Ch 7 Membrane Structure and Function Separates cell
Ch. 7 Membrane Structure and Function Separates cell from environment Controls traffic Selectively permeable Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
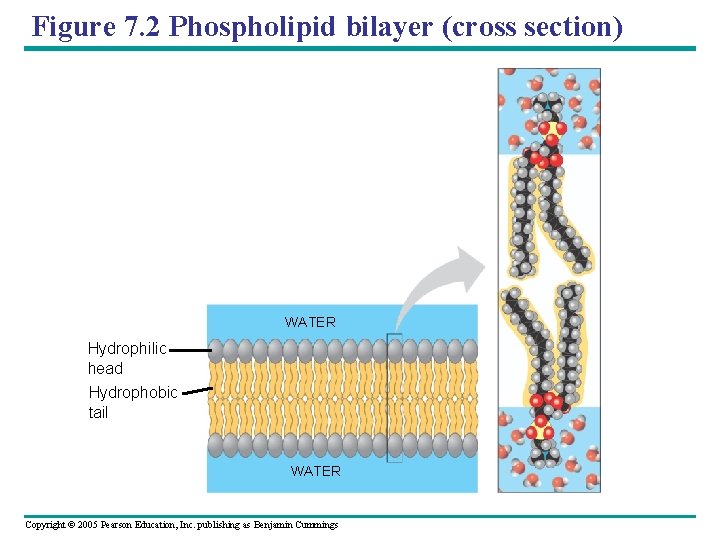
Figure 7. 2 Phospholipid bilayer (cross section) WATER Hydrophilic head Hydrophobic tail WATER Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
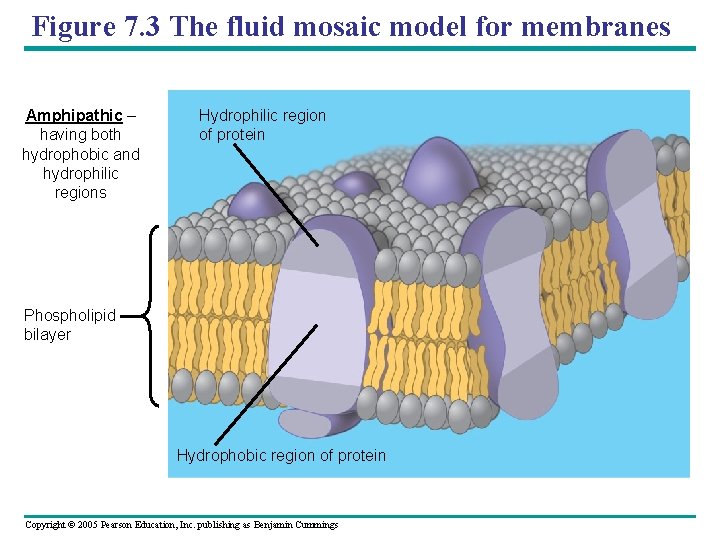
Figure 7. 3 The fluid mosaic model for membranes Amphipathic – having both hydrophobic and hydrophilic regions Hydrophilic region of protein Phospholipid bilayer Hydrophobic region of protein Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
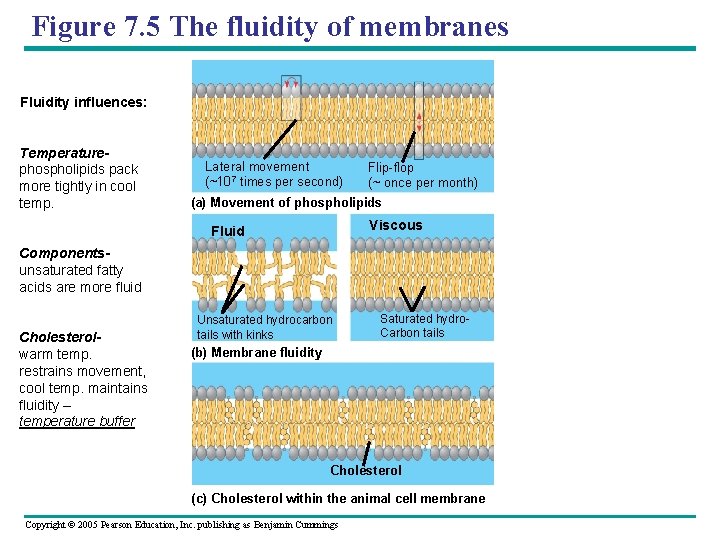
Figure 7. 5 The fluidity of membranes Fluidity influences: Temperaturephospholipids pack more tightly in cool temp. Lateral movement (~107 times per second) Flip-flop (~ once per month) (a) Movement of phospholipids Viscous Fluid Componentsunsaturated fatty acids are more fluid Cholesterolwarm temp. restrains movement, cool temp. maintains fluidity – temperature buffer Unsaturated hydrocarbon tails with kinks Saturated hydro. Carbon tails (b) Membrane fluidity Cholesterol (c) Cholesterol within the animal cell membrane Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
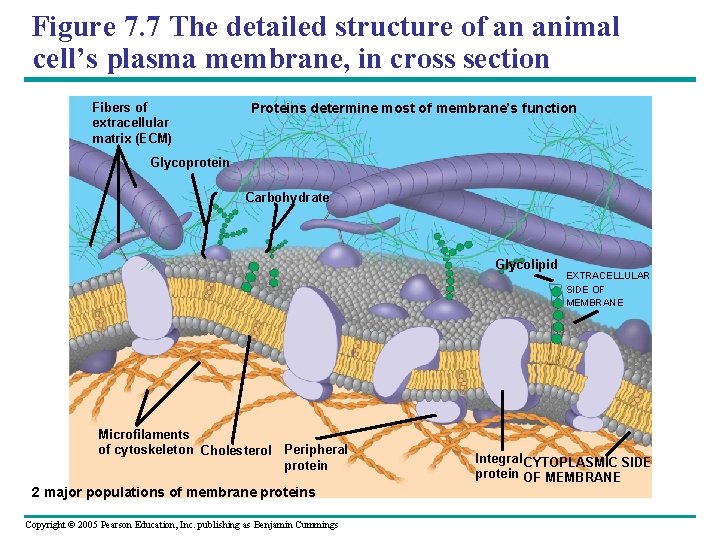
Figure 7. 7 The detailed structure of an animal cell’s plasma membrane, in cross section Fibers of extracellular matrix (ECM) Proteins determine most of membrane’s function Glycoprotein Carbohydrate Glycolipid Microfilaments of cytoskeleton Cholesterol Peripheral protein 2 major populations of membrane proteins Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings EXTRACELLULAR SIDE OF MEMBRANE Integral CYTOPLASMIC SIDE protein OF MEMBRANE
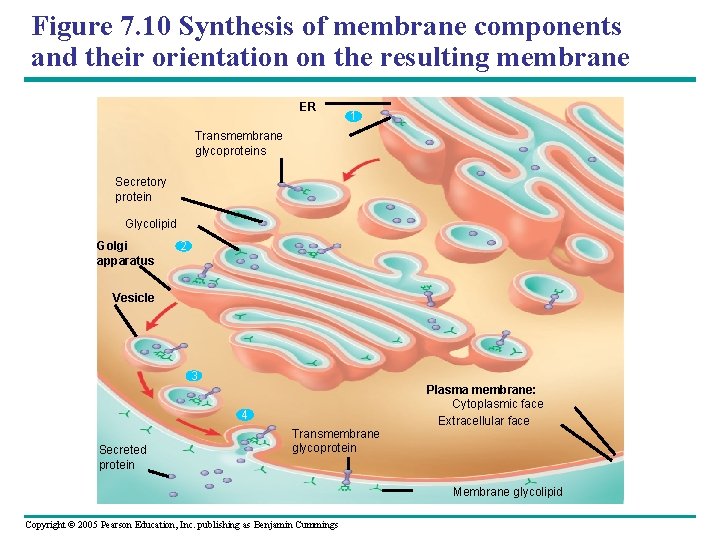
Figure 7. 10 Synthesis of membrane components and their orientation on the resulting membrane ER 1 Transmembrane glycoproteins Secretory protein Glycolipid Golgi apparatus 2 Vesicle 3 4 Secreted protein Transmembrane glycoprotein Plasma membrane: Cytoplasmic face Extracellular face Membrane glycolipid Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
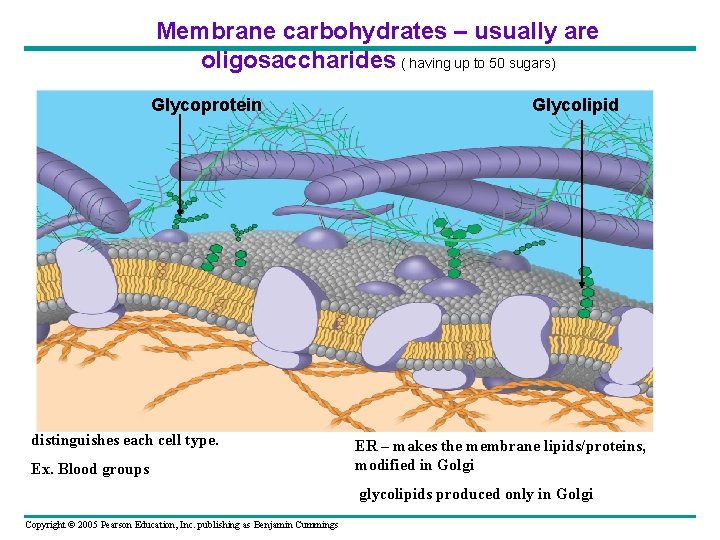
Membrane carbohydrates – usually are oligosaccharides ( having up to 50 sugars) Glycoprotein distinguishes each cell type. Ex. Blood groups Glycolipid ER – makes the membrane lipids/proteins, modified in Golgi glycolipids produced only in Golgi Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
Membrane carbohydrates • Recognition – cells bind to surface molecules on the plasma membrane • Basis for rejection of foreign cells by immune sys. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
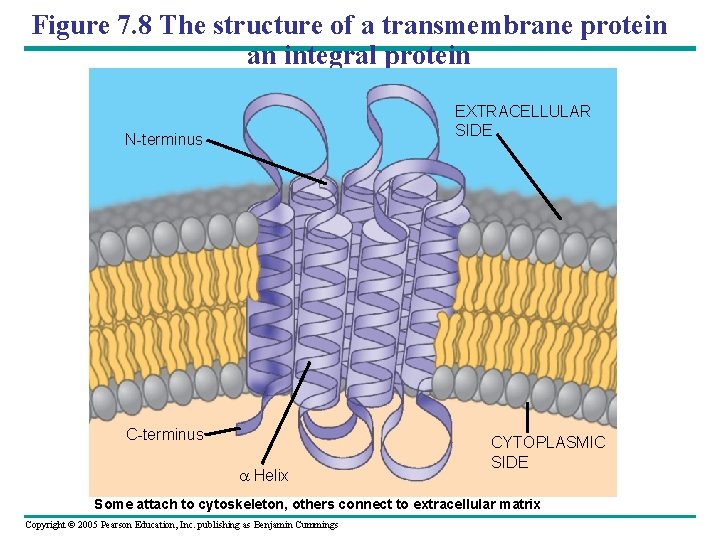
Figure 7. 8 The structure of a transmembrane protein an integral protein EXTRACELLULAR SIDE N-terminus C-terminus a Helix CYTOPLASMIC SIDE Some attach to cytoskeleton, others connect to extracellular matrix Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
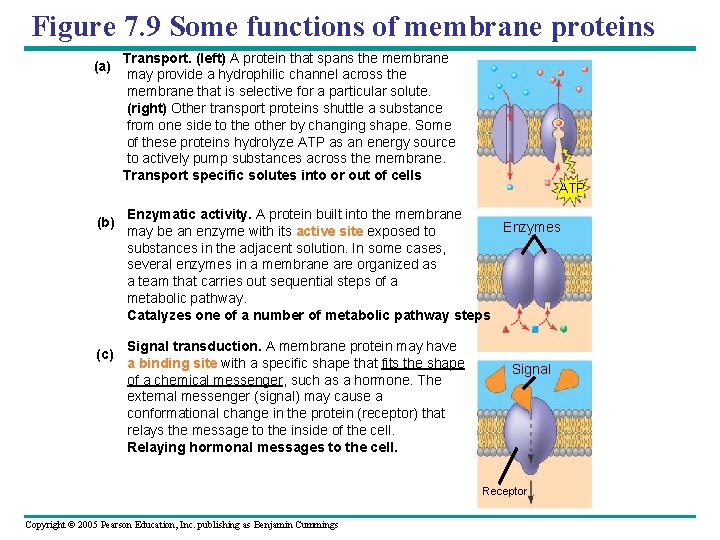
Figure 7. 9 Some functions of membrane proteins (a) (b) (c) Transport. (left) A protein that spans the membrane may provide a hydrophilic channel across the membrane that is selective for a particular solute. (right) Other transport proteins shuttle a substance from one side to the other by changing shape. Some of these proteins hydrolyze ATP as an energy source to actively pump substances across the membrane. Transport specific solutes into or out of cells ATP Enzymatic activity. A protein built into the membrane Enzymes may be an enzyme with its active site exposed to substances in the adjacent solution. In some cases, several enzymes in a membrane are organized as a team that carries out sequential steps of a metabolic pathway. Catalyzes one of a number of metabolic pathway steps Signal transduction. A membrane protein may have a binding site with a specific shape that fits the shape of a chemical messenger, such as a hormone. The external messenger (signal) may cause a conformational change in the protein (receptor) that relays the message to the inside of the cell. Relaying hormonal messages to the cell. Signal Receptor Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
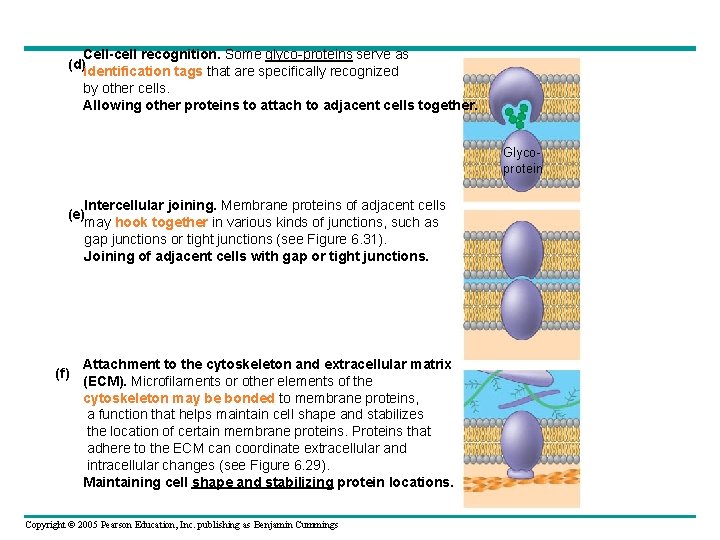
Cell-cell recognition. Some glyco-proteins serve as (d)identification tags that are specifically recognized by other cells. Allowing other proteins to attach to adjacent cells together. Glycoprotein Intercellular joining. Membrane proteins of adjacent cells (e) may hook together in various kinds of junctions, such as gap junctions or tight junctions (see Figure 6. 31). Joining of adjacent cells with gap or tight junctions. (f) Attachment to the cytoskeleton and extracellular matrix (ECM). Microfilaments or other elements of the cytoskeleton may be bonded to membrane proteins, a function that helps maintain cell shape and stabilizes the location of certain membrane proteins. Proteins that adhere to the ECM can coordinate extracellular and intracellular changes (see Figure 6. 29). Maintaining cell shape and stabilizing protein locations. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
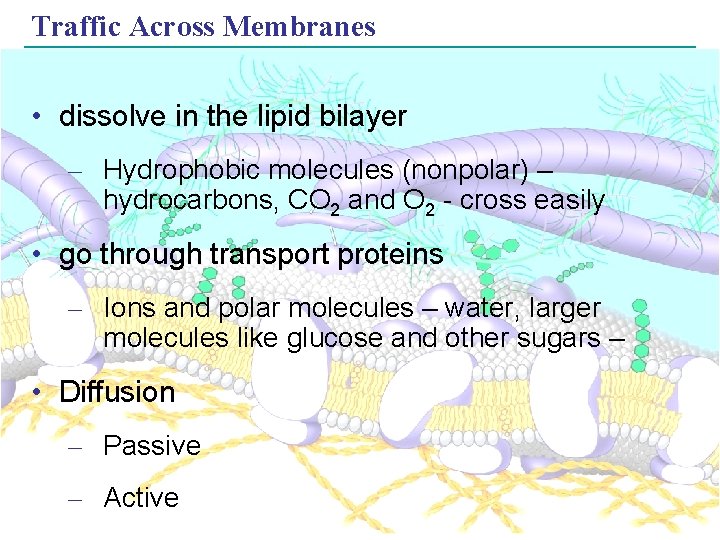
Traffic Across Membranes • dissolve in the lipid bilayer – Hydrophobic molecules (nonpolar) – hydrocarbons, CO 2 and O 2 - cross easily • go through transport proteins – Ions and polar molecules – water, larger molecules like glucose and other sugars – • Diffusion – Passive – Active Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
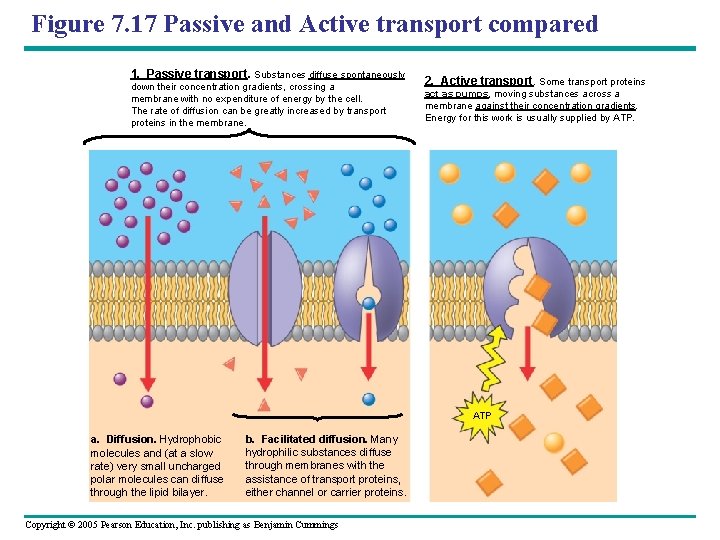
Figure 7. 17 Passive and Active transport compared 1. Passive transport. Substances diffuse spontaneously down their concentration gradients, crossing a membrane with no expenditure of energy by the cell. The rate of diffusion can be greatly increased by transport proteins in the membrane. 2. Active transport. Some transport proteins act as pumps, moving substances across a membrane against their concentration gradients. Energy for this work is usually supplied by ATP a. Diffusion. Hydrophobic molecules and (at a slow rate) very small uncharged polar molecules can diffuse through the lipid bilayer. b. Facilitated diffusion. Many hydrophilic substances diffuse through membranes with the assistance of transport proteins, either channel or carrier proteins. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
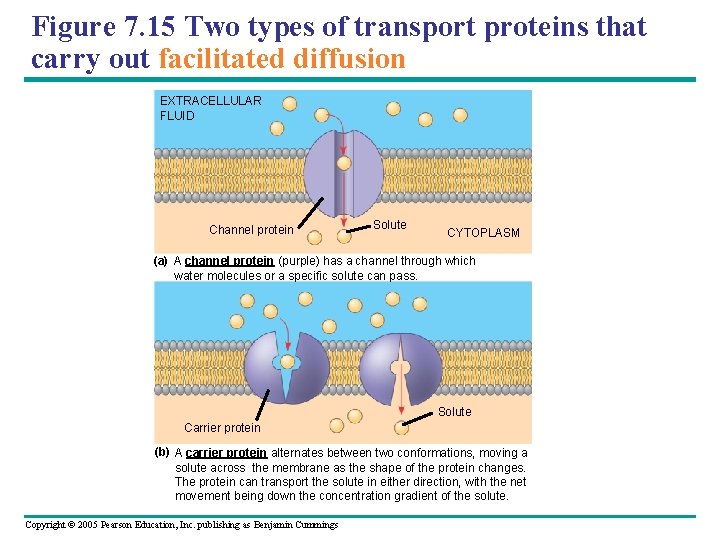
Figure 7. 15 Two types of transport proteins that carry out facilitated diffusion EXTRACELLULAR FLUID Channel protein Solute CYTOPLASM (a) A channel protein (purple) has a channel through which water molecules or a specific solute can pass. Solute Carrier protein (b) A carrier protein alternates between two conformations, moving a solute across the membrane as the shape of the protein changes. The protein can transport the solute in either direction, with the net movement being down the concentration gradient of the solute. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
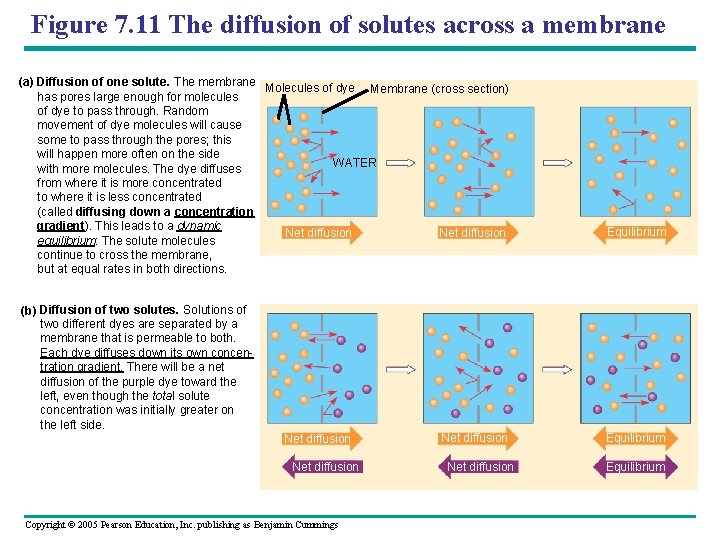
Figure 7. 11 The diffusion of solutes across a membrane (a) Diffusion of one solute. The membrane Molecules of dye Membrane (cross section) has pores large enough for molecules of dye to pass through. Random movement of dye molecules will cause some to pass through the pores; this will happen more often on the side WATER with more molecules. The dye diffuses from where it is more concentrated to where it is less concentrated (called diffusing down a concentration gradient). This leads to a dynamic Net diffusion equilibrium: The solute molecules continue to cross the membrane, but at equal rates in both directions. Equilibrium (b) Diffusion of two solutes. Solutions of two different dyes are separated by a membrane that is permeable to both. Each dye diffuses down its own concentration gradient. There will be a net diffusion of the purple dye toward the left, even though the total solute concentration was initially greater on the left side. Net Osmosis diffusion Net diffusion Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Net diffusion Equilibrium
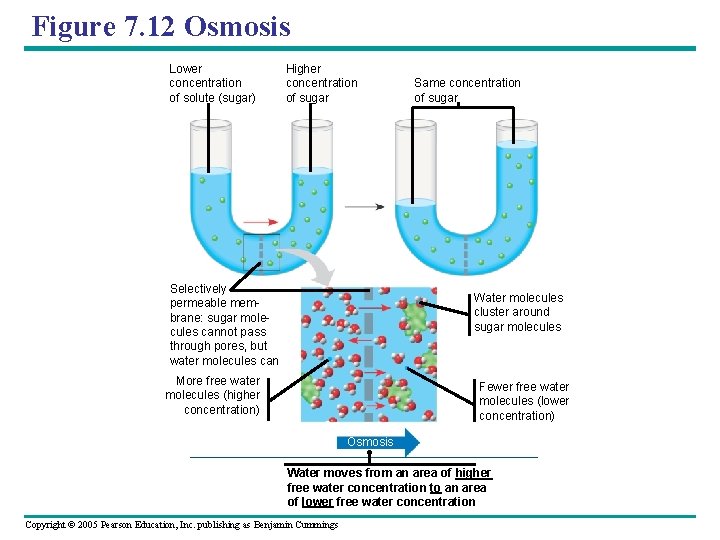
Figure 7. 12 Osmosis Lower concentration of solute (sugar) Higher concentration of sugar Same concentration of sugar Selectively permeable membrane: sugar molecules cannot pass through pores, but water molecules can Water molecules cluster around sugar molecules More free water molecules (higher concentration) Fewer free water molecules (lower concentration) Osmosis Water moves from an area of higher free water concentration to an area of lower free water concentration Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
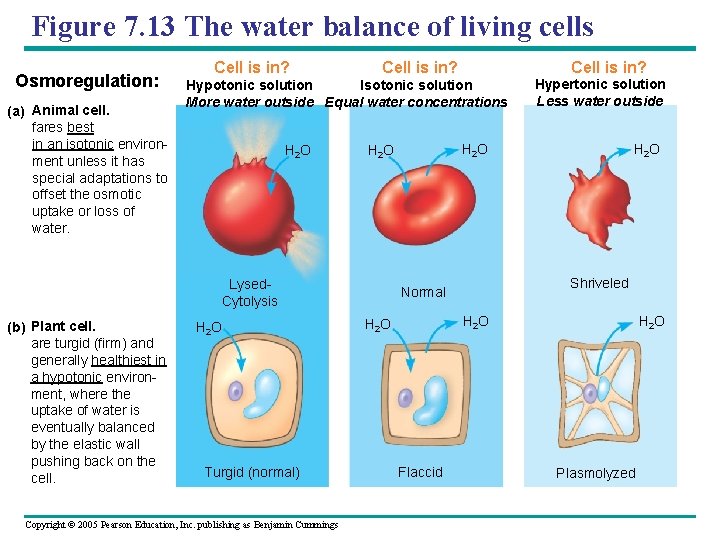
Figure 7. 13 The water balance of living cells Osmoregulation: (a) Animal cell. fares best in an isotonic environment unless it has special adaptations to offset the osmotic uptake or loss of water. Cell is in? Hypotonic solution Isotonic solution More water outside Equal water concentrations H 2 O Turgid (normal) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Hypertonic solution Less water outside H 2 O Lysed. Cytolysis (b) Plant cell. are turgid (firm) and generally healthiest in a hypotonic environment, where the uptake of water is eventually balanced by the elastic wall pushing back on the cell. Cell is in? H 2 O Shriveled Normal H 2 O Flaccid H 2 O Plasmolyzed
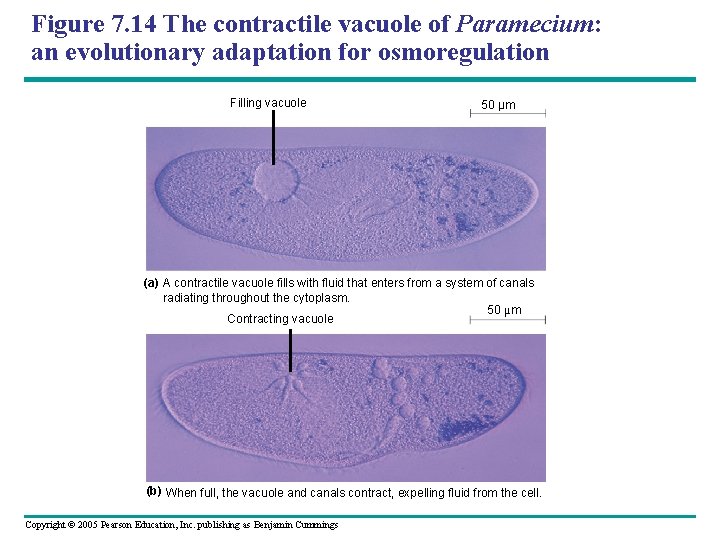
Figure 7. 14 The contractile vacuole of Paramecium: an evolutionary adaptation for osmoregulation Filling vacuole 50 µm (a) A contractile vacuole fills with fluid that enters from a system of canals radiating throughout the cytoplasm. 50 µm Contracting vacuole (b) When full, the vacuole and canals contract, expelling fluid from the cell. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Figure 7. 16 The sodium-potassium pump (protein): a specific case of active transport 1 Cytoplasmic Na+ binds to. EXTRACELLULAR+ [Na ] high the sodium-potassium FLUID [K+] low + Na pump. 2 Na+ binding stimulates phosphorylation by ATP. Na+ hydrolyze ATP as an energy source Na+ [Na+] low Na+ + CYTOPLASM [K ] high 3 K+ is released and Na+ sites are receptive again; The cycle repeats. P ADP Na+ ATP Phosphorylation causes protein to change its conformation, expelling Na+ to the outside. 4 the Na+ K+ P K+ 5 Loss of the phosphate restores the protein’s original conformation. 6 K+ K+ P Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Pi Extracellular K+ binds to the protein, triggering release of the Phosphate group. *Used to maintain these gradients – 3 NA+outside and 2 K+ inside
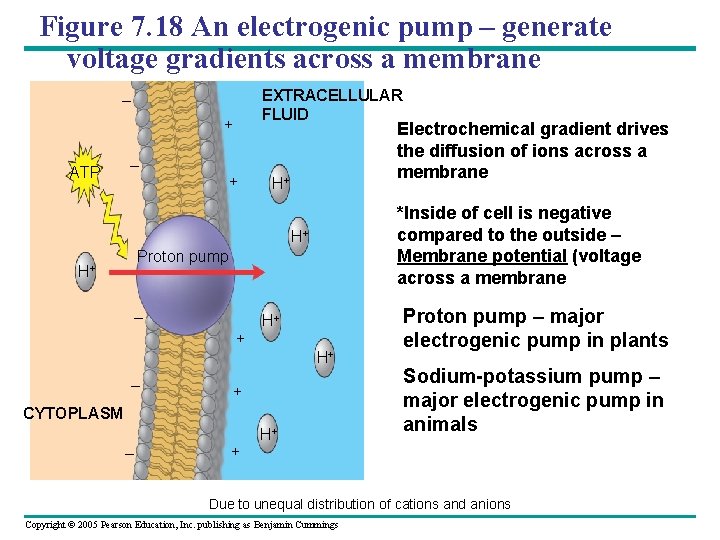
Figure 7. 18 An electrogenic pump – generate voltage gradients across a membrane EXTRACELLULAR FLUID – + ATP – + Electrochemical gradient drives the diffusion of ions across a membrane H+ *Inside of cell is negative compared to the outside – Membrane potential (voltage across a membrane H+ Proton pump H+ – + H+ H+ – + CYTOPLASM – + H+ Proton pump – major electrogenic pump in plants Sodium-potassium pump – major electrogenic pump in animals Due to unequal distribution of cations and anions Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
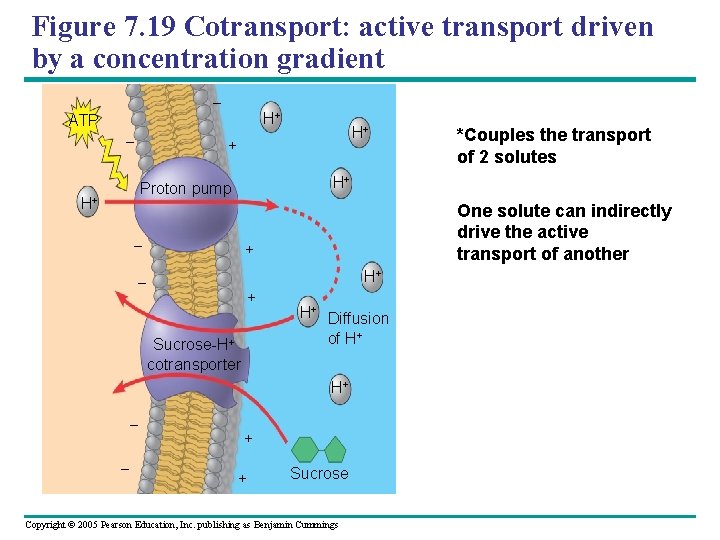
Figure 7. 19 Cotransport: active transport driven by a concentration gradient – ATP – + H+ H+ Proton pump H+ – One solute can indirectly drive the active transport of another + H+ – + Sucrose-H+ cotransporter H+ Diffusion of H+ H+ – – *Couples the transport of 2 solutes + + Sucrose Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
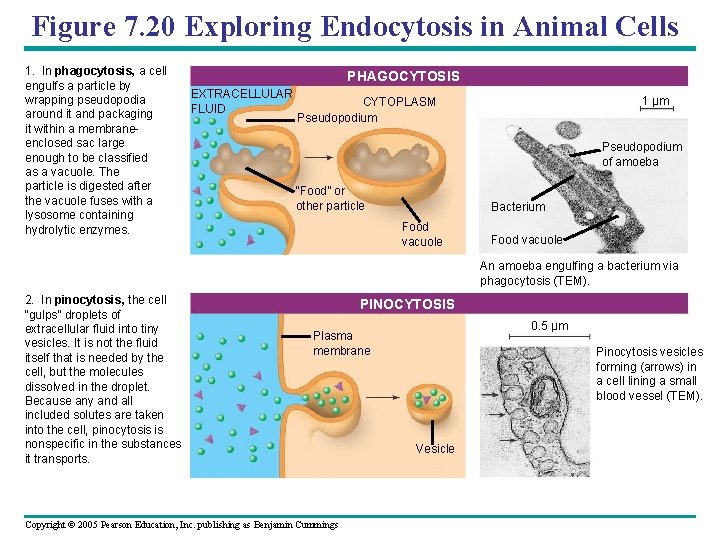
Figure 7. 20 Exploring Endocytosis in Animal Cells 1. In phagocytosis, a cell engulfs a particle by wrapping pseudopodia around it and packaging it within a membraneenclosed sac large enough to be classified as a vacuole. The particle is digested after the vacuole fuses with a lysosome containing hydrolytic enzymes. PHAGOCYTOSIS EXTRACELLULAR FLUID 1 µm CYTOPLASM Pseudopodium of amoeba “Food” or other particle Bacterium Food vacuole An amoeba engulfing a bacterium via phagocytosis (TEM). 2. In pinocytosis, the cell “gulps” droplets of extracellular fluid into tiny vesicles. It is not the fluid itself that is needed by the cell, but the molecules dissolved in the droplet. Because any and all included solutes are taken into the cell, pinocytosis is nonspecific in the substances it transports. PINOCYTOSIS 0. 5 µm Plasma membrane Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Pinocytosis vesicles forming (arrows) in a cell lining a small blood vessel (TEM). Vesicle
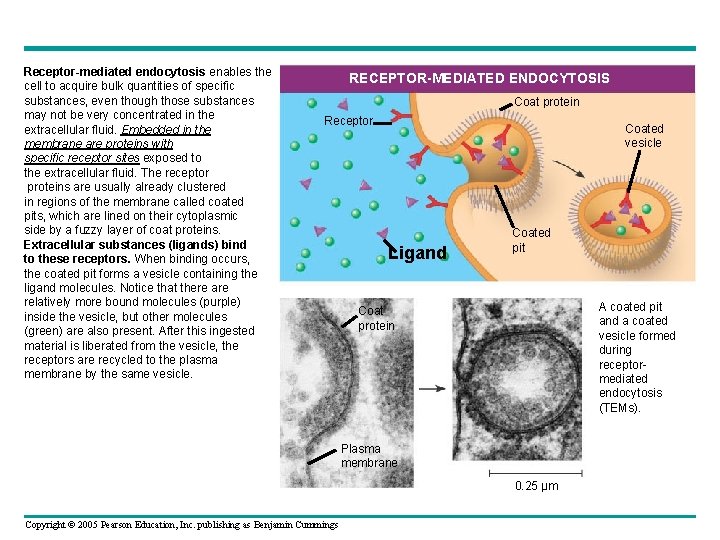
Receptor-mediated endocytosis enables the cell to acquire bulk quantities of specific substances, even though those substances may not be very concentrated in the extracellular fluid. Embedded in the membrane are proteins with specific receptor sites exposed to the extracellular fluid. The receptor proteins are usually already clustered in regions of the membrane called coated pits, which are lined on their cytoplasmic side by a fuzzy layer of coat proteins. Extracellular substances (ligands) bind to these receptors. When binding occurs, the coated pit forms a vesicle containing the ligand molecules. Notice that there are relatively more bound molecules (purple) inside the vesicle, but other molecules (green) are also present. After this ingested material is liberated from the vesicle, the receptors are recycled to the plasma membrane by the same vesicle. RECEPTOR-MEDIATED ENDOCYTOSIS Coat protein Receptor Coated vesicle Ligand Coated pit A coated pit and a coated vesicle formed during receptormediated endocytosis (TEMs). Coat protein Plasma membrane 0. 25 µm Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
- Slides: 23