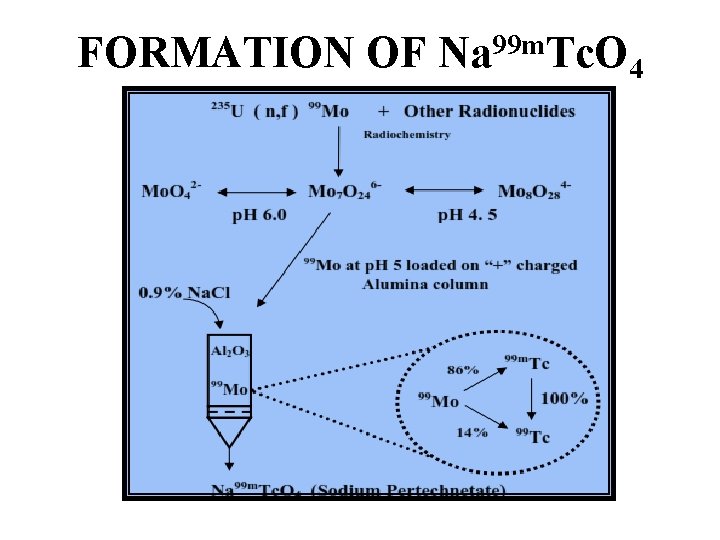
FORMATION OF Na 99 m. Tc. O 4

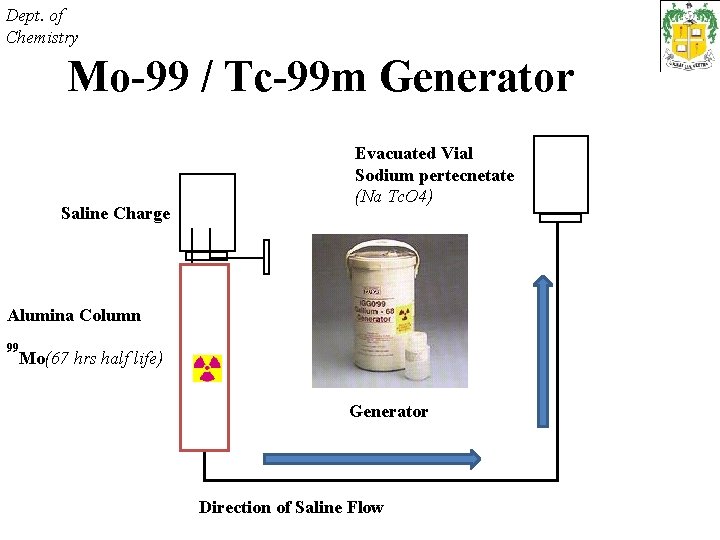
Dept. of Chemistry Mo-99 / Tc-99 m Generator Saline Charge Evacuated Vial Sodium pertecnetate (Na Tc. O 4) Alumina Column 99 Mo(67 hrs half life) Generator Direction of Saline Flow

Dept. of Chemistry Radiolabeling: Addition of required amount of radioactivity in the form of Na. Tc. O 4 into the vial. Kit components – vial 1. Reducing agent, Sn. Cl 2 2. Carrier ligand 3. Antioxidant 4. Buffer Radiochemical purity analysis: Ascending Paper chromatography Techniques 1. Whatmann filter paper and 80% methanol or saline. 2. Free 99 m. Tc-sodium pertechnetate 3. 99 m. Tc –carrier ligand - different Rf values In vitro and invivo stability of the radiocomplex –Organ specificity. Bio-distribution studies Toxicity Assay- to fix the lowest dose

Dept. of Chemistry Hot Lab Preparation Radio pharmacy Kit

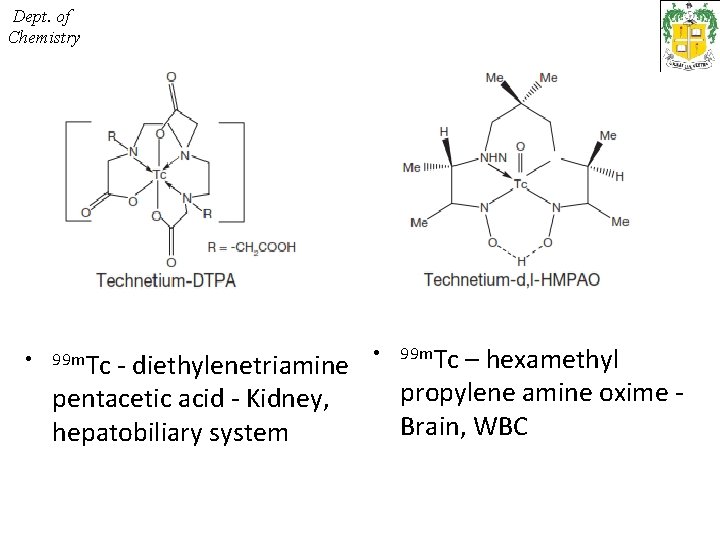
Dept. of Chemistry • 99 m. Tc - diethylenetriamine • pentacetic acid - Kidney, hepatobiliary system 99 m. Tc – hexamethyl propylene amine oxime - Brain, WBC

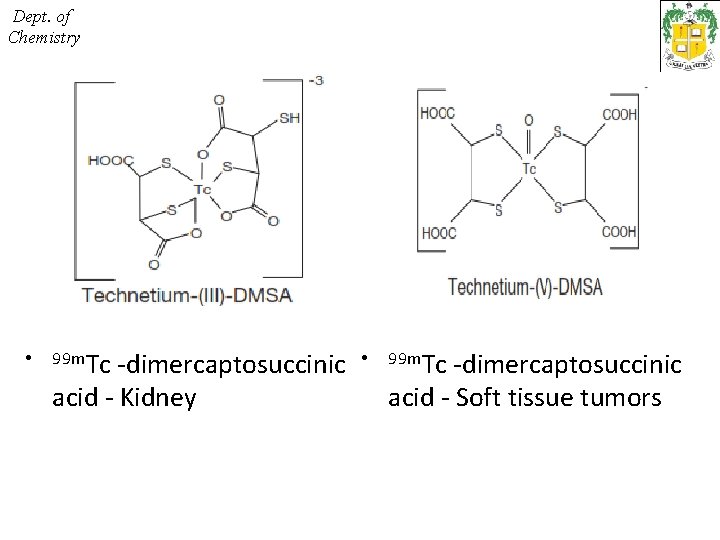
Dept. of Chemistry • 99 m. Tc -dimercaptosuccinic • acid - Kidney 99 m. Tc -dimercaptosuccinic acid - Soft tissue tumors

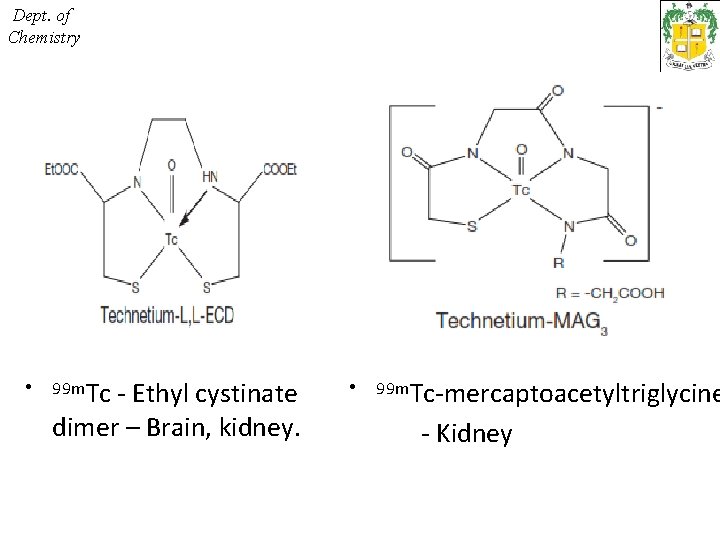
Dept. of Chemistry • 99 m. Tc - Ethyl cystinate dimer – Brain, kidney. • 99 m. Tc-mercaptoacetyltriglycine - Kidney

Dept. of Chemistry Gamma Cameras Detector Patient Table

Dept. of Chemistry Nuclear Medicine Imaging - rays

Dept. of Chemistry Scintillation Event PMT Light Detector Na. I crystal (Thallium activated sodium iodine ) Gamma Ray

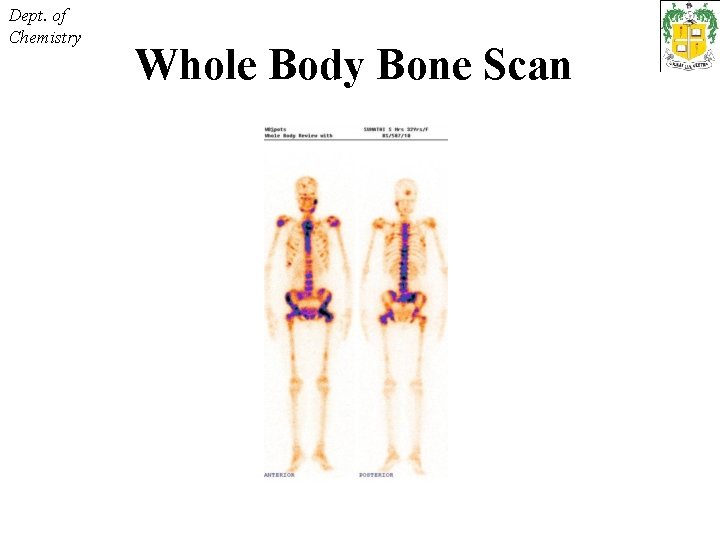
Dept. of Chemistry Whole Body Bone Scan

Nuclear imaging studies

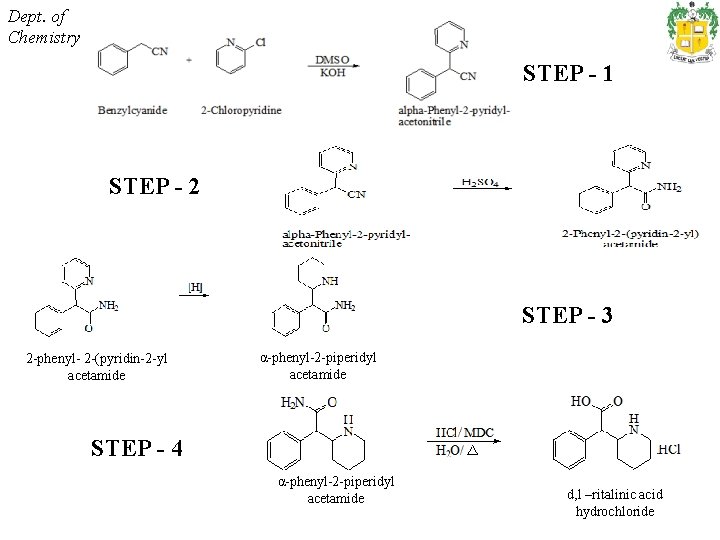
Synthesis, characterization and technetium-99 m radiolabeling of d, l – ritalinic acid hydrochloride a. Anbalagan Thangavelu, M. Prabhu, K. Eswaramurthy, S. Bharathi, A. Jeyarajendran* • d, l – ritalinic acid hydrochloride was synthesized from benzylcyanide and 2 -chloropyridine in a four step process and was characterized • The ligand was then radiolabeled with 99 m. Tc – sodiumpertechnetate by the direct pertechnetate addition using stannous chloride dihydrate as reducing agent. • The percentage labeling efficiency varies from 12 -80% at a p. H range of 5 -6. 5. The optimum labeling was obtained at a ratio of 2: 4: 0. 5 of d, l – ritalinic acid hydrochloride, ascorbic acid and stannous chloride dihydrate. • Radiochemical purity was evaluated using ascending paper chromatography and was found to be 72. 28 ± 6. 15 and stable up to 6 hrs after preparation at room temperature.

Dept. of Chemistry STEP - 1 STEP - 2 STEP - 3 2 -phenyl- 2 -(pyridin-2 -yl acetamide α-phenyl-2 -piperidyl acetamide STEP - 4 α-phenyl-2 -piperidyl acetamide d, l –ritalinic acid hydrochloride

Dept. of Chemistry a) b) c) d) e) f) i. iii. iv. Overall steps To identify suitable ligands To synthesize potential ligands and to characterize. 99 m To label with Tc. To assess the percent labeling. To do the bio-distribution study, if percent labeling is >95%, If bio-distribution study reveals any useful and specific organ accumulation of the injected 99 m. Tc-complex, do all the Radiopharmaceutical QC. QUALITY CONTROL Stability Sterility Apyrogenicity Safeness/ toxicity

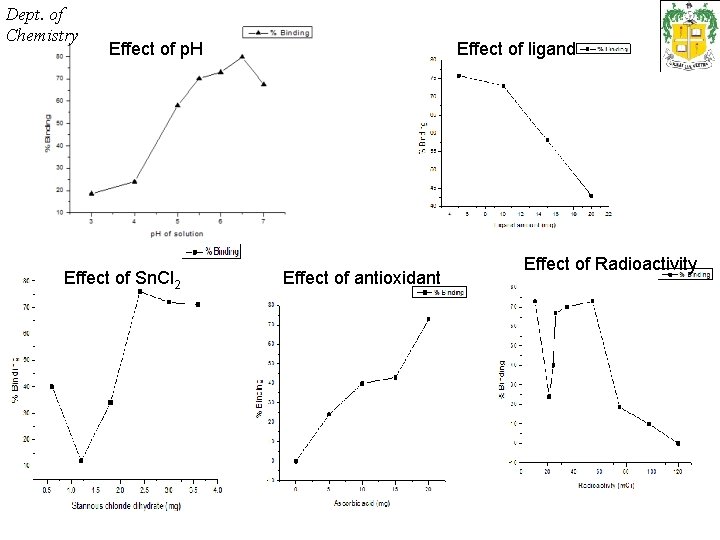
Dept. of Chemistry RADIOCHEMICAL PURITY • The percent labeling efficiency were evaluated using the formula – • % hydrolyzed 99 m. Tc fraction was evaluated from the saline chromatogram % H = Activity at the bottom of the strip x 100 Activity at the (top+bottom) of strip • % free 99 m. Tc fraction was calculated from the acetone chromatogram % F = Activity at the top of the strip x 100 Activity at the (top+bottom) of strip • % bound 99 m. Tc is calculated as % B = 100 – (%H + %F)

Dept. of Chemistry Effect of p. H Effect of Sn. Cl 2 Effect of ligand Effect of antioxidant Effect of Radioactivity

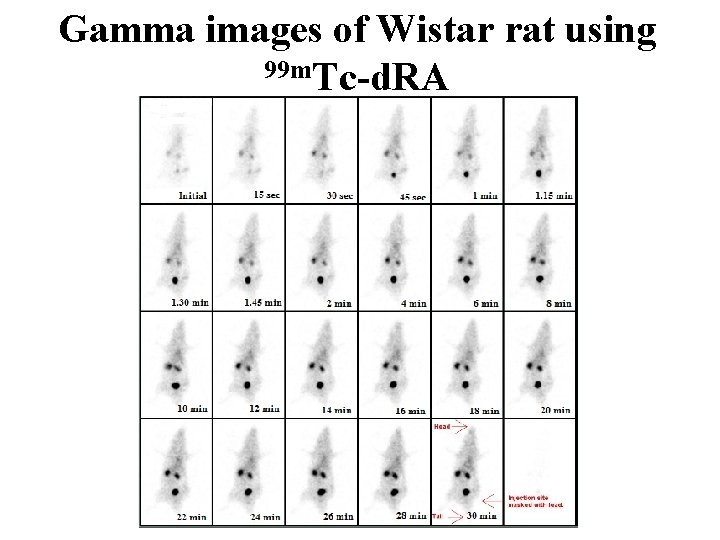
Gamma images of Wistar rat using 99 m. Tc-d. RA

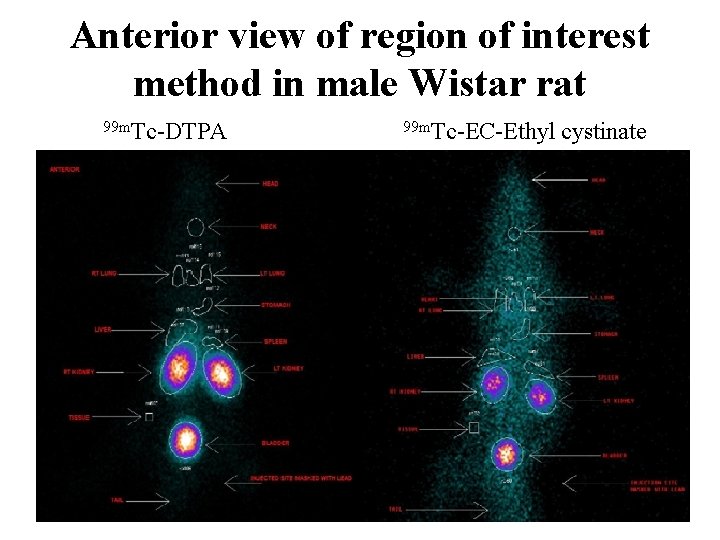
Anterior view of region of interest method in male Wistar rat 99 m. Tc-DTPA 99 m. Tc-EC-Ethyl cystinate

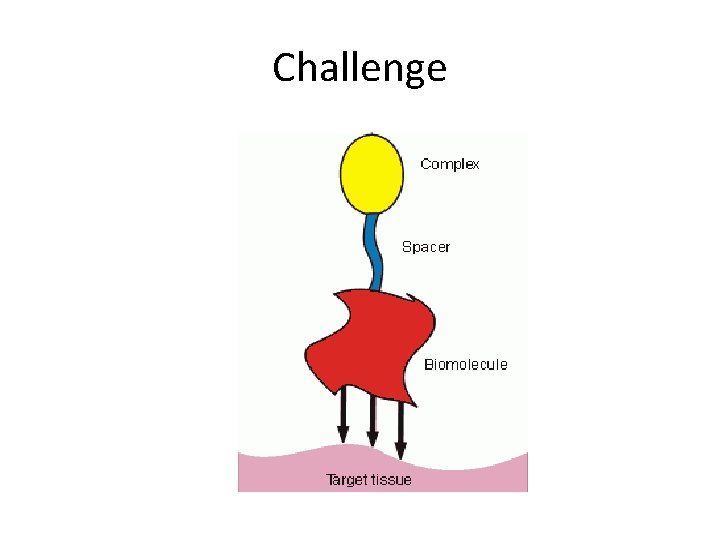
Challenge

Thank You Research Scholars Mr. T. Anbalagan – M. Sc, M. Phil, DMRIT, RSO (Diploma in medical radio isotope technique, BARC, Mumbai) - Certification course – Radiation safety officer(RSO) Mr. M. Prabhu, Mr. K. Eswara Moorthy – Organic synthesis (Malladi Pharmaceuticals) Dr. I. R. Celine Rose, Dr. F. Michael Raj –Nano technology Mr. Soven Dhawa, Ms. Vigneswari, Mr. Serma Rajan S. Bharathi – M. Phil (Nuclear Imaging)