Chagas Tests Development and Standardization Glucia ParanhosBaccal Emerging

Chagas Tests: Development and Standardization Gláucia Paranhos-Baccalà Emerging Pathogens Department bio. Mérieux International Biological Reference Preparations for Chagas Diagnostic Test WHO – Geneva January, 26 th and 28 th 2009 1
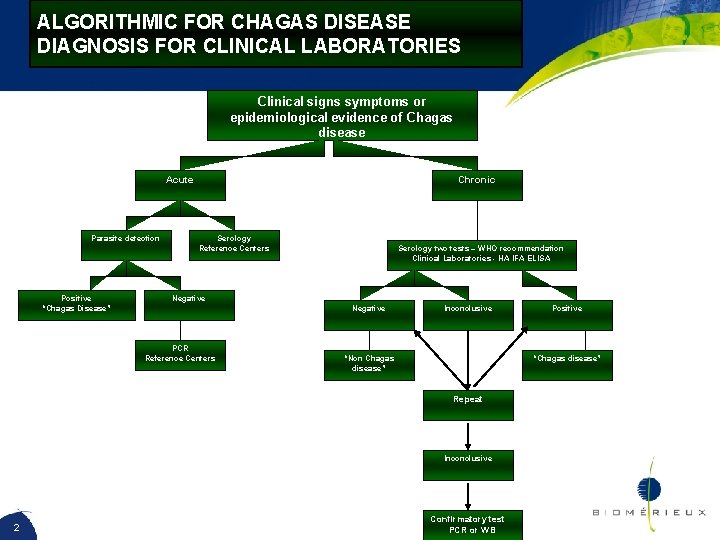
ALGORITHMIC FOR CHAGAS DISEASE DIAGNOSIS FOR CLINICAL LABORATORIES Clinical signs symptoms or epidemiological evidence of Chagas disease Acute Parasite detection Positive “Chagas Disease” Chronic Serology Reference Centers Serology two tests – WHO recommendation Clinical Laboratories - HA IFA ELISA Negative PCR Reference Centers Inconclusive “Non Chagas disease” “Chagas disease” Repeat Inconclusive 2 Positive Confirmatory test PCR or WB

Chagas Tests: Diagnosis Eu Market Blood Screening Ø Direct Transmission through transfusions, by organ transplantation or laboratory accident Adequate cost tests Needs for Chagas test High Sensitivity Good Specificity 3 Antibody detection

Epidemiology In France: In 3 years 14 cases against 5 in 30 years. ØNumber of migrants from Latin or Central America not well known in EU Real emergence or cluster effect? 4
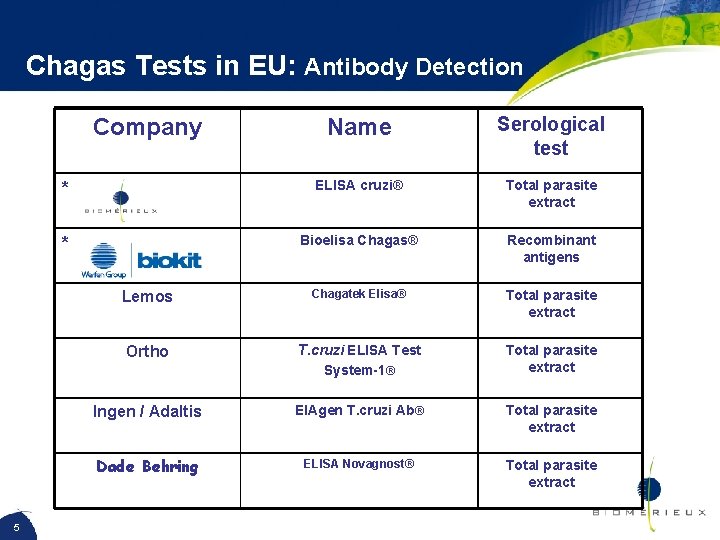
Chagas Tests in EU: Antibody Detection Name Serological test * ELISA cruzi® Total parasite extract * Bioelisa Chagas® Recombinant antigens Lemos Chagatek Elisa® Total parasite extract Ortho T. cruzi ELISA Test System-1® Total parasite extract Ingen / Adaltis EIAgen T. cruzi Ab® Total parasite extract Dade Behring ELISA Novagnost® Total parasite extract Company 5

T. cruzi antigens preparations for diagnostic tests based on Ab detection § Crude epimastigote extracts § Epimastigote alcaline extractions § Purified epimastigotes fractions, … Antigen preparation could present variations Batch to batch changes Lost of some epimastigotes epitopes or strain specific? 6

bio. Mérieux Vironostika® 7

Kit Elisa Cruzi: for antibody detection Kit: Ref: Number of tests: ELISA CRUZI 35117 192 tests 2 microplates of 96 tests: 12 barrettes de 8 wells. Human serum or plasma: citrate, heparine, oxalate ou EDTA. Epimastigotes antigens Results: Positif / Doubts / Negatif. adsorbance index : Timing: Controls : 1 < Retest < 0, 8 70 minutes of incubation. 2 pos Controls and 3 neg Controls in each test. If necessary: one well reserved for the R 3 diluant solution to callibrate the reader (blanc). 8

Chagas Test Standardization for antibody detection The following steps are used for standardization: • Sera panel set-up (negative and positive controls); • Antigen production and titers: batch to batch; • anti-Ig. G human conjugate; • Cut-off; • The test interpretation. 9

The Chagas disease immunoassay standardization test for antibody detection at bio. Mérieux is based on the Sensitivity, Specificity and Reproducibility with the following steps listed bellow: 1. Sera panel set-up: A positive and negative sera panel with samples from a serological screening of blood donors in an hemocenter service is obtained -positive sera panel is: Each lot of Chagas kit is tested with about 200 positive sera from chagasic patients of different endemic regions from Brazil with varying degrees of reactivity. The positive panel was evaluated in at least in two reference tests as Immunofluorescence assay, indirect hemagglutination. The titers of each positive serum included in the panel was previously determined. Bio. Mérieux has a positive sera panel (n=40) representing others endemic areas located at Argentina, Bolivia, Venezuela and Mexico. This precious panel is used to evaluate the final lot. -negative sera panel is: Each lot of Chagas kit is tested with about 2000 negative samples and positive for others pathologies as: hepatitis, malaria, syphilis, lupus, HIV, HCV, HTLV-I. -potential cross-reactivity panel is: Each lot of Chagas kit is tested with about 80 Leishmaniasis sera samples. This test has been calibrated against in house standards. test. 10

The Chagas disease immunoassay standardization test for antibody detection at bio. Mérieux Antigen titers: bio. Mérieux used as antigen a crude extract obtained from alkaline extraction from T. cruzi II epimastigotes forms. • The total protein of the parasite is estimated by a colorimetric method in each lot of the T. cruzi production. • Evaluation with lot to lot antigen production in terms of title of antigen dilution with the Chagas panel sera and a target value obtained from an already tested lot. • The stability of the antigen production is also evaluated during the time and temperature. • The antigen preparation should present NO variation between serum or lot to lot. The cut-off is evaluated for each antigen production after stability and must be have a variation with less than 10%. At each antigen preparation three pilot lots are produced. 11

The Chagas disease immunoassay standardization test for antibody detection at bio. Mérieux Human conjugate: The mouse monoclonal anti-human Ig. G antibodies conjugated to enzyme is commercially acquired analyzed in each lot to assure the performance of the Chagas disease kit production. The cut-off: The cut off is calculated from results obtained from negative sera panel. Negative sera panel should included positive sera from possible cross-reactive infections. The exact cut-off and indeterminate values are determined by by Roc curve and Youden coefficient. 12
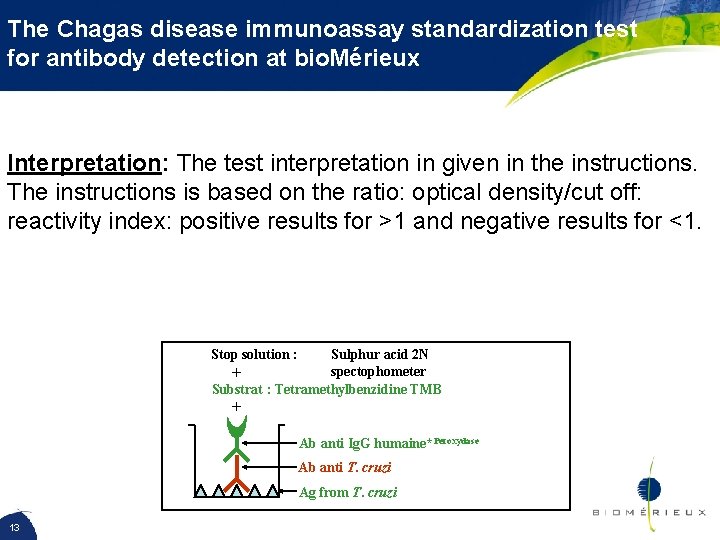
The Chagas disease immunoassay standardization test for antibody detection at bio. Mérieux Interpretation: The test interpretation in given in the instructions. The instructions is based on the ratio: optical density/cut off: reactivity index: positive results for >1 and negative results for <1. Stop solution : Sulphur acid 2 N spectophometer + Substrat : Tetramethylbenzidine TMB + Ab anti Ig. G humaine*Peroxydase Ab anti T. cruzi Ag from T. cruzi 13

Confirmatory tests for Chagas Disease Blood Donors and co-endemic areas Western Blot: TESA cruzi WB (bio. Mérieux) - Trypomastigote excreted-secreted antigens 14

15

TESAcruzi - Parameters Serum panel bio. Mérieux + serum from blood banks 16

APPLICATIONS 17

SEROEPIDEMIOLOGICAL SURVEY ON CHAGAS DISEASE PREVALENCE AMONG CHILDREN N = 100, 000 Blood samples on filter paper Central Laboratory ELISA - IIF Reference Laboratory 10% of blood samples + Reagents + Inconclusives IIF, ELISA and IHA Reagents or inconclusives – TESA cruzi Results Reagents samples Identification of childen + contactants Venous Blood 18

SEROEPIDEMIOLOGICAL SURVEY ON CHAGAS DISEASE PREVALENCE AMONG CHILDREN Central Laboratory – 80, 000 blood samples ELISA - IIF Reference Laboratory – 8, 788 10% of blood samples + Reagents + Inconclusives Indirect Immunofluorescence -1/40 - 313 (3. 6%) ELISA - 166 (1. 9%) TESAcruzi – 77 (0. 9%) 19
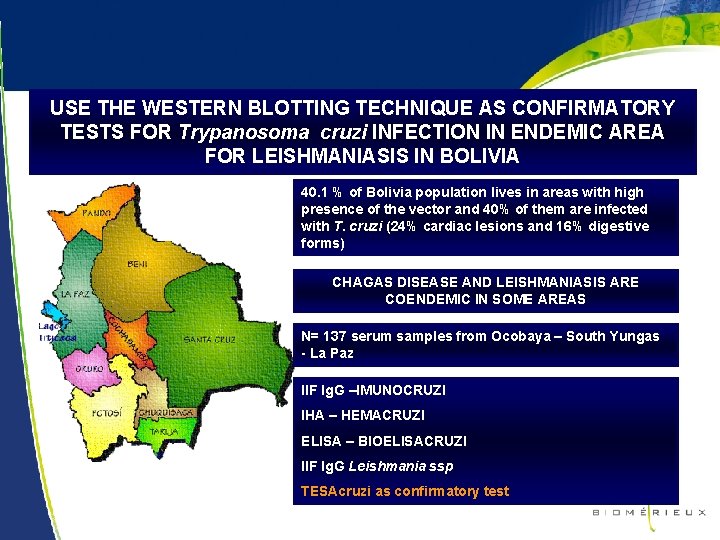
USE THE WESTERN BLOTTING TECHNIQUE AS CONFIRMATORY TESTS FOR Trypanosoma cruzi INFECTION IN ENDEMIC AREA FOR LEISHMANIASIS IN BOLIVIA 40. 1 % of Bolivia population lives in areas with high presence of the vector and 40% of them are infected with T. cruzi (24% cardiac lesions and 16% digestive forms) CHAGAS DISEASE AND LEISHMANIASIS ARE COENDEMIC IN SOME AREAS N= 137 serum samples from Ocobaya – South Yungas - La Paz IIF Ig. G –IMUNOCRUZI IHA – HEMACRUZI ELISA – BIOELISACRUZI IIF Ig. G Leishmania ssp TESAcruzi as confirmatory test 20
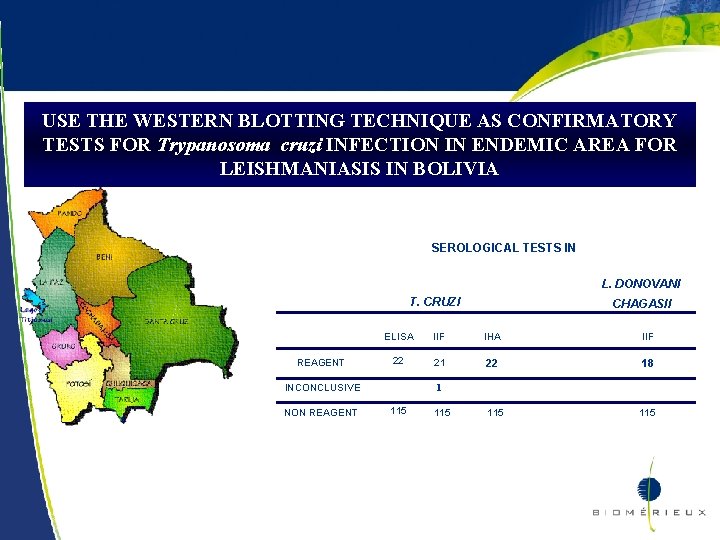
USE THE WESTERN BLOTTING TECHNIQUE AS CONFIRMATORY TESTS FOR Trypanosoma cruzi INFECTION IN ENDEMIC AREA FOR LEISHMANIASIS IN BOLIVIA SEROLOGICAL TESTS IN L. DONOVANI T. CRUZI REAGENT ELISA IIF IHA IIF 22 21 22 18 115 INCONCLUSIVE NON REAGENT 21 CHAGASII 1 115
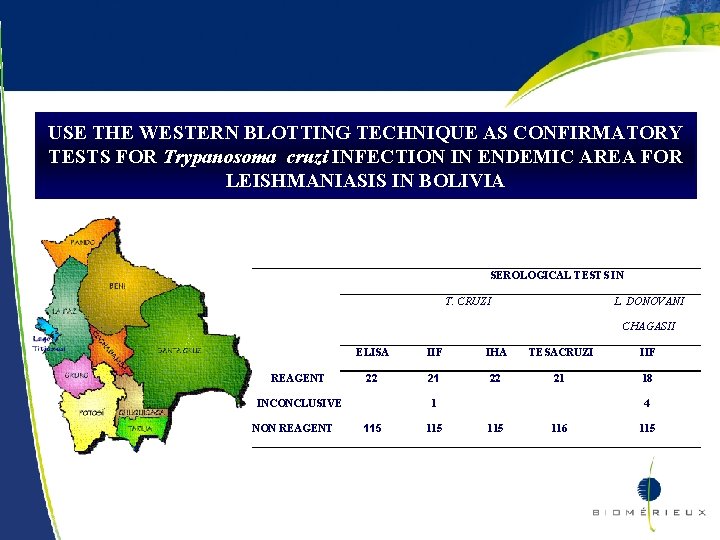
USE THE WESTERN BLOTTING TECHNIQUE AS CONFIRMATORY TESTS FOR Trypanosoma cruzi INFECTION IN ENDEMIC AREA FOR LEISHMANIASIS IN BOLIVIA SEROLOGICAL TESTS IN T. CRUZI L. DONOVANI CHAGASII REAGENT ELISA IIF IHA TESACRUZI IIF 22 21 18 INCONCLUSIVE NON REAGENT 22 1 115 4 115 116 115

Emerging Pathogens Department bio. Mérieux SA Tour CERVI IFR 128 Bio. Sciences Lyon Gerland 23
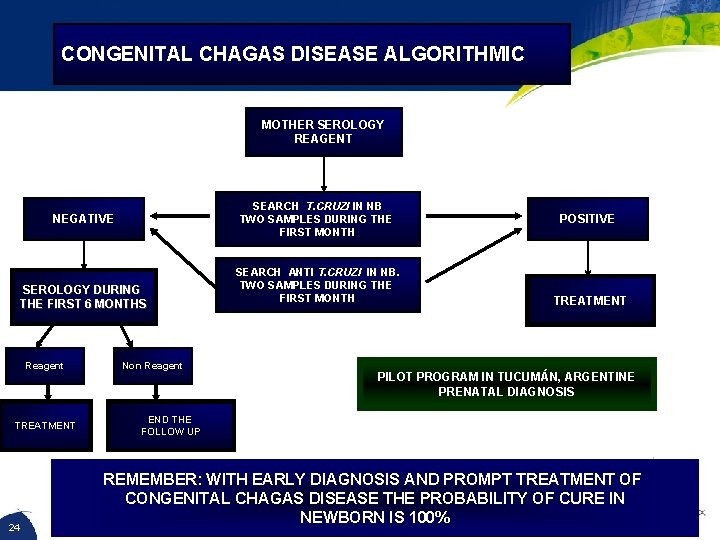
CONGENITAL CHAGAS DISEASE ALGORITHMIC MOTHER SEROLOGY REAGENT SEARCH T. CRUZI IN NB TWO SAMPLES DURING THE FIRST MONTH NEGATIVE SEROLOGY DURING THE FIRST 6 MONTHS Reagent TREATMENT 24 Non Reagent SEARCH ANTI T. CRUZI IN NB. TWO SAMPLES DURING THE FIRST MONTH POSITIVE TREATMENT PILOT PROGRAM IN TUCUMÁN, ARGENTINE PRENATAL DIAGNOSIS END THE FOLLOW UP REMEMBER: WITH EARLY DIAGNOSIS AND PROMPT TREATMENT OF CONGENITAL CHAGAS DISEASE THE PROBABILITY OF CURE IN NEWBORN IS 100%
Vironostika® 25 Kit Composition:
- Slides: 25