CH 7 Intro to Coordination Compounds 1 Innersphere


![Nomenclature ex: [Cu(OH 2)6]2+ is hexaaquacopper(II) or hexaaquacopper(2+) [Cu. Cl 4] is tetrachlorocuprate(III) or Nomenclature ex: [Cu(OH 2)6]2+ is hexaaquacopper(II) or hexaaquacopper(2+) [Cu. Cl 4] is tetrachlorocuprate(III) or](https://slidetodoc.com/presentation_image_h/95df142226c050aff7ee474a2359a00e/image-4.jpg)
![Nomenclature 6. To write the formula: [metal, then anionic ligands, then neutral ligands] net Nomenclature 6. To write the formula: [metal, then anionic ligands, then neutral ligands] net](https://slidetodoc.com/presentation_image_h/95df142226c050aff7ee474a2359a00e/image-6.jpg)


![Cluster compounds direct M-M bonding ex: [Re 2 Cl 8]2 octachlorodirhenate(III) D 4 h Cluster compounds direct M-M bonding ex: [Re 2 Cl 8]2 octachlorodirhenate(III) D 4 h](https://slidetodoc.com/presentation_image_h/95df142226c050aff7ee474a2359a00e/image-12.jpg)






- Slides: 30

CH 7. Intro to Coordination Compounds 1

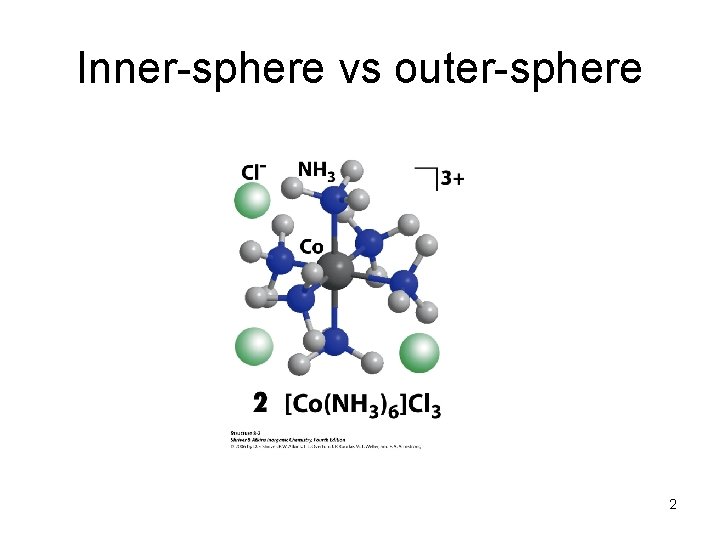
Inner-sphere vs outer-sphere 2

Nomenclature 1. Learn common ligand names (Table 7. 1) Ex: : OH 2 : O 2 : CN : Br : NH 3 aqua oxo (oxido) cyano (cyanido) bromo (bromido) ammine Note that anionic ligands end in “o” 2. List ligands in alphabetical order 3. Metal name at end, add “ate” if it’s an anionic complex some common names – ferrate, stannate, plumbate, cuprate 4. Add (and metal oxidation number in Roman numerals) or add metal (and total complex charge in Arabic numerals) 3
![Nomenclature ex CuOH 262 is hexaaquacopperII or hexaaquacopper2 Cu Cl 4 is tetrachlorocuprateIII or Nomenclature ex: [Cu(OH 2)6]2+ is hexaaquacopper(II) or hexaaquacopper(2+) [Cu. Cl 4] is tetrachlorocuprate(III) or](https://slidetodoc.com/presentation_image_h/95df142226c050aff7ee474a2359a00e/image-4.jpg)
Nomenclature ex: [Cu(OH 2)6]2+ is hexaaquacopper(II) or hexaaquacopper(2+) [Cu. Cl 4] is tetrachlorocuprate(III) or tetrachloridocuprate(III) 5. Add prefixes to indicate number of each ligand type mono, di, tri, tetra, penta, hexa or use bis, tris, tetrakis if less confusing due to ligand name ex: [Pt. Br 2{P(CH 3)3}2 ] is dibromobis(trimethylphosphine)platinum(II) ~ C 2 v ~D 2 h Stereoisomers cis- and transplatin. The cis isomer is an anticancer drug. 4

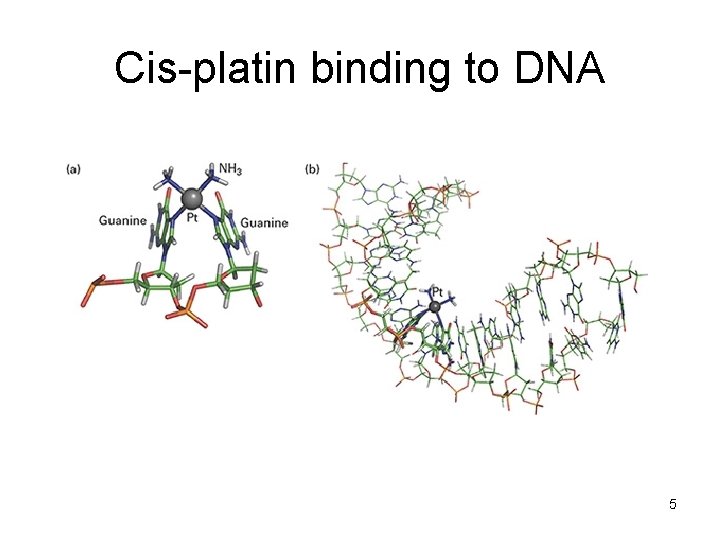
Cis-platin binding to DNA 5
![Nomenclature 6 To write the formula metal then anionic ligands then neutral ligands net Nomenclature 6. To write the formula: [metal, then anionic ligands, then neutral ligands] net](https://slidetodoc.com/presentation_image_h/95df142226c050aff7ee474a2359a00e/image-6.jpg)
Nomenclature 6. To write the formula: [metal, then anionic ligands, then neutral ligands] net charge superscript 7. Special ligands: a. ambidentate -SCN (thicyanato) vs NCS (isothiocyanato) [Pt(SCN)4]2 D 4 h [Cr(NCS)(NH 3)5]2+ NO 2 (nitrito) vs tetrathiocyanatoplatinate(II) pentaammineisothiocyanatochromium(III) ONO (isonitrito) 6

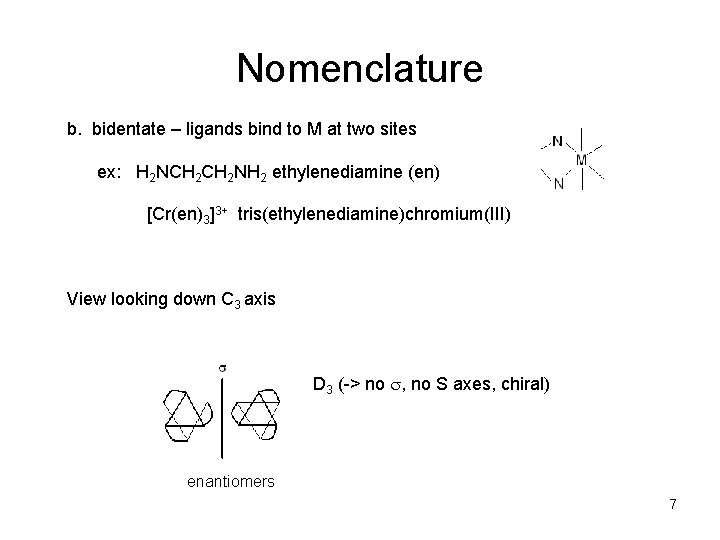
Nomenclature b. bidentate – ligands bind to M at two sites ex: H 2 NCH 2 NH 2 ethylenediamine (en) [Cr(en)3]3+ tris(ethylenediamine)chromium(III) View looking down C 3 axis D 3 (-> no , no S axes, chiral) enantiomers 7

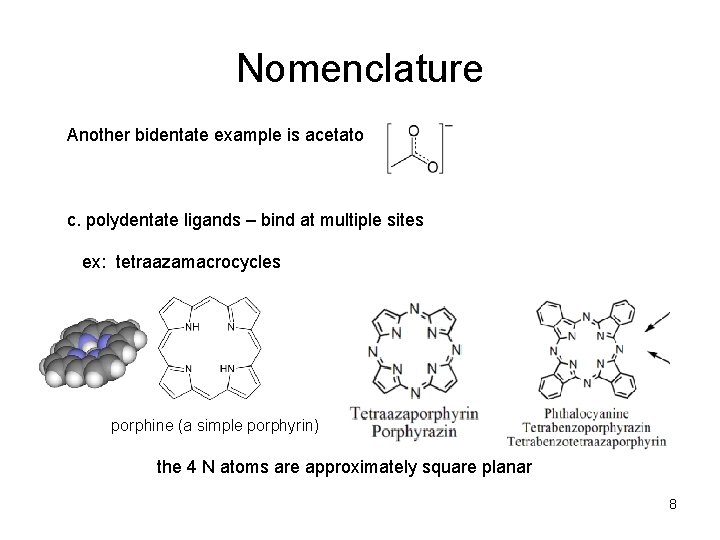
Nomenclature Another bidentate example is acetato c. polydentate ligands – bind at multiple sites ex: tetraazamacrocycles porphine (a simple porphyrin) the 4 N atoms are approximately square planar 8

Geometric Isomers There have distinct physical and chemical properties Oh coordination MX 5 Y 1 isomer MX 4 Y 2 2 isomers (cis or trans) MX 3 Y 3 2 isomers (fac = C 3 V or mer = C 2 V ) ex: [Co. Cl 2(NH 3)4]+ tetraamminedichlorocobalt(III) cis – purple trans – green 9

Optical Isomers Enantiomers = non-superimposable mirror images of a chiral molecule enantiomers have identical physical properties (except in a chiral environment, for example retention times on a chiral column are not the same) enantiomers rotate the plane of polarized light in opposite directions (optical isomers) 10

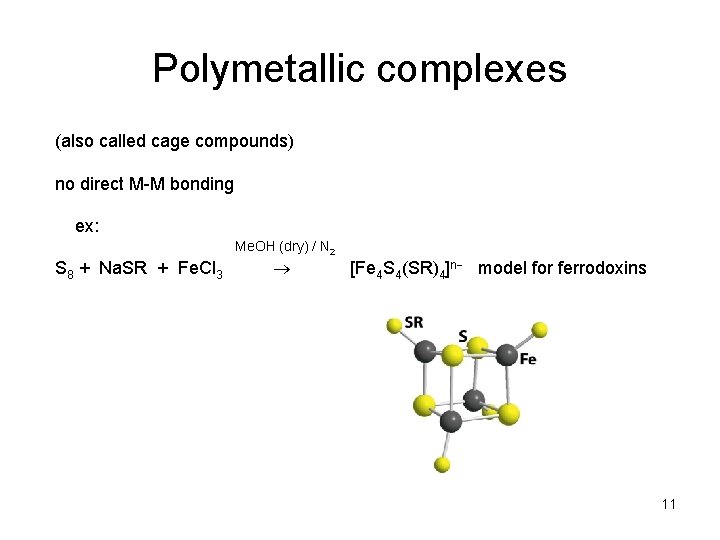
Polymetallic complexes (also called cage compounds) no direct M-M bonding ex: Me. OH (dry) / N 2 S 8 + Na. SR + Fe. Cl 3 [Fe 4 S 4(SR)4]n model for ferrodoxins 11
![Cluster compounds direct MM bonding ex Re 2 Cl 82 octachlorodirhenateIII D 4 h Cluster compounds direct M-M bonding ex: [Re 2 Cl 8]2 octachlorodirhenate(III) D 4 h](https://slidetodoc.com/presentation_image_h/95df142226c050aff7ee474a2359a00e/image-12.jpg)
Cluster compounds direct M-M bonding ex: [Re 2 Cl 8]2 octachlorodirhenate(III) D 4 h (eclipsed) 12

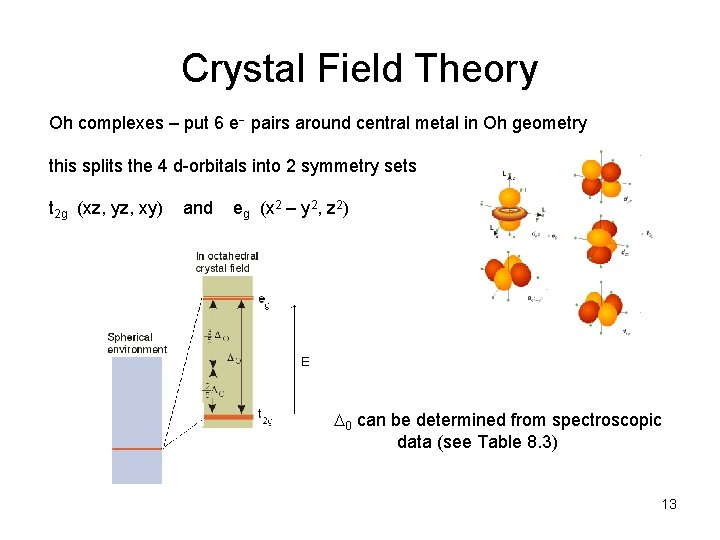
Crystal Field Theory Oh complexes – put 6 e pairs around central metal in Oh geometry this splits the 4 d-orbitals into 2 symmetry sets t 2 g (xz, yz, xy) and eg (x 2 – y 2, z 2) 0 can be determined from spectroscopic data (see Table 8. 3) 13

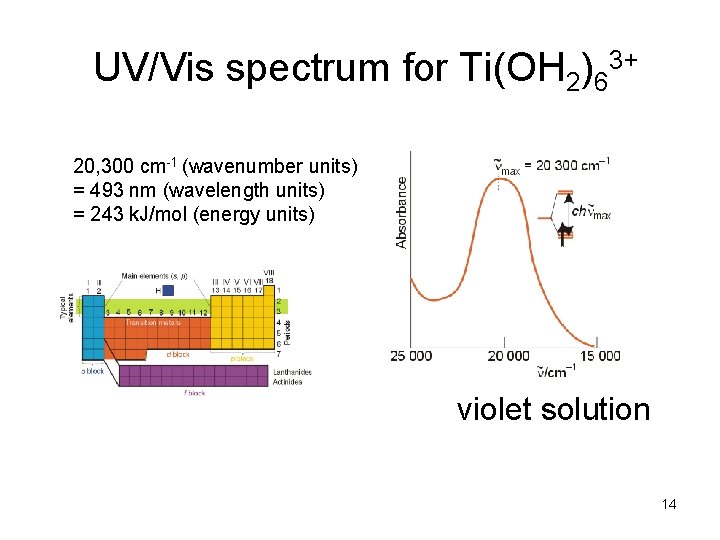
UV/Vis spectrum for Ti(OH 2)63+ 20, 300 cm-1 (wavenumber units) = 493 nm (wavelength units) = 243 k. J/mol (energy units) violet solution 14

Crystal Field Theory 0 depends on: 1. ligand (spectrochemical series) 0 I < Br < Cl < F < OH < NH 3 < CN < CO weak field strong field more complete list in text 2. metal ion 0 greater for higher oxidation number – stronger, shorter M-L interaction 0 greater going down a group – more diffuse d-orbitals interact more strongly with ligands 0 Mn 2+ < Fe 3+ < Ru 3+ < Pd 4+ < Pt 4+ small large 15

Ligand Field Stabilization Energy the LFSE = (0. 4 x 0. 6 y) 0 for electronic config t 2 gx egy high spin case # d electrons 0 1 2 3 4 e config - t 2 g 1 t 2 g 2 t 2 g 3 LFSE ( 0) 0 0. 4 0. 8 # unpaired e 0 1 2 5 6 7 8 9 10 t 2 g 3 eg 1 t 2 g 3 eg 2 t 4 eg 2 t 5 eg 2 t 2 g 6 eg 3 t 2 g 6 eg 4 1. 2 0. 6 0 0. 4 0. 8 1. 2 0. 6 0 3 4 5 4 3 2 1 0 depends of relative values of 0 and pairing energy. 16

High spin vs low spin d 4 t 2 g 3 eg 1 t 2 g 4 LSFE = 0. 6 0 LFSE = 1. 6 0 PE high spin low spin (weak field) (strong field) [Cr(OH 2)6]2+ [Cr(CN)6]4 17

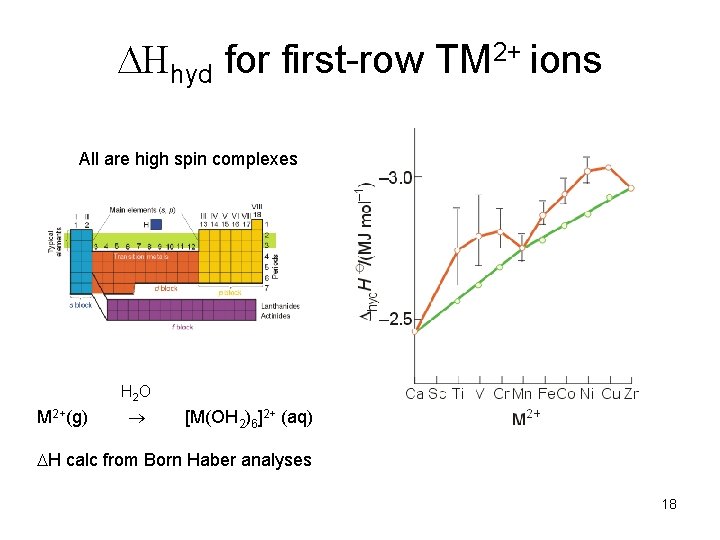
Hhyd for first-row TM 2+ ions All are high spin complexes H 2 O M 2+(g) [M(OH 2)6]2+ (aq) H calc from Born Haber analyses 18

Magnetic Measurements Magnetic moment ( ) is the attractive force towards a magnetic field (H) ≈ [N(N + 2)]1/2 B where N = number of unpaired electrons N / B 1 2 3 4 5 1. 73 2. 83 3. 87 4. 90 5. 92 this is the paramagnetic contribution from unpaired e spin only, it ignores both spinorbit coupling and diamagnetic contributions ex: [Mn(NCS)6]4 experimental / B = 6. 06, Mn(II) is d 5 it must be a high spin complex 19

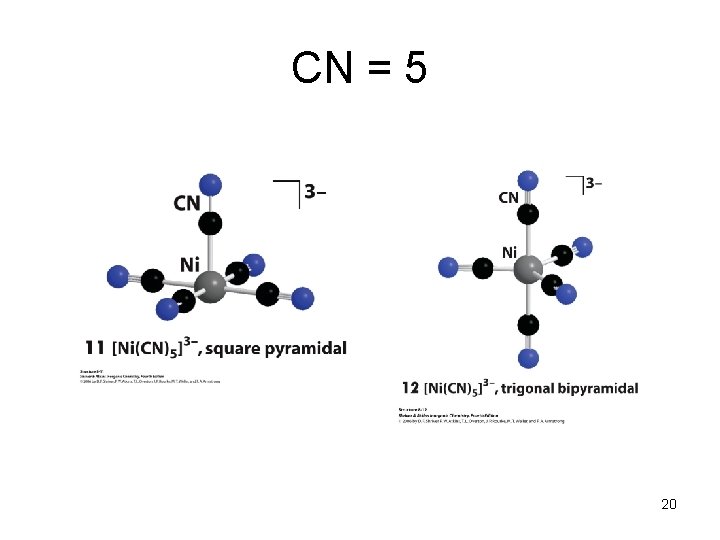
CN = 5 20

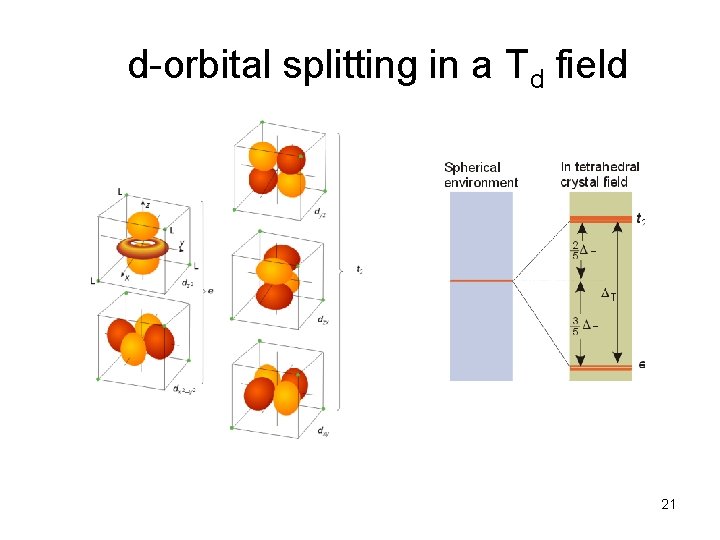
d-orbital splitting in a Td field 21

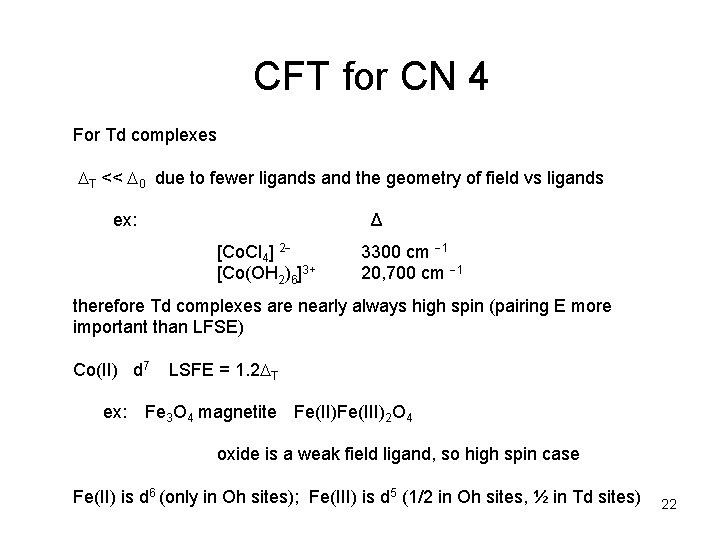
CFT for CN 4 For Td complexes T << 0 due to fewer ligands and the geometry of field vs ligands ex: Δ [Co. Cl 4] 2 [Co(OH 2)6]3+ 3300 cm 1 20, 700 cm 1 therefore Td complexes are nearly always high spin (pairing E more important than LFSE) Co(II) d 7 ex: LSFE = 1. 2 T Fe 3 O 4 magnetite Fe(II)Fe(III)2 O 4 oxide is a weak field ligand, so high spin case Fe(II) is d 6 (only in Oh sites); Fe(III) is d 5 (1/2 in Oh sites, ½ in Td sites) 22

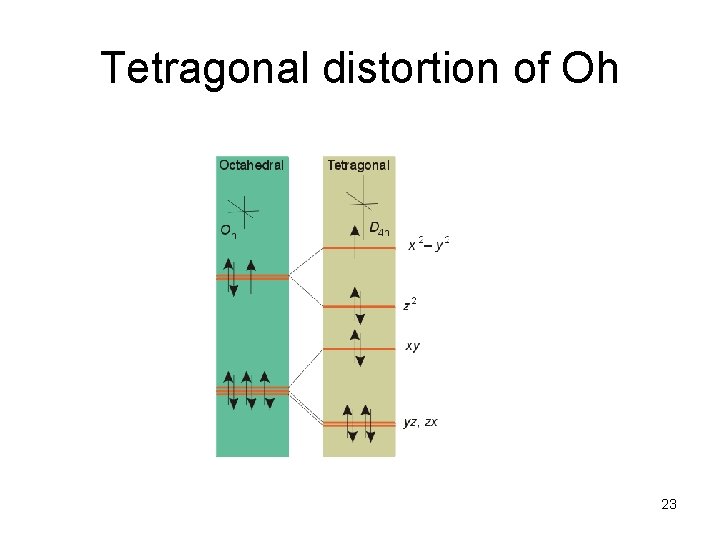
Tetragonal distortion of Oh 23

Square planar complexes D 4 h is a common structure for d 8 complexes (full z 2, empty x 2 – y 2 orbitals) Group 9: Rh(I), Ir(I) Group 10: Pt(II), Pd(II) Group 11: Au(III), for example Au. Cl 4 Note: [Ni(CN)4]2 is D 4 h but [Ni. Cl 4]2 is Td Ni(II) has a smaller than Pd, PT so Td is common but we see D 4 h with strong field ligands 24

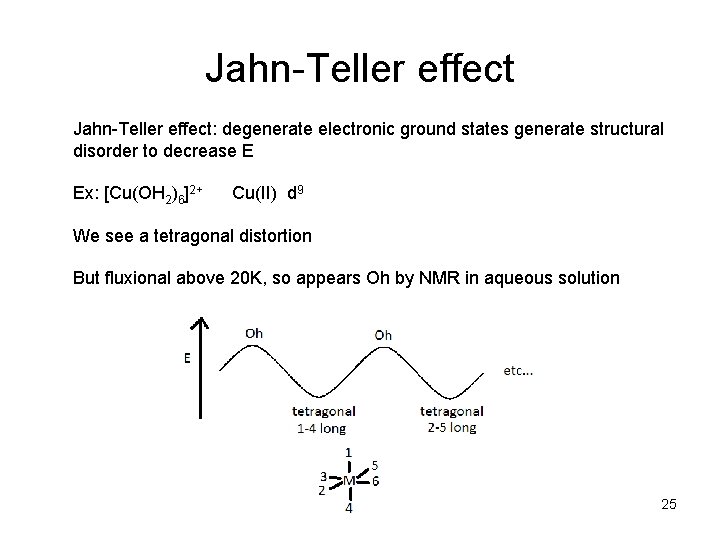
Jahn-Teller effect: degenerate electronic ground states generate structural disorder to decrease E Ex: [Cu(OH 2)6]2+ Cu(II) d 9 We see a tetragonal distortion But fluxional above 20 K, so appears Oh by NMR in aqueous solution 25

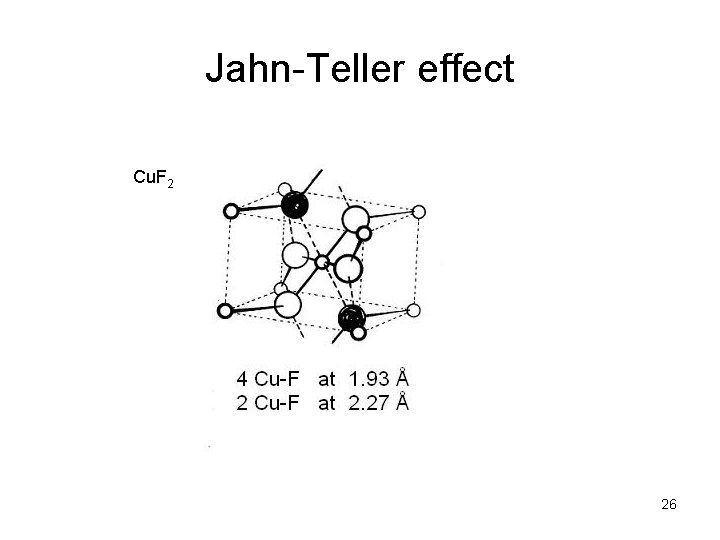
Jahn-Teller effect Cu. F 2 26

Ligand Field Theory CFT does not explain ligand field strengths; MO theory can Start with SALCs that are ligand combinations shown to the right 27

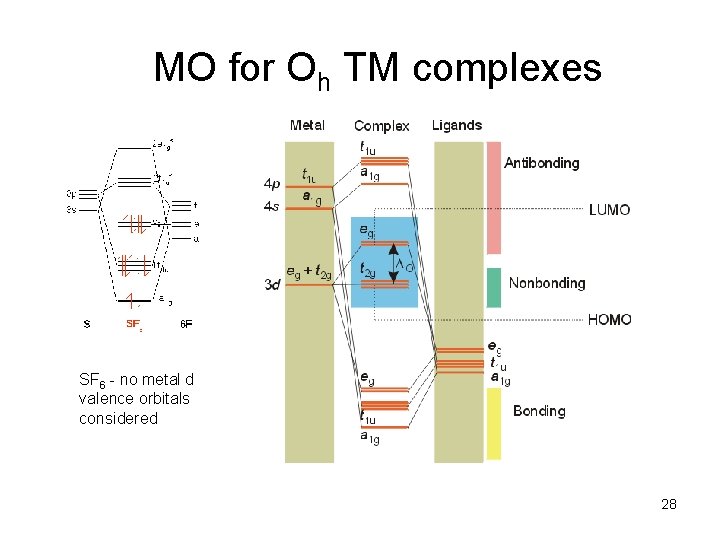
MO for Oh TM complexes SF 6 - no metal d valence orbitals considered 28

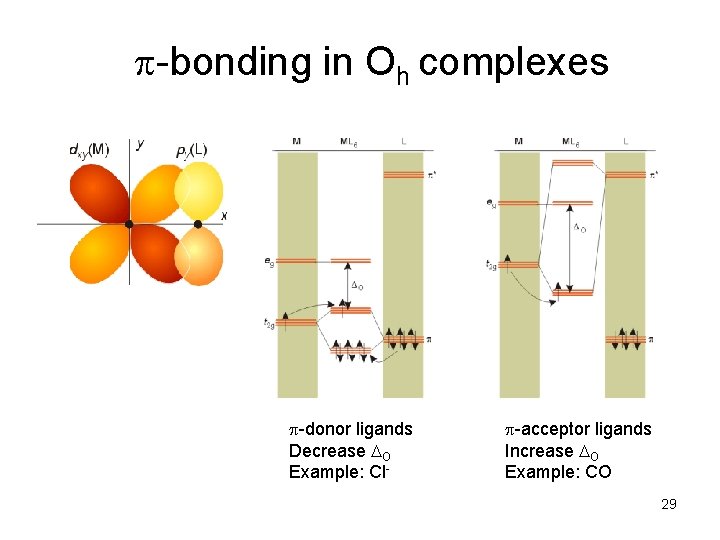
p-bonding in Oh complexes p-donor ligands Decrease O Example: Cl- p-acceptor ligands Increase O Example: CO 29

Oh character table 30