CH 6 Thermochemistry Renee Y Becker Valencia Community

CH 6: Thermochemistry Renee Y. Becker Valencia Community College CHM 1045 1

Energy • Energy: is the capacity to do work, or supply heat. Energy = Work + Heat • Kinetic Energy: is the energy of motion. EK = 1/2 mv 2 (1 Joule = 1 kg m 2/s 2) (1 calorie = 4. 184 J) • Potential Energy: is stored energy. 2
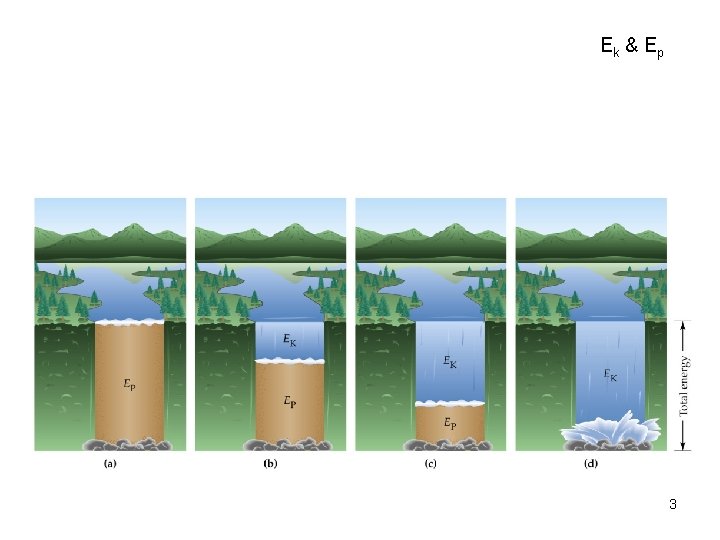
Ek & E p 3

Example 1: KE Which of the following has the greatest kinetic energy? 1. A 12 kg toy car moving at 5 mph? 2. A 12 kg toy car standing at the top of a large hill? 4

Energy • Thermal Energy is the kinetic energy of molecular motion • Thermal energy is proportional to the temperature in degrees Kelvin. Ethermal T(K) • Heat is the amount of thermal energy transferred between two objects at different temperatures. 5

• In an experiment: Reactants and products are the system; everything else is the surroundings. • Energy flow from the system to the surroundings has a negative sign (loss of energy). (- E or - H) • Energy flow from the surroundings to the system has a positive sign (gain of energy). (+ E or + H) 6
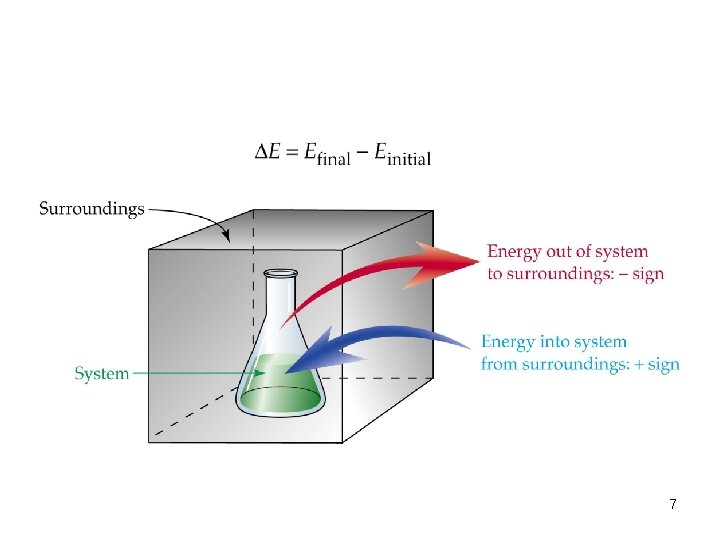
7

• The law of the conservation of energy: Energy cannot be created or destroyed. • The energy of an isolated system must be constant. • The energy change in a system equals the work done on the system + the heat added. E = Efinal – Einitial = E 2 – E 1 = q + w q = heat, w = work 8

• Pressure is the force per unit area. (1 N/m 2 = 1 Pa) (1 atm = 101, 325 Pa) • Work is a force (F) that produces an object’s movement, times the distance moved (d): Work = Force x Distance 9
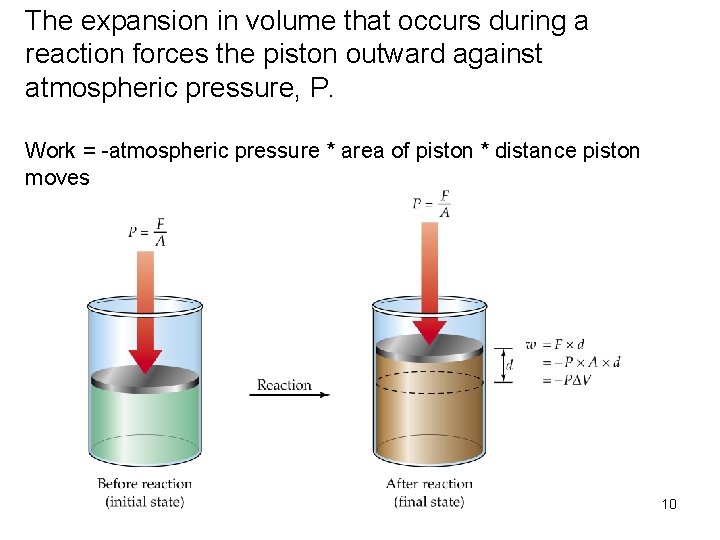
The expansion in volume that occurs during a reaction forces the piston outward against atmospheric pressure, P. Work = -atmospheric pressure * area of piston * distance piston moves 10
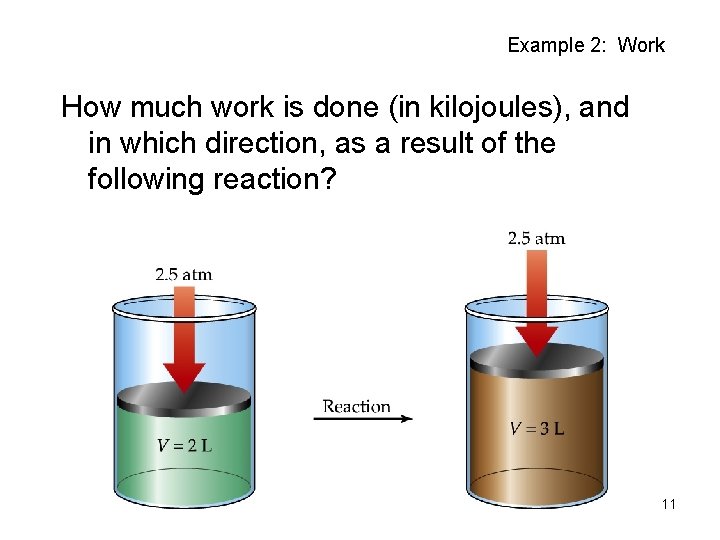
Example 2: Work How much work is done (in kilojoules), and in which direction, as a result of the following reaction? 11

• The amount of heat exchanged between the system and the surroundings is given the symbol q. q = E + P V At constant volume ( V = 0): qv = E At constant pressure: qp = E + P V = H Enthalpy change: H = Hproducts – Hreactants 12

Example 3: Work The explosion of 2. 00 mol of solid TNT with a volume of approximately 0. 274 L produces gases with a volume of 489 L at room temperature. How much PV (in kilojoules) work is done during the explosion? Assume P = 1 atm, T = 25°C. 2 C 7 H 5 N 3 O 6(s) 12 CO(g) + 5 H 2(g) + 3 N 2(g) + 2 C(s) 13

• Enthalpies of Physical Change: Enthalpy is a state function, the enthalpy change from solid to vapor does not depend on the path taken between the two states. Hsubl = Hfusion + Hvap 14

• Enthalpies of Chemical Change: Often called heats of reaction ( Hreaction). Endothermic: Heat flows into the system from the surroundings and H has a positive sign. Exothermic: Heat flows out of the system into the surroundings and H has a negative sign. 15

16

• Reversing a reaction changes the sign of H for a reaction. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) H = – 2219 k. J 3 CO 2(g) + 4 H 2 O(l) C 3 H 8(g) + 5 O 2(g) H = +2219 k. J • Multiplying a reaction increases H by the same factor. 3 [C 3 H 8(g) + 15 O 2(g) 9 CO 2(g) + 12 H 2 O(l)] H = 3(-2219) k. J H = -6657 k. J 17

Example 4: Heat • How much heat (in kilojoules) is evolved or absorbed in each of the following reactions? a) Burning of 15. 5 g of propane: C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) H = – 2219 k. J/mole b) Reaction of 4. 88 g of barium hydroxide octahydrate with ammonium chloride: Ba(OH)2· 8 H 2 O(s) + 2 NH 4 Cl(s) Ba. Cl 2(aq) + 2 NH 3(aq) + 10 H 2 O(l) H = +80. 3 k. J/mole 18

• Thermodynamic Standard State: Most stable form of a substance at 1 atm pressure and 25°C; 1 M concentration for all substances in solution. • These are indicated by a superscript ° to the symbol of the quantity reported. • Standard enthalpy change is indicated by the symbol H°. 19

Example 5: Is an endothermic reaction a favorable process thermodynamically speaking? 1) Yes 2) No 20

Hess’s Law • Hess’s Law: The overall enthalpy change for a reaction is equal to the sum of the enthalpy changes for the individual steps in the reaction. (not a physical change, chemical change) 3 H 2(g) + N 2(g) 2 NH 3(g) H° = – 92. 2 k. J 21

• Reactants and products in individual steps can be added and subtracted to determine the overall equation. (1) 2 H 2(g) + N 2(g) N 2 H 4(g) H° 1 = ? (2) N 2 H 4(g) + H 2(g) 2 NH 3(g) H° 2 = – 187. 6 k. J (3) 3 H 2(g) + N 2(g) 2 NH 3(g) H° 3 = – 92. 2 k. J H° 1 + H° 2 = H°reaction Then H° 1 = H°reaction - H° 2 H° 1 = H° 3 – H° 2 = (– 92. 2 k. J) – (– 187. 6 k. J) = +95. 4 k. J 22

Example 6: Hess’s Law • The industrial degreasing solvent methylene chloride (CH 2 Cl 2, dichloromethane) is prepared from methane by reaction with chlorine: CH 4(g) + 2 Cl 2(g) CH 2 Cl 2(g) + 2 HCl(g) Use the following data to calculate H° (in kilojoules) for the above reaction: CH 4(g) + Cl 2(g) CH 3 Cl(g) + HCl(g) H° = – 98. 3 k. J CH 3 Cl(g) + Cl 2(g) H° = – 104 k. J CH 2 Cl 2(g) + HCl(g) 23

• Standard Heats of Formation ( H°f): The enthalpy change for the formation of 1 mole of substance in its standard state from its constituent elements in their standard states. • The standard heat of formation for any element in its standard state is defined as being ZERO. H°f = 0 for an element in its standard state 24

Standard Heats of Formation • Calculating H° for a reaction: H° = H°f (Products) – H°f (Reactants) • For a balanced equation, each heat of formation must be multiplied by the stoichiometric coefficient. a. A + b. B c. C + d. D H° = [c H°f (C) + d H°f (D)] – [a H°f (A) + b H°f (B)] 25
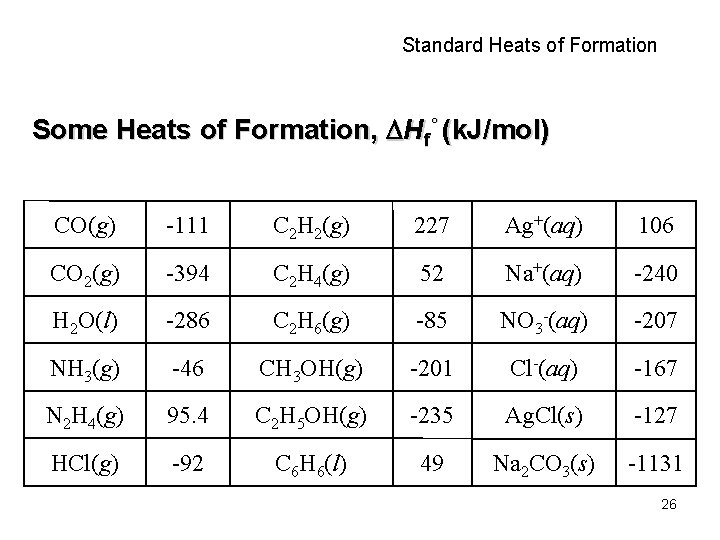
Standard Heats of Formation Some Heats of Formation, Hf° (k. J/mol) CO(g) -111 C 2 H 2(g) 227 Ag+(aq) 106 CO 2(g) -394 C 2 H 4(g) 52 Na+(aq) -240 H 2 O(l) -286 C 2 H 6(g) -85 NO 3 -(aq) -207 NH 3(g) -46 CH 3 OH(g) -201 Cl-(aq) -167 N 2 H 4(g) 95. 4 C 2 H 5 OH(g) -235 Ag. Cl(s) -127 HCl(g) -92 C 6 H 6(l) 49 Na 2 CO 3(s) -1131 26

Example 7: Standard heat of formation Calculate H° (in kilojoules) for the reaction of ammonia with O 2 to yield nitric oxide (NO) and H 2 O(g), a step in the Ostwald process for the commercial production of nitric acid. 27

Example 8: Standard heat of formation Calculate H° (in kilojoules) for the photosynthesis of glucose and O 2 from CO 2 and liquid water, a reaction carried out by all green plants. 28

Example 9: Which of the following would indicate an endothermic reaction? Why? 1. - H 2. + H 29

Heat of Phase Transitions from H f Calculate the heat of vaporization, H vap of water, using standard enthalpies of formation H 2 O(g) H 2 O(l) H f -241. 8 k. J/mol -285. 8 k. J/mol 30

Calorimetry and Heat Capacity • Calorimetry is the science of measuring heat changes (q) for chemical reactions. There are two types of calorimeters: • Bomb Calorimetry: A bomb calorimeter measures the heat change at constant volume such that q = E. • Constant Pressure Calorimetry: A constant pressure calorimeter measures the heat change at constant pressure such that q = H. 31
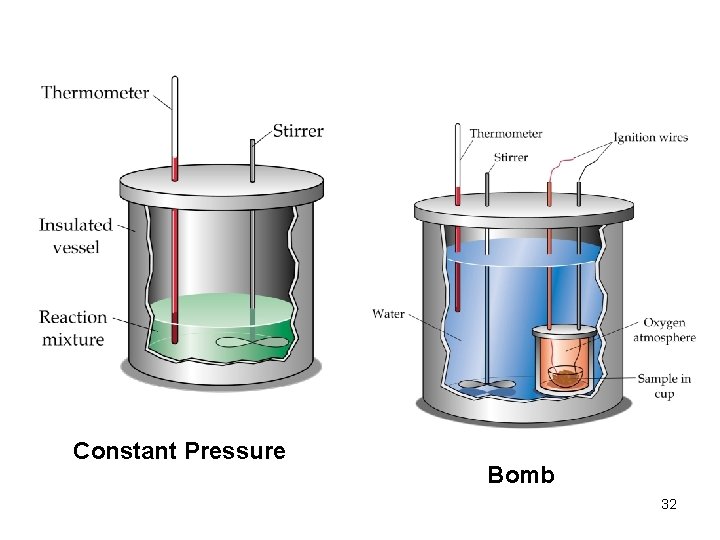
Constant Pressure Bomb 32

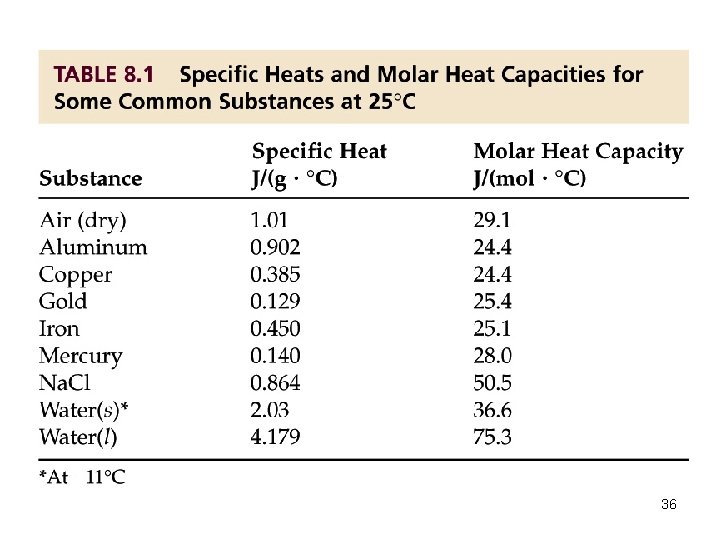
Calorimetry and Heat Capacity • Heat capacity (C) is the amount of heat required to raise the temperature of an object or substance a given amount. Specific Heat: The amount of heat required to raise the temperature of 1. 00 g of substance by 1. 00°C. q = s x m x t q = heat required (energy) s = specific heat m = mass in grams t = Tf - Ti 33

Calorimetry and Heat Capacity • Molar Heat: The amount of heat required to raise the temperature of 1. 00 mole of substance by 1. 00°C. q = MH x n x t q = heat required (energy) MH = molar heat n = moles t = Tf - Ti 34

Example 10: Specific Heat What is the specific heat of lead if it takes 96 J to raise the temperature of a 75 g block by 10. 0°C? 35
36

Example 11: Specific Heat How much energy (in J) does it take to increase the temperature of 12. 8 g of Gold from 56 C to 85 C? 37

Example 12: Molar Heat • How much energy (in J) does it take to increase the temperature of 1. 45 x 104 moles of water from 69 C to 94 C? 38
- Slides: 38