Chair of Microbiology Virology and Immunology Doctrine about

Chair of Microbiology, Virology, and Immunology Doctrine about antibiotics. Antimicrobial Chemotherapy. Clinical use of antibiotics Lecturer As. O. B. Kuchmak

Lecture schedule 1. History of antibiotics discovery. 2. Classification of antibiotics. 3. Examination of bacterial susceptibility to antibiotics. Complication of antibioticotherapy.

Diarrheal diseases 4 billions cases, Malaria 500 mln, acute infection of respiratory tract 395 mln, sexual transmitted diseases 330 mln, measles 42 mln, whooping cough 40 mln tuberculosis – 1, 9 bln of infected persons, 9 mln of new cases of diseases AIDS – 50 mln cases, 6 mln people died SARS, hemorrhagic fever

Tremendous quantities of antibiotics are produced and released into the environment. 90 – 180 million kg/year of antibiotics are used (enough for 25 BILLION full treatment courses ~ 4 per human/yr!) About 10 X more antibiotics are used in agriculture than to treat people. (Levy 1997 estimated 30 X more in animals than in people).

Modern chemotherapy has been dated to the work of Paul Ehrlich in Germany, who sought systematically to discover effective agents to treat trypanosomiasis and syphilis. He discovered p-rosaniline, which has antitrypanosomal effects, and arsphenamine, which is effective against syphilis. Ehrlich postulated that it would be possible to find chemicals that were selectively toxic for parasites but not toxic to humans. This idea has been called the "magic bullet" concept

Paul Ehrlich
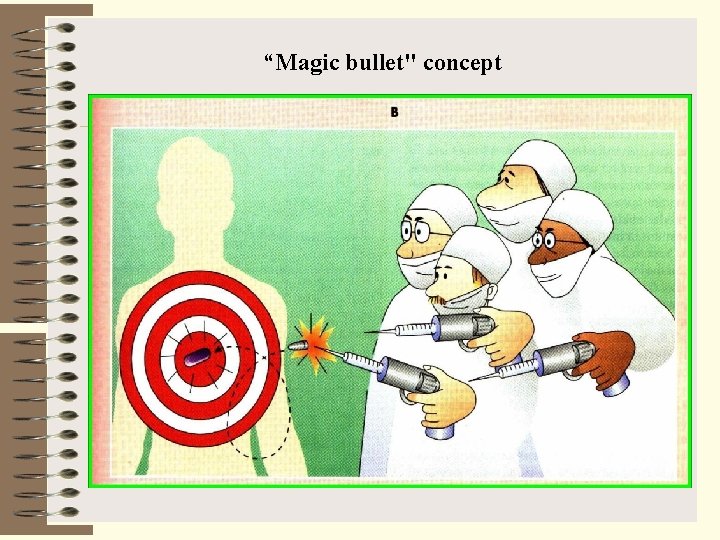
“Magic bullet" concept

Sulfonamides • Analogues of para-aminobenzoic acid • Broad spectrum • Competitive inhibitors of dihydropteroate synthase – needed for folic acid synthesis • Cidal in urine • Mechanisms of resistance Gerhard Domagk gets a Nobel for Medicine, 1939. – Altered affinity of enzyme for drug – Decreased permeability or active efflux – New pathway of folic acid synthesis It had little success until the 1930 s, when Gerhard Domagk discovered the protective effects of prontosil, the forerunner of sulfonamide.

Sir A. Fleming and Penicillin Ironically, penicillin G was discovered fortuitously in 1929 by Fleming, who did not initially appreciate the magnitude of his discovery.

Sir A. Fleming

In 1939 Florey and colleagues at Oxford University again isolated penicillin G. Florey E. Chainy

S. Waksman In 1944 S. Waksman isolated streptomycin and subsequently found agents such as chloramphenicol, tetracyclines, and erythromycin in soil samples.

What are Antibiotics? • Antibiotics means: anti – against, bios – life = • “against life” The term “antibiotics” was proposed in 1942 by S. Waksman • Antibiotics are molecules that stop microbes, both bacteria, viruses, protpzoa, and fungi, from growing or kill them outright. • Antibiotics can be either natural products or manmade synthetic chemicals.

Microbial antagonism is the basis of modern use of antibiotics L. Pasteur

Peculiarities of antibiotics - high level of biological activity - high election specificity Activity of antibiotics is evaluated in International Unit or µg/ml


Spectrum – range of activity of a drug Narrow-spectrum – effective on a small range of microbes target a specific cell component that is found only in certain microbes Broad-spectrum – greatest range of activity target cell components common to most pathogens

Antimicrobial agents may be either: Bacteriostatic Bactericidal Bacteriostatic antibiotics inhibit growth and reproduction of bacteria without killing them. Bacteriostatic agents must work with the immune system to remove the microorganisms from the body. Bacteriostatic antibiotics hamper the growth of bacteria by interfering with bacterial: Protein production DNA replication Cellular metabolism A bacteriocide is a substance that kills the bacteria of choice and, preferably, nothing else. Microbe death is usually achieved by disruption of the bacterial cell membrane leading to lysis. E. g. : Penicillins, Cephalosporins Bactericidal agents are more effective, but bacteriostatic agents can be extremely beneficial since they permit the normal defenses of the host to destroy the microorganisms

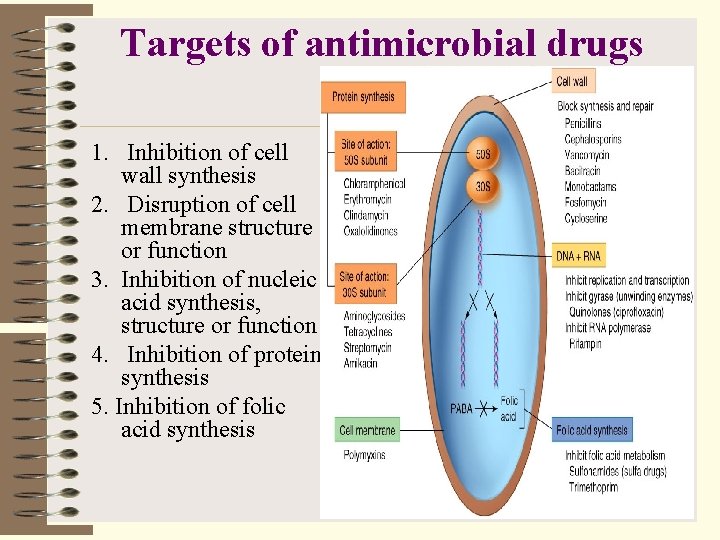
Targets of antimicrobial drugs 1. Inhibition of cell wall synthesis 2. Disruption of cell membrane structure or function 3. Inhibition of nucleic acid synthesis, structure or function 4. Inhibition of protein synthesis 5. Inhibition of folic acid synthesis
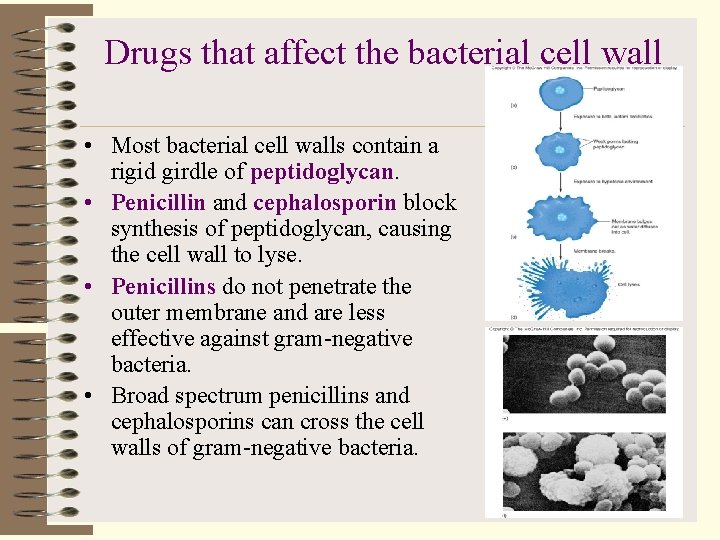
Drugs that affect the bacterial cell wall • Most bacterial cell walls contain a rigid girdle of peptidoglycan. • Penicillin and cephalosporin block synthesis of peptidoglycan, causing the cell wall to lyse. • Penicillins do not penetrate the outer membrane and are less effective against gram-negative bacteria. • Broad spectrum penicillins and cephalosporins can cross the cell walls of gram-negative bacteria.
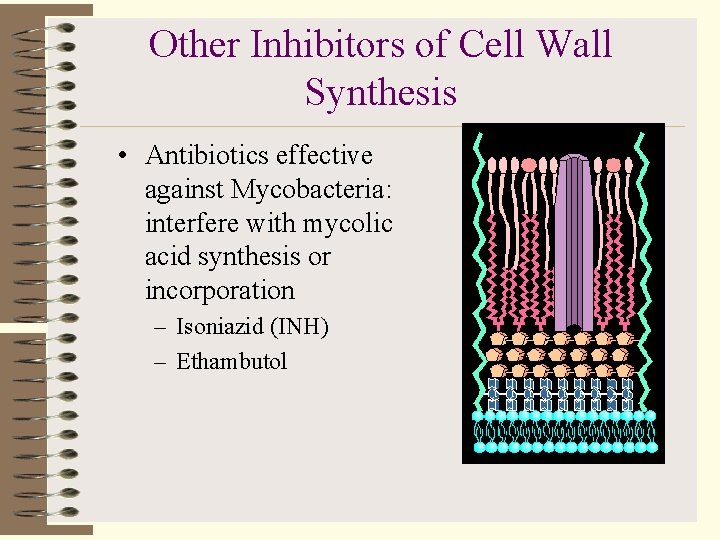
Other Inhibitors of Cell Wall Synthesis • Antibiotics effective against Mycobacteria: interfere with mycolic acid synthesis or incorporation – Isoniazid (INH) – Ethambutol
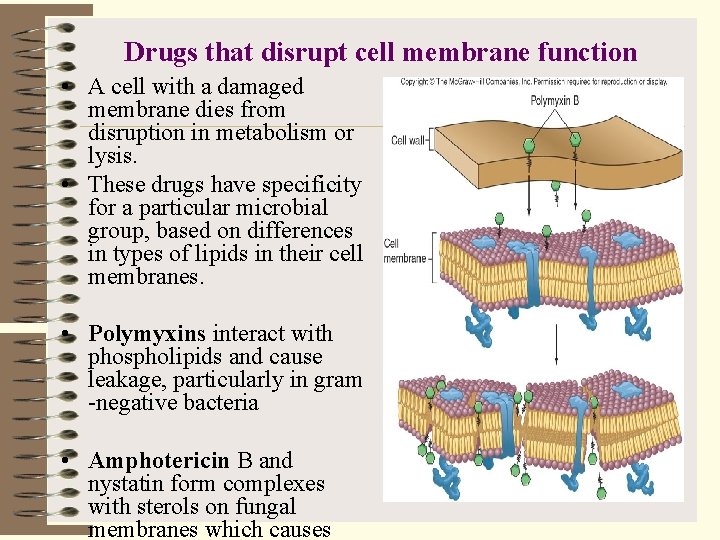
Drugs that disrupt cell membrane function • A cell with a damaged membrane dies from disruption in metabolism or lysis. • These drugs have specificity for a particular microbial group, based on differences in types of lipids in their cell membranes. • Polymyxins interact with phospholipids and cause leakage, particularly in gram -negative bacteria • Amphotericin B and nystatin form complexes with sterols on fungal membranes which causes

Inhibitors of Nucleic Acid Synthesis • Rifamycin – Inhibits RNA synthesis – Antituberculosis • Quinolones and fluoroquinolones – Ciprofloxacin – Inhibits DNA gyrase – Urinary tract infections

Drugs that inhibit nucleic acid synthesis • may block synthesis of nucleotides, inhibit replication, or stop transcription • Sulfonamides and trimethoprim block enzymes required for tetrahydrofolate synthesis needed for DNA & RNA synthesis. • competitive inhibition – drug competes with normal substrate for enzyme’s active site • synergistic effect – an additive effect, achieved by multiple drugs working together, requiring a lower dose of each

Drugs that block protein synthesis Ribosomes of eucaryotes differ in size and structure from procaryotes, so antimicrobics usually have a selective action against procaryotes. But they can also damage the eucaryotic mitochondria. ØAminoglycosides (streptomycin, gentamicin) insert on sites on the 30 S subunit and cause misreading of m. RNA. ØTetracyclines block attachment of t. RNA on the A acceptor site and stop further synthesis. ØMacrolides: Erythromycin (gram +, used in children) ØChloramphenicol
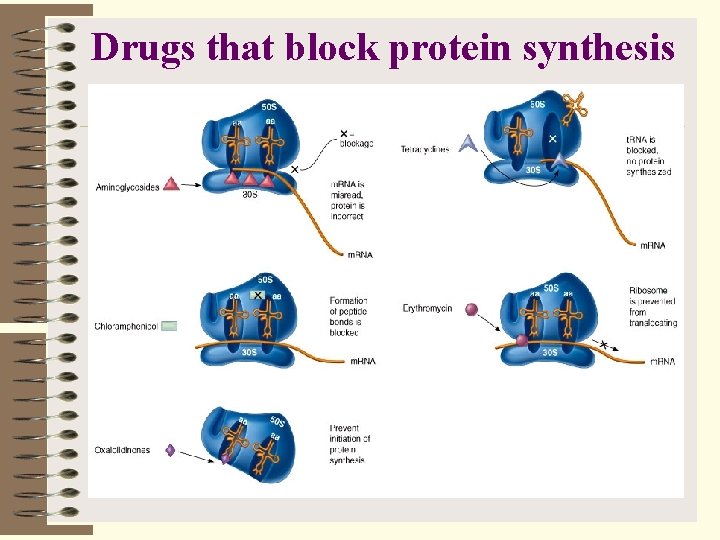
Drugs that block protein synthesis

Penicillins • Large diverse group of compounds • The R group is responsible for the activity of the drug, and cleavage of the beta-lactam ring will render the drug inactive. • Penicillins G and V most important natural forms • Penicillin is the drug of choice for gram-positive cocci (streptococci) and some gram-negative bacteria (meningococci and syphilis spirochete) • Semisynthetic penicillins – ampicillin, carbenicillin & amoxicillin have broader spectra – gram negative enterics rods • Penicillinase-resistant – methicillin, nafcillin, cloxacillin • Primary problems – allergies and resistant strains of bacteria
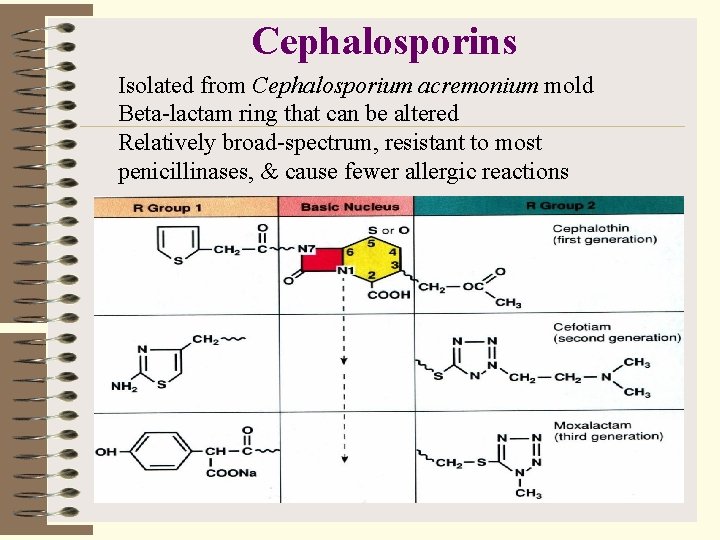
Cephalosporins Isolated from Cephalosporium acremonium mold Beta-lactam ring that can be altered Relatively broad-spectrum, resistant to most penicillinases, & cause fewer allergic reactions

Cephalosporins 5 generations exist Ø 1 st generation – cefazolin, cephalothin – most effective against gram-positive cocci Ø 2 nd generation – cefuroxime, cefaclor, cefonacid – more effective against gram-negative bacteria Ø 3 rd generation – ceftriaxone, cephalexin, cefotaxime – broad-spectrum activity against enteric bacteria with beta-lactamases Ceftriaxone – semisynthetic broad-spectrum drug for treating wide variety of infections Ø 4 th generation - cefepime Ø 5 th generation - ceftobiprole Ø Differ in spectrum, resistance to beta lactamases, penetration into CNS
Vancomycin (Vancocin) Ø Mechanism of action Ø Inhibits bacterial cell wall synthesis Ø Bactericidal? Ø Spectrum – Gram positive bacteria – Methicillin resistant Staphylococcus aureus – Clostridium difficile (oral)
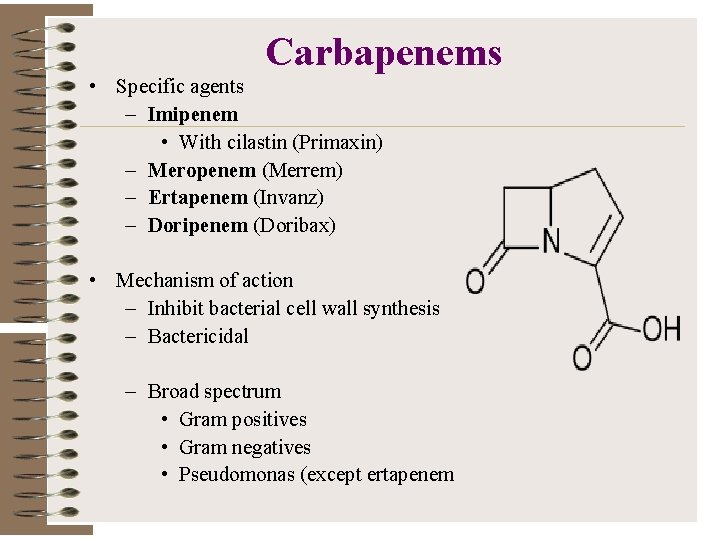
Carbapenems • Specific agents – Imipenem • With cilastin (Primaxin) – Meropenem (Merrem) – Ertapenem (Invanz) – Doripenem (Doribax) • Mechanism of action – Inhibit bacterial cell wall synthesis – Bactericidal – Broad spectrum • Gram positives • Gram negatives • Pseudomonas (except ertapenem
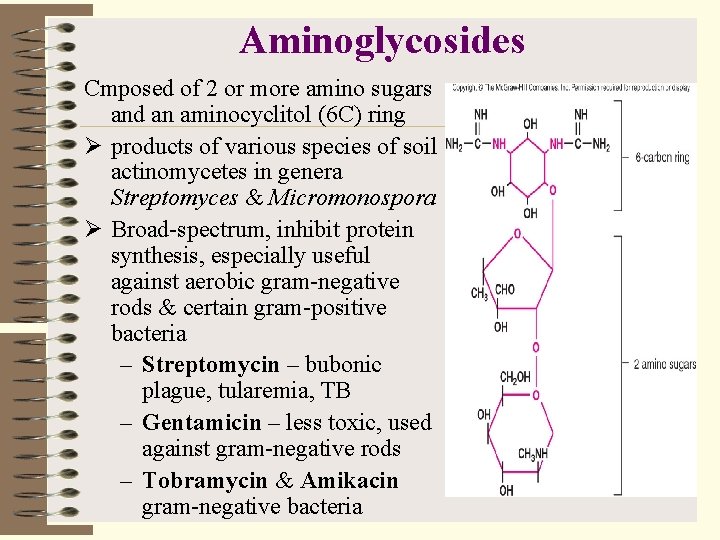
Aminoglycosides Cmposed of 2 or more amino sugars and an aminocyclitol (6 C) ring Ø products of various species of soil actinomycetes in genera Streptomyces & Micromonospora Ø Broad-spectrum, inhibit protein synthesis, especially useful against aerobic gram-negative rods & certain gram-positive bacteria – Streptomycin – bubonic plague, tularemia, TB – Gentamicin – less toxic, used against gram-negative rods – Tobramycin & Amikacin gram-negative bacteria
Macrolides Mechanism of action – Inhibit bacterial protein synthesis – Bacteriostatic • Mechanism of resistance – Decreased permeability of drug into the microbe – Modification of target sites – Hydrolysis of macrolide by bacterial enzymes Specific agents: Erythromycin (attaches to ribosomal 50 s subunit) Azithromycin (Zithromax) Clarithromycin (Biaxin)
Clindamycin • Antimicrobial spectrum – Anaerobes – Some gram positives • Inhibits bacterial protein synthesis • Bacteriostatic • Adverse effects – Nausea, diarrhea – Clostridium difficile
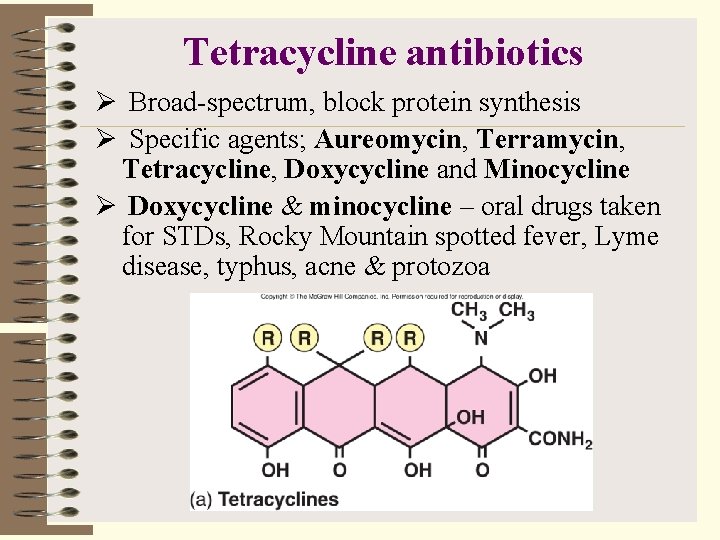
Tetracycline antibiotics Ø Broad-spectrum, block protein synthesis Ø Specific agents; Aureomycin, Terramycin, Tetracycline, Doxycycline and Minocycline Ø Doxycycline & minocycline – oral drugs taken for STDs, Rocky Mountain spotted fever, Lyme disease, typhus, acne & protozoa
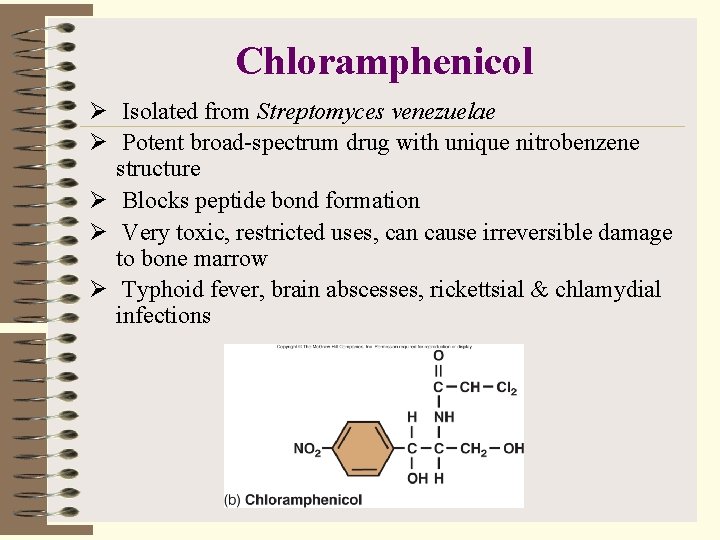
Chloramphenicol Ø Isolated from Streptomyces venezuelae Ø Potent broad-spectrum drug with unique nitrobenzene structure Ø Blocks peptide bond formation Ø Very toxic, restricted uses, can cause irreversible damage to bone marrow Ø Typhoid fever, brain abscesses, rickettsial & chlamydial infections
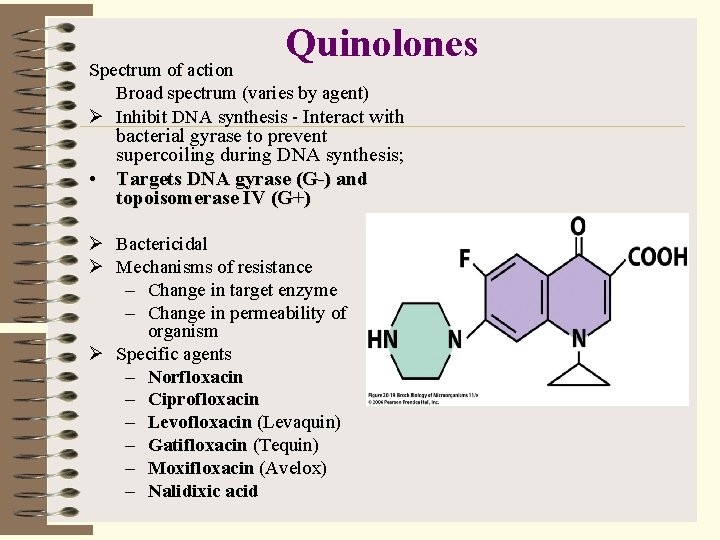
Quinolones Spectrum of action Broad spectrum (varies by agent) Ø Inhibit DNA synthesis - Interact with bacterial gyrase to prevent supercoiling during DNA synthesis; • Targets DNA gyrase (G-) and topoisomerase IV (G+) Ø Bactericidal Ø Mechanisms of resistance – Change in target enzyme – Change in permeability of organism Ø Specific agents – Norfloxacin – Ciprofloxacin – Levofloxacin (Levaquin) – Gatifloxacin (Tequin) – Moxifloxacin (Avelox) – Nalidixic acid
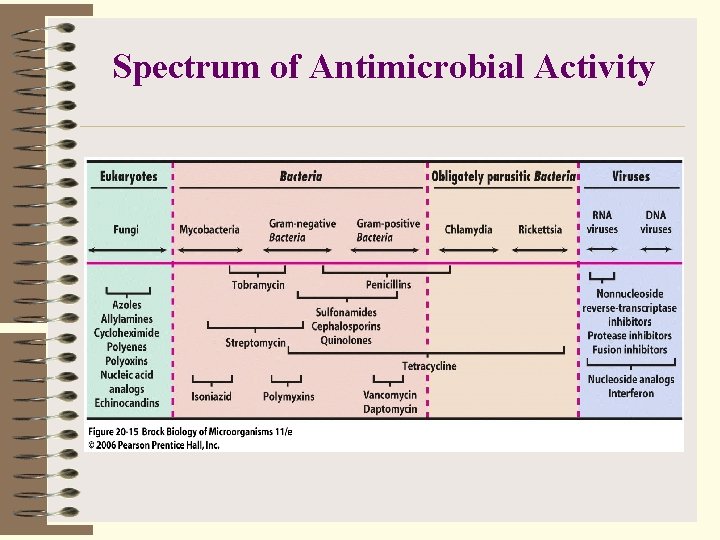
Spectrum of Antimicrobial Activity

Considerations in Selecting an Antimicrobial Drug Testing for Drug Susceptibility: The MIC and Therapeutic Index • In vitro activity of a drug is not always correlated with in vivo effect. – If therapy fails, a different drug, combination of drugs, or different administration must be considered. • Best to choose a drug with highest level of selectivity but lowest level toxicity – measured by therapeutic index – the ratio of the dose of the drug that is toxic to humans as compared to its minimum effective dose • High index is desirable.

Examination of bacteria susceptibility to antibiotics Ø Serial dilutions: - in a liquid medium in a solid medium Ø Disc diffusion method Ø Rapid methods

Demands to nutrient media Ø to be standard and provide optimal conditions for microbial growth; Ø do not have inhibitors of bacterial growth and a lot of stimulators; Ø do not have substances, which inhibit antibiotic activity
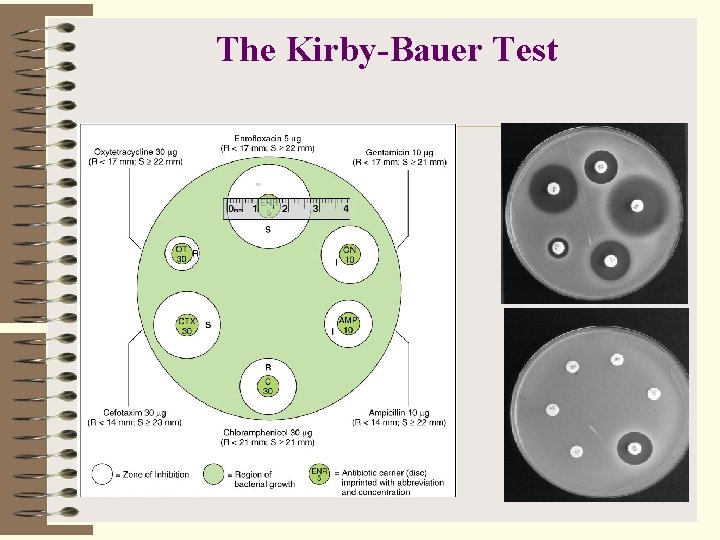
The Kirby-Bauer Test

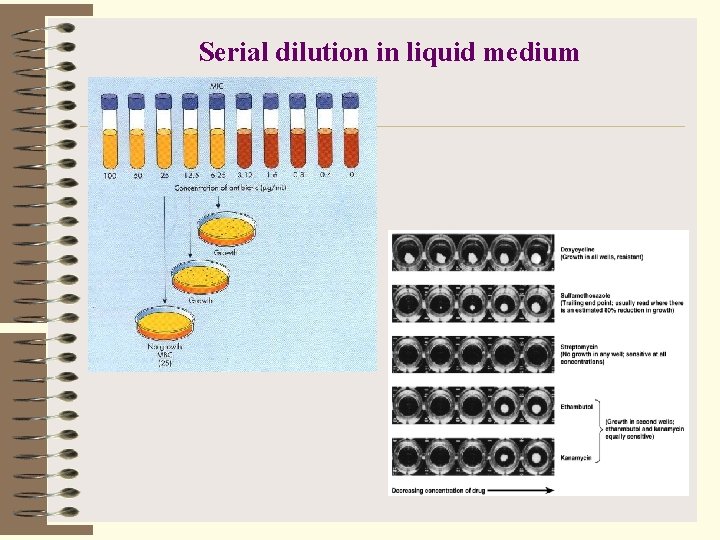
Serial dilution in liquid medium

• Minimum inhibitory concentration (MIC)smallest concentration of drug that visibly inhibits growth • Therapeutic index – the ratio of the dose of the drug that is toxic to humans as compared to its minimum effective dose
Serial dilution in solid medium
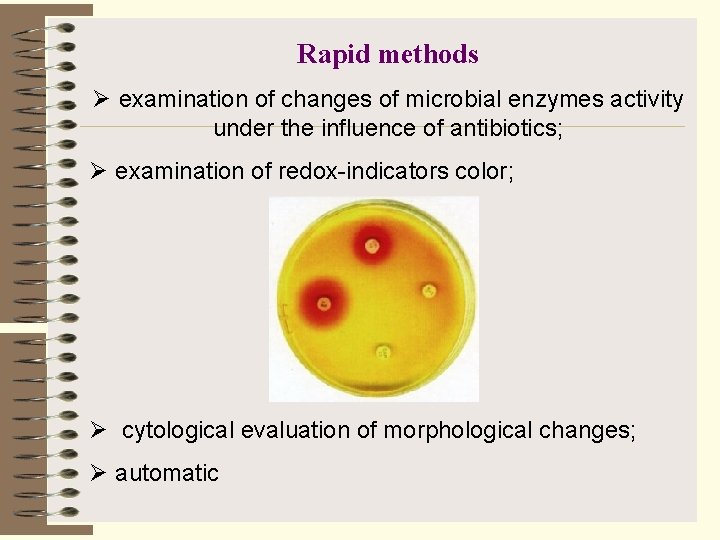
Rapid methods Ø examination of changes of microbial enzymes activity under the influence of antibiotics; Ø examination of redox indicators color; Ø cytological evaluation of morphological changes; Ø automatic
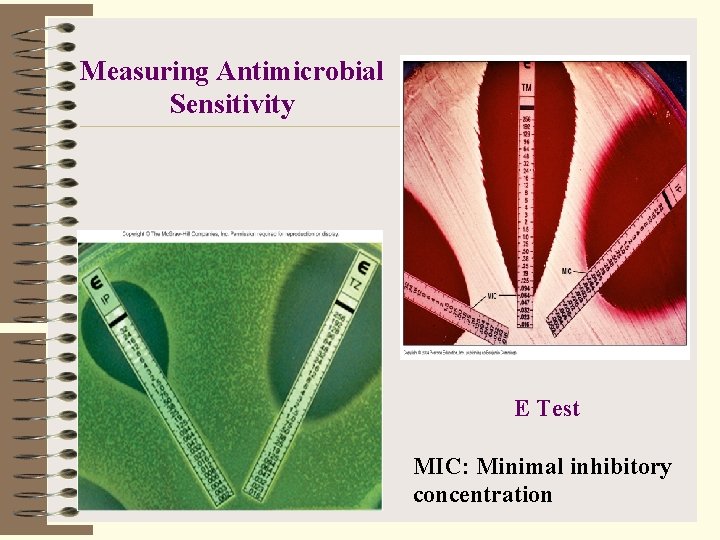
Measuring Antimicrobial Sensitivity E Test MIC: Minimal inhibitory concentration
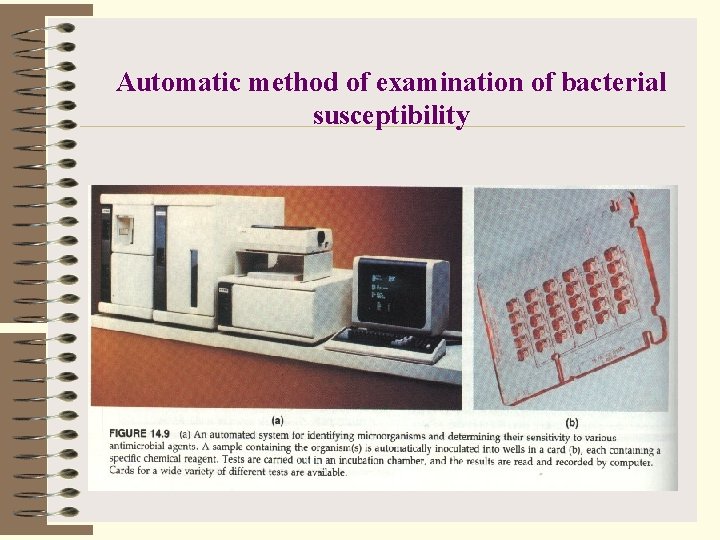
Automatic method of examination of bacterial susceptibility

Considerations in selecting an antimicrobial drug 1. Nature of microbe causing infection 2. Ddegree of microbe’s sensitivity to various drugs 3. Overall medical condition of patient

General principles 1. The first question to ask before prescribing an antibiotic is whether its use is really necessary. There is no point in prescribing it if, for instance, the disease is not due to an infection (fever does not always indicate the presence of an infection), or if the infection is due to agents such as viruses, which do not respond to antibiotics. All therapy is a calculated risk in which the probable benefits must outweigh the draw backs, and antibiotics are no exception to this rule. To use them when they are not indicated and when the "probable benefits" are non existent means exposing the patient to the risk of adverse reactions, or worse.

2. Patients with similar infections react differently. This may be due to previous contact with the same pathogen or to the individual immune response. The presence of hepatic or renal disease may necessitate changes in the dosage or the choice of antibiotic. Knowledge of any past adverse reactions to antibiotics is also essential. 3. The doctor must be familiar with the typical response of infections to proper antibiotic treatment. Acute infection with group A streptococci or pneumococci responds rapidly (usually within 48 hours) to penicillin G, while the temperature curve in typhoid fever treated with chloramphenicol may not show any change for four or five days.

4. The doctor must know which bacteria are commonly found in which situations, for instance Pseudomonas in extensive burns (sepsis is frequent and often fatal) and in the expectoration of children with cystic fibrosis, or Streptococcus pneumoniae and Haemophilus influenzae in chronic bronchitis of the adult.

5. Ideally, treatment with antibiotics should not be instituted before samples for sensitivity testing have been collected. Such tests can be dispensed with, however, when the causative organism is known and its response to the antibiotic is predictable. But the sensitivity of, for instance, many gram negative strains can change, even during treatment, making an alternative treatment necessary. In addition, the clinical results may be at odds with the findings of the sensitivity tests. Even a severe infection may show a satisfactory clinical response despite apparent lack of sensitivity.

Failure of antibiotic therapy Antibiotic treatment is considered a failure if no response is seen within three days. Failure may be due to various causes: 1. Wrong diagnosis (a viral infection does not respond to antibiotics). 2. Wrong choice of antibiotic. 3. Wrong dosage (wrongly dosed by doctor or poor patient compliance). 4. Development of resistance during therapy (as sometimes occurs in tuberculosis and infections due to gram negative pathogens). 5. Superinfection by resistant bacteria. 6. Accumulation of pus necessitating surgical drainage (buttock abscess). 7. Underlying disease (lymphoma, neoplasia) of which the infection is only an intercurrent complication. 8. Drug fever.

Secondary action of antibiotics І. Allergic reactions Ø dangerous for life (anaphylactic shock, angioneurotic oedema of larynx) Ø dangerous for life (skin itching, urticaria, rash, rhinitis, glossitis, conjunctivitis, photodermatoses (tetracyclines) ІІ. Toxic reactions Ø dangerous for life (agranulocytosis, aplastic anemia, endotoxic shock) Ø non dangerous (neuritis of N. vestibularis and N. auricularis aminoglycosides; periferal neuritis, vomiting, nausea, diarrhea, hepatotoxic and nephrotoxic effects, embriotoxic effect (pigmentation of the teeth)
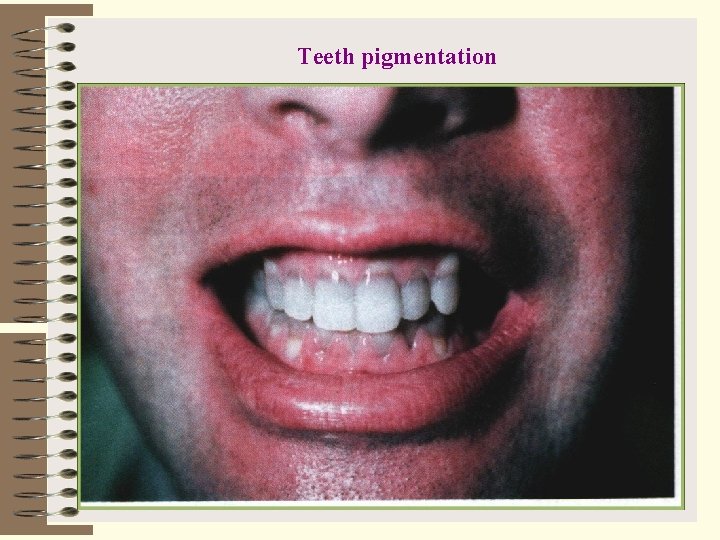
Teeth pigmentation
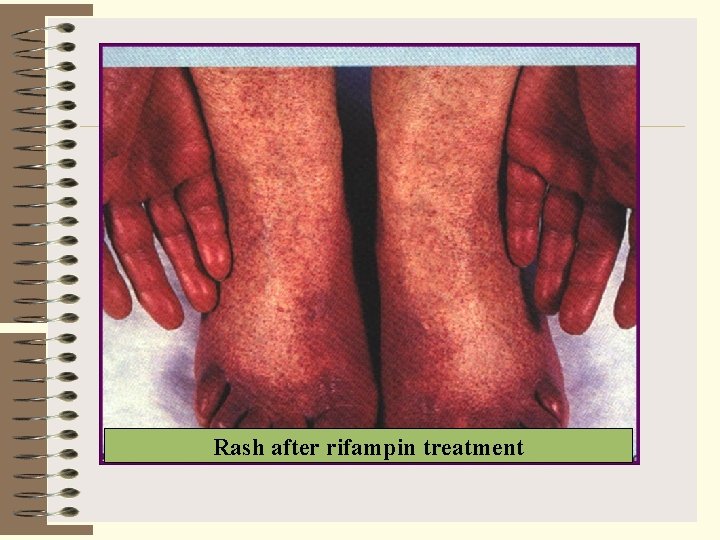
Rash after rifampin treatment
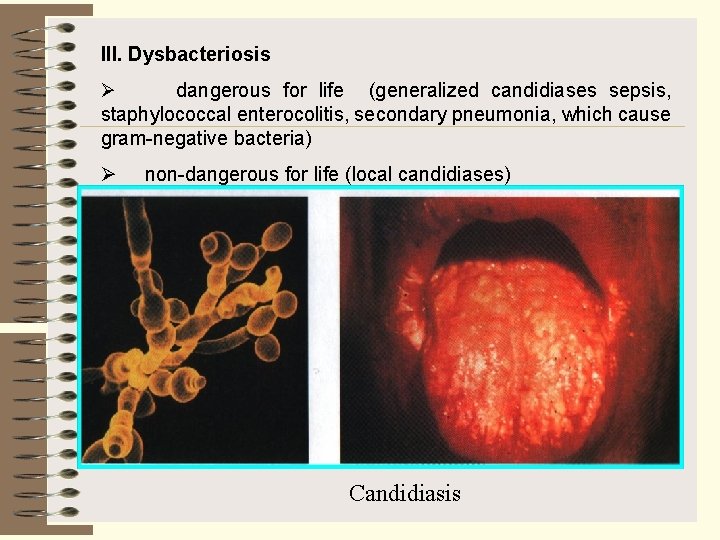
ІІІ. Dysbacteriosis Ø dangerous for life (generalized candidiases sepsis, staphylococcal enterocolitis, secondary pneumonia, which cause gram negative bacteria) Ø non dangerous for life (local candidiases) Candidiasis

Antibiotic Resistance – What is it? Antibiotic resistance – when bacteria change eliminating the effectiveness of the drug designed to cure or prevent infection – How it happens? • Bacteria survive antibiotic control and continue to multiply into resistant strains • Sensitive “S” • Resistant “R” - microbe is inhibited. - microbe unaffected.

Timeline of Antibiotic Resistance Ø 1929 – Alexander Fleming discovers the first antibiotic, Penicillin Ø 1942 – Penicillin available through mass production Ø 1954 – 2 million pounds of antibiotics produced in the United States annually Ø 1960’s – Various resistant strains emerging due to abused antibiotic use Ø Today – 50 million pounds of antibiotics produced in the United There is probably no States annually chemotherapeutic drug to which in suitable circumstances the bacteria cannot react in some way acquiring fastness (resistance) Alexander Fleming

Antibiotic Resistance Ø Innate resistance Ø Acquired resistance (primary or secondary): Ø Mutations of existing genes (stepwise) Ø Acquisition of foreign DNA • Plasmid exchange (conjugation, transduction) • Transformation • Transposons Ø Clonal spread Ø All promoted by antibiotic use!

Resistance Mechanisms Ø Microbe lacks structure the antibiotic attaches to Ø Microbe impermeable to antibiotic Ø Microbe can modify antibiotic to inactive form (Drug inactivation – penicillinases) Ø Microbe may modify the target of the antibiotic Ø Microbe may develop a resistant biochemical pathway Microbe may pump out an antibiotic entering the cell
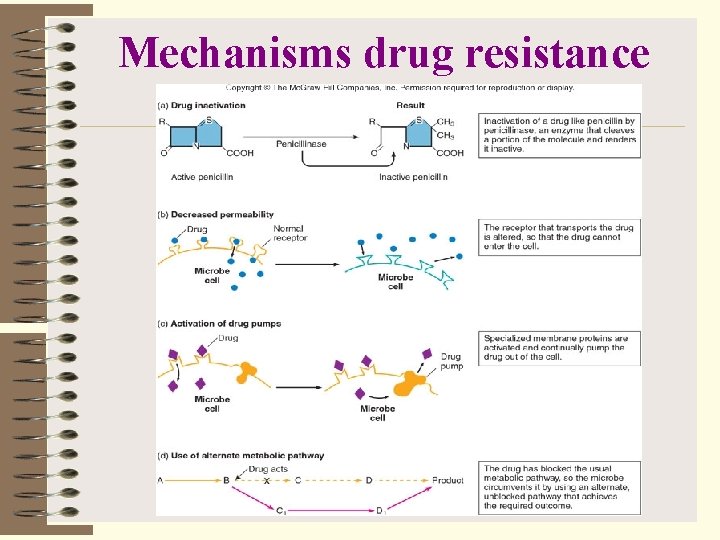
Mechanisms drug resistance
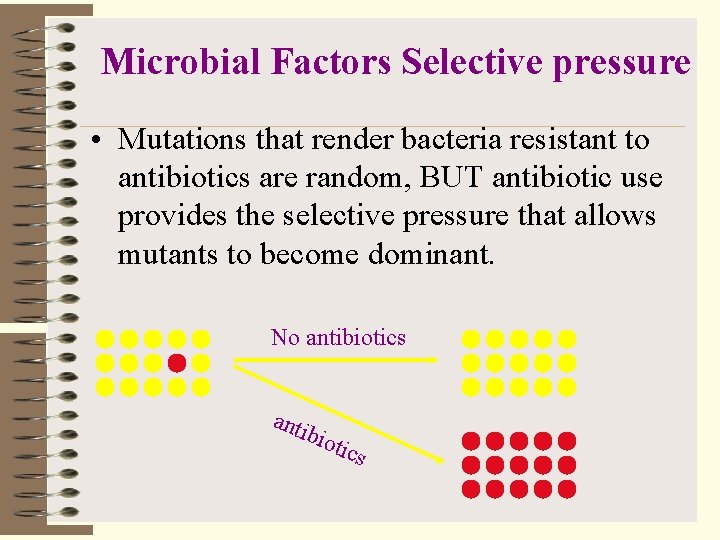
Microbial Factors Selective pressure • Mutations that render bacteria resistant to antibiotics are random, BUT antibiotic use provides the selective pressure that allows mutants to become dominant. No antibiotics anti bio tics
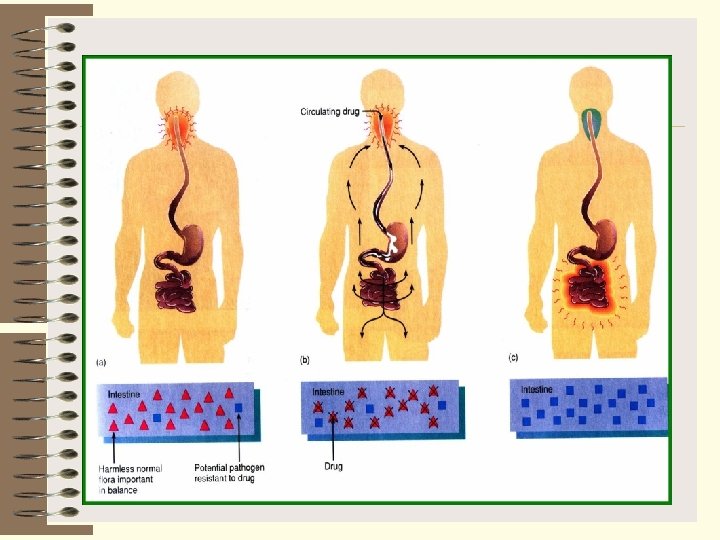

Resistance by Replication • Resistant bacteria usually have a gene that makes the antibiotic ineffective – Surviving bacteria will replicate • Bacteria have plasmids that allow genes to move between different types of bacteria – Bacteria that was previously susceptible to antibiotic now have the resistant gene • Dead bacterial cells give off DNA that can be incorporated into living bacteria allowing it to become resistant (transformation)
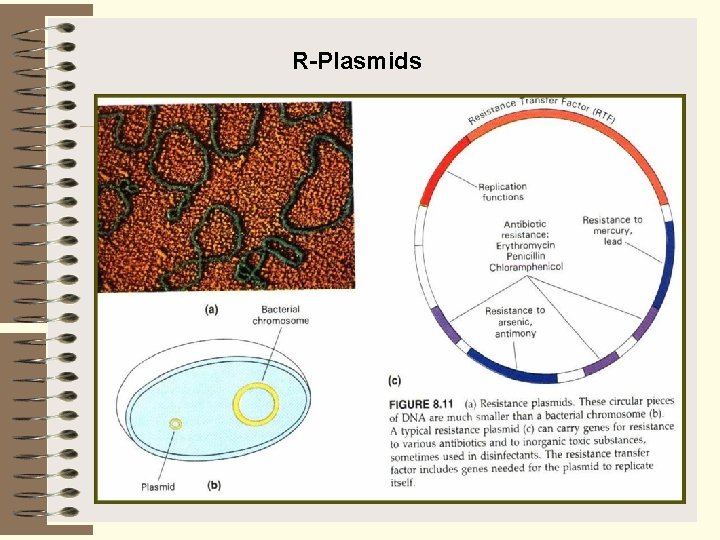
R-Plasmids
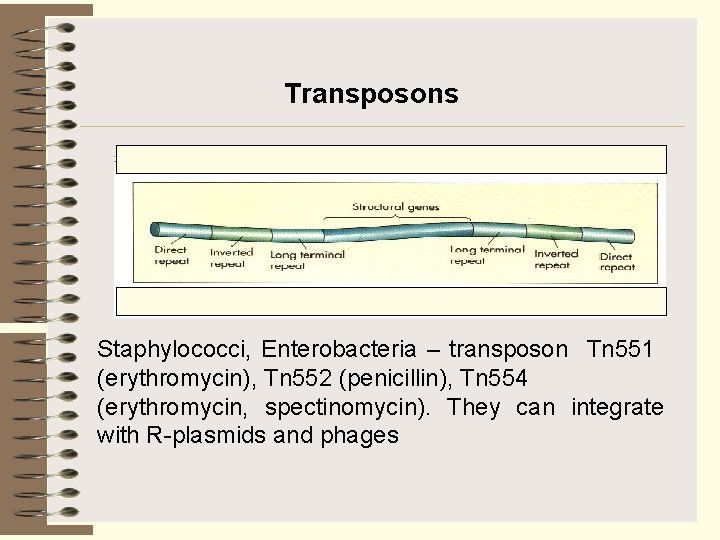
Transposons Staphylococci, Enterobacteria – transposon Tn 551 (erythromycin), Tn 552 (penicillin), Tn 554 (erythromycin, spectinomycin). They can integrate with R plasmids and phages

Overcoming Antibiotic Resistance Ø Altering the use of existing antibiotics: – Decrease the duration of the antibiotic so the organism does not create resistance – Increase the dosage of the antibiotic for a higher concentration of drug – Discontinue use of an antibiotic for a period of time Ø Rotation of antibiotics used in treatment – Especially useful when used with last resort treatments Ø Combination of antibiotics in a treatment – Minimizes possibility of resistance since the organism needs two ways to get rid of antibiotics http: //www. pharmacist. com/pdf/combating_antibiotic_res_sr. pdf

Prevention of Antibiotic Resistance Ø Only use an antibiotic when they are likely to be beneficial Ø Do not take an antibiotic for a viral infection like a cold, most sore throats or flu Ø Do not save any of your antibiotic prescription Ø Take an antibiotic exactly as the doctor tells you Ø Do not take an antibiotic that is prescribed for someone else

Alternative Treatments for Antibiotics Ø Inhibitors that will neutralize the organism’s ability to become resistant Ø Antibiotic will attack a different site of bacteria than normal that allows it to be effective Ø Chemicals with antibiotic qualities are being used to kill organisms Ø Current antibiotics are made from microbes and fungi Ø Bacteriophages (virus that attacks bacteria) are being altered http: //www. pharmacist. com/pdf/combating_antibiotic_res_sr. pdf

The Future of Chemotherapeutic Agents Antisense agents – Complementary DNA or peptide nucleic acids that binds to a pathogen's virulence gene(s) and prevents transcription

- Slides: 75