CHALAPATHI INSTITUTE OF PHARMACEUTICAL SCIENCES Approved by AICTE

CHALAPATHI INSTITUTE OF PHARMACEUTICAL SCIENCES Approved by AICTE, PCI & Affiliated to ANU Recognized by Department of Technical Education, Govt of A. P. Certified By ISO 9001: 2008 www. chalapathipharmacy. in

PRINCIPLE AND INSTRUMENTATION OF ULTRAVIOLET SPECTROSCOPY

� Electromagnetic radiations are form of energy consisting of discrete packets of energy moving with definite velocity in the presence or absence of a transmission medium. Each packet of energy is called photon. � Most important consequence of interaction is the energy is absorbed or emitted by the matter in discrete amount called as quanta. .
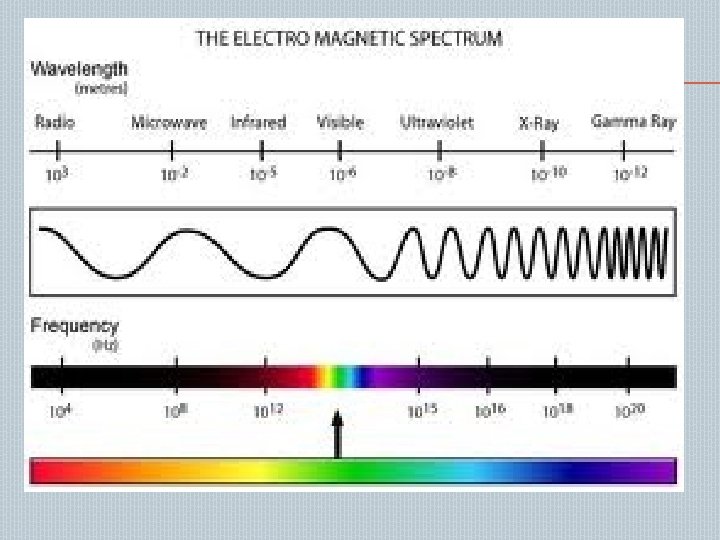

� UV radiations starts at blue end of visible light(400 nm)and end at 200 nm � It is divided into 2 spectral region: � � Near UV— 200 -400 nm Vacuum UV--<200 nm ORIGIN AND THEORY OF SPECTRA UV absorption spectra arises from transitions of electrons. Electrons undergoes transitions from ground( E 0)to higher (E 1) energy level, this energy difference given by E = hυ E 1 ─ E 0 = hυ
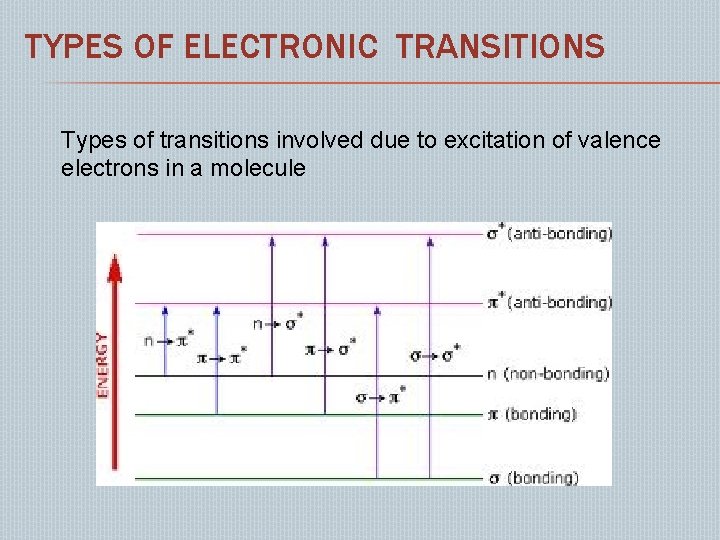
TYPES OF ELECTRONIC TRANSITIONS Types of transitions involved due to excitation of valence electrons in a molecule

σ-σ*TRANSITIONS: � � This type of transitions occurs in saturated compounds. Ex: methane, propane, cyclopropane etc Shows absorption at normal uv range i. e. 180 -400 nm The usual spectroscopy technique cannot be used below 200 nm, since oxygen begins to absorb strongly. n-σ* TRANSITION: This type of transitions takes place in saturated compounds containing hetero atom with lone pair of electrons. Ex: saturated halides, alcohols, ethers, aldehydes, ketones, amines etc.

� π-π* TANSITIONS: � This type of transitions occurs in unsaturated molecules. � � � eg: alkenes, alkynes, carbonyl compounds, azo compounds, cyanides. In conjugated alkenes, absorption band appear around 170 to 190 nm. n-π* TANSITIONS: - � Require less amount of energy hence exhibit weak bands in absorption spectrum. � Eg: saturated aldehydes, ketones, nitrocompound

SOLVENT EFFECTS: . � � � By increasing the polarity of the solvent compounds, like dienes and conjugated hydrocarbons i. e non polar compounds do not experience any appreciable shift. So the max wavelength for the non polar compounds is same in alcohol and hexane. The lambda max for polar compounds is usually shifted with change in polarity of the solvent

BEER’S LAW: States that when a beam of monochromatic light radiation passes through an absorbing medium the intensity of the incident radiation decreases exponentially with increase in the concentration of the absorbing medium -d. I/dc ∞ I -d. I/dc=KI I=I e‾kc

LAMBERT’S LAW � States that “when monochromatic light passes through a transparent medium the rate of decrease in the intensity with the thickness of the medium is proportional to intensity of incident radiation”. d. I/dt ∞ I I=Ie‾kt. BEER-LAMBERT’S LAW I=I e‾kct A=εbc

MOLAR ABSORPTIVITY: - A=A 1% 1 cmbc A=εbc ε – molar absorptivity, (if the concentration is taken in moles per litre) SIGNIFICANCE: � � For identification of compounds. When molecular weight of a compound is unknown, then it can be calculated by ε = a x mol. Wt/10

DEVIATIONS OF BEERS LAW According to beers law, there exists is a direct relationship between the absorbance and concentration. A graph is plotted between absorbance on y-axis concentration on x-axis

TYPES OF DEVIATIONS: � REAL DEVIATIONS � INSTRUMENTAL � CHEMICAL DEVIATIONS REAL DEVIATIONS �These �At are fundamental deviation concentrations molecules will undergo collisions and charge distribution results in deviations.

INSTRUMENTAL DEVIATIONS: - POLYCHROMATIC LIGHT : beers law obeyed only when a monochromatic radiations is used. STRAY LIGHT: -is any radiations reaching the � photodetectors other than the narrow range of wavelength normally transmitted by monochromator. Most important sources are imperfection and defects of the dispersing elements, irregularities and contaminants on the optical surface.

MISMATCHED CELLS If the cells holding the analyte and blank solution are not of equal path length and are not equivalent in optical characteristics. CHEMICAL DEVIATIONS It arises if the absorbing species undergoes chemical changes such as association, dissociation, complex formation, hydrogen bonding, ionization or polymerization. Ex : POLYMERIZATION polymeric form of benzyl alcohol absorbs radiation at 3. 004 Monomeric form → at 2. 750 to 2. 765 Ex: IONIZATION Unionized form of benzoic acid → 273 nm; while ionized form absorbs at→ 268 nm.

PRESENCE OF IMPURITIES: - may causes fluorescence. Chemical deviations can be corrected by using appropriate wavelength, buffers, selecting suitable solvents. CHROMOPHORE: - a covalently unsaturated organic functional group responsible for characteristic absorption of light by a molecule is called chromophore. Ex: ethylenic, acetylenic, carbonyl, acids, nitriles, azo compounds etc. AUXOCHROME: - saturated or unsaturated group with non bonded electrons which do not have any characteristic absorption but when attached to a chromophore it alters the intensity of absorption Ex: -OH, -OR, -NH 2, -NHR, -NR 2, -SH etc. ,
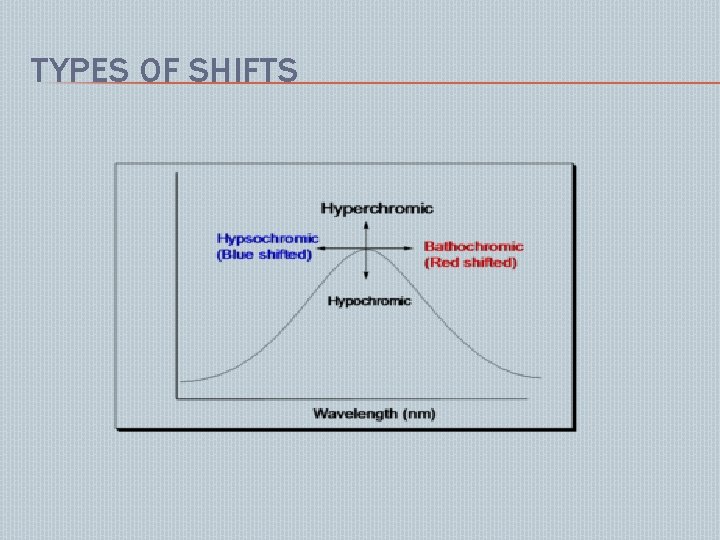
TYPES OF SHIFTS

BATHOCHROMIC SHIFTS: Those groups which � deepens the color of the chromogen causes displacement to longer wavelength. ex: -NH 2, -NHR, NHR 2 Substitution effect: addition of alkyl substitution or addition of double or triple bond in a molecule. � Polarity of the solvent HYPSOCHROMIC SHIFT: Absorption max shifted towards shorter wavelength. Occurs due to removal of conjugation or change in the polarity of the solvents. ex: acetylation of OH, OR, NH 2 ex: aniline absorption max at 280 nm. But in acidic solution it exhibits blue shift at 200 nm

HYPERCHROMIC SHIFT: there is an increase in the intensity of absorption. May be due to the introduction of an auxochrome ex: pyridine 257 nm at Emax 2750 2 -methyl pyridine 262 nm at Emax 3560 HYPOCHROMIC SHIFT: there is a decrease in the intensity of absorption due to certain groups which may deforms the geometry of the molecule. ex: methyl group at 2 position of biphenyl group brings molecular deformation. nd

INSTRUMENTATION SOURCES OF RADIATION: � � deuterium discharge lamp Mercury arc lamp Tungsten halogen lamp Xenon discharge lamp MONOCHROMATORS: Device which converts � � polychromatic light into monochromatic light. Two types Prism monochromators Grating monochromators

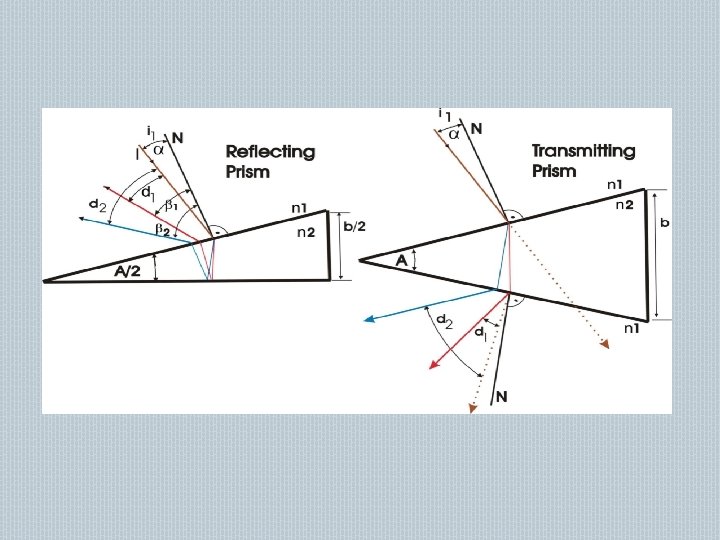
PRISM MONOCHROMATER: Refractive type Reflective type � Made of glass, quartz, fused silica They disperse polychromatic light falling on it into its component rainbow colors according to their wavelengths. � Demerits : gives non linear dispersions � Resolution changes with wavelength �
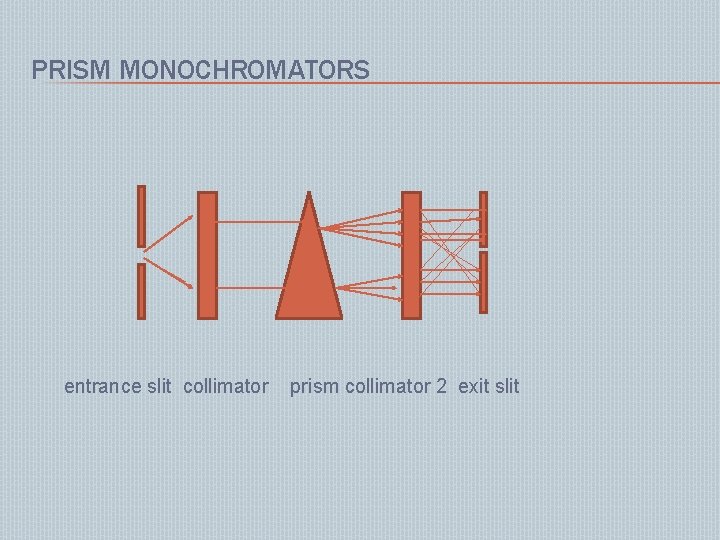
PRISM MONOCHROMATORS entrance slit collimator prism collimator 2 exit slit
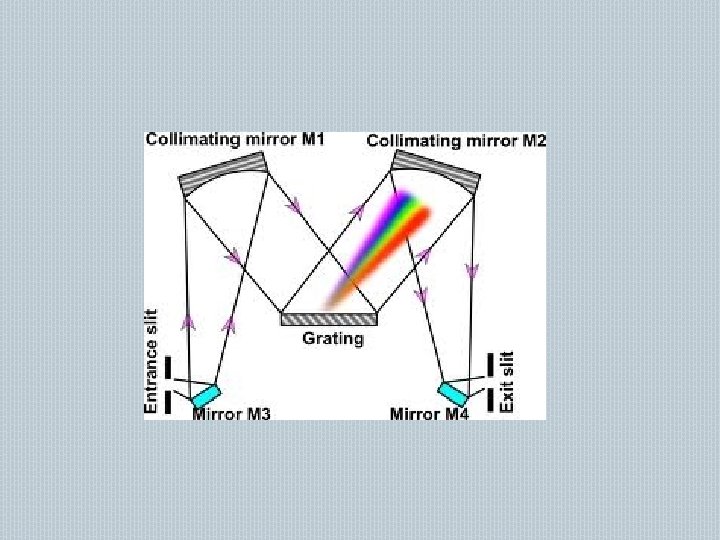

� � � GRATING MONOCHROMATOR: - Made up of glass, quartz, alkyl halides like KBr, Na. Br Large no of equi spaced lines on a glass coated with aluminium Grating does not depends on wavelength Gives a resolution of +/- 0. 1 nm Mechanism is reinforcement No of rulling - 3600 or more grooves/mm for the uv and visible region FILTERS ABSORPTION FILTERS: � � Made of pigmented glass or dyed gelatin Glass filters have a wide bandwidth 150 nm Gelatin filters have a bandwidth 25 nm Disadvantage- intensity of radiation decreases due to absorption of filter
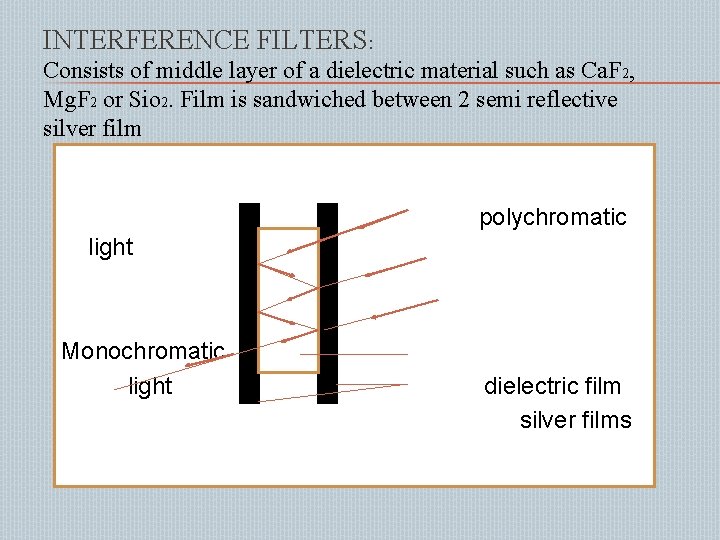
INTERFERENCE FILTERS: Consists of middle layer of a dielectric material such as Ca. F 2, Mg. F 2 or Sio 2. Film is sandwiched between 2 semi reflective silver film polychromatic light Monochromatic light dielectric film silver films
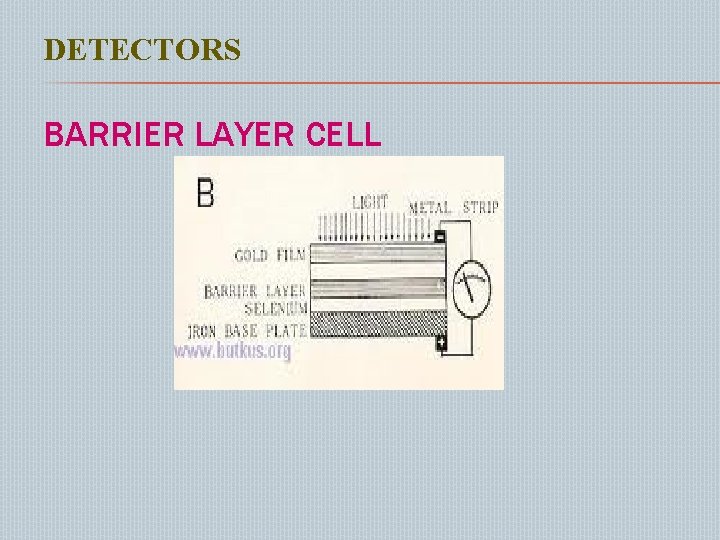
DETECTORS BARRIER LAYER CELL
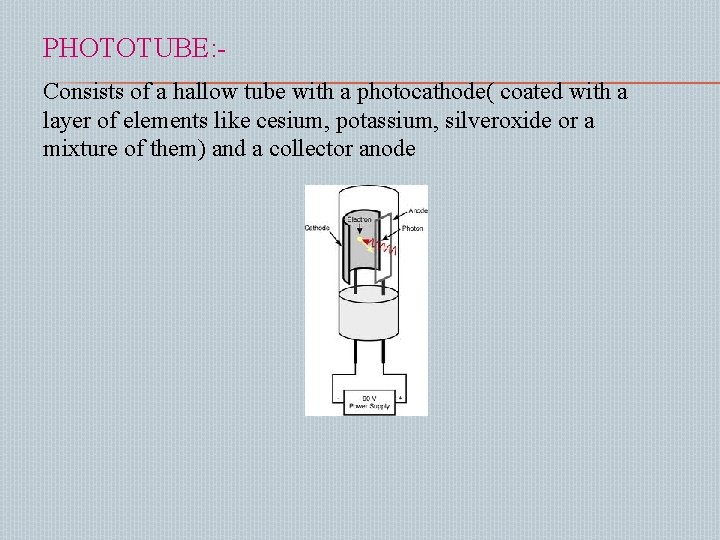
PHOTOTUBE: Consists of a hallow tube with a photocathode( coated with a layer of elements like cesium, potassium, silveroxide or a mixture of them) and a collector anode
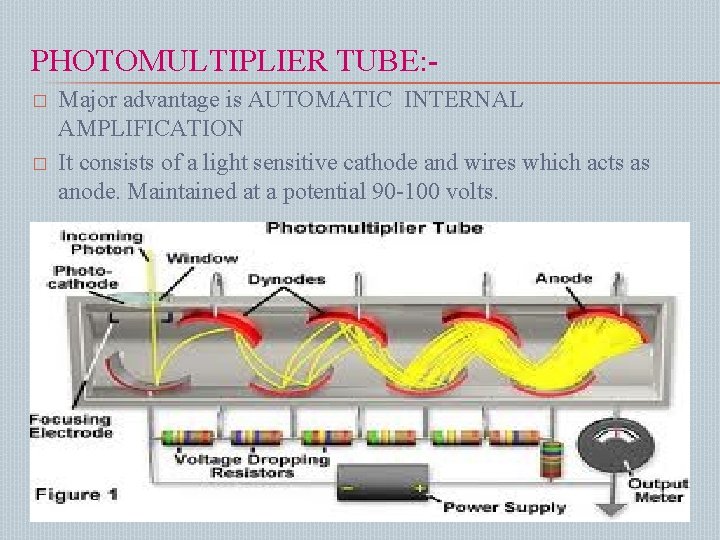
PHOTOMULTIPLIER TUBE: � � Major advantage is AUTOMATIC INTERNAL AMPLIFICATION It consists of a light sensitive cathode and wires which acts as anode. Maintained at a potential 90 -100 volts.
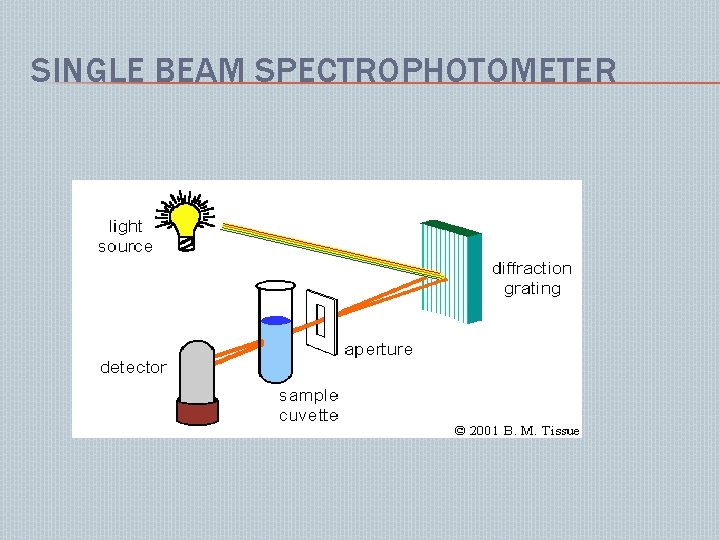
SINGLE BEAM SPECTROPHOTOMETER
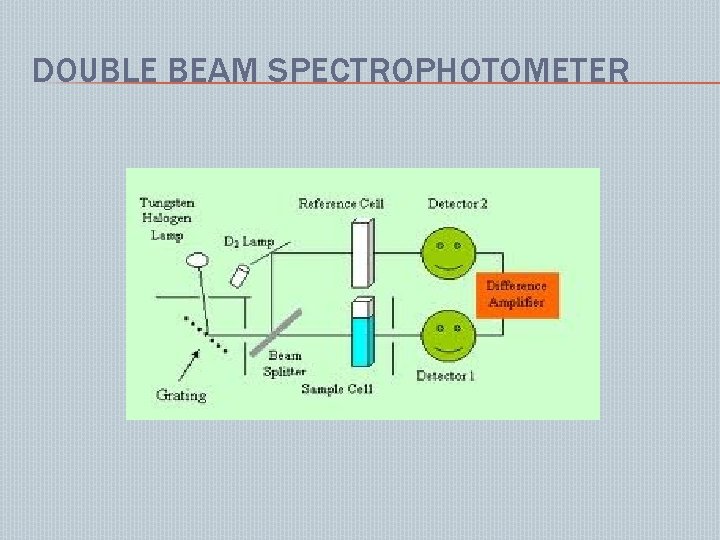
DOUBLE BEAM SPECTROPHOTOMETER

REFERENCES � Pavia, Lampman, Kriz ; Introduction to spectroscopy; 3 rd edition; pg- 353 -360. � Skoog, West, Holler, Crouch; Fundamentals of analytical chemistry; 8 th edition; pg-728 -761 � Y. R. Sharma; Elementary organic chemistry; pg 9 -25 � Gorgor Sandor, ultra violet spectroscopy in pharmaceutical analysis; pg-8 -27.

THANK YOU
- Slides: 34