Ch 6 The Periodic Table and Periodic Law
Ch 6: The Periodic Table and Periodic Law Honors Chemistry

Section 6. 1 Development of the Modern Periodic Table Chemist’s first began to organize known elements in the 1700’s. Many have made contributions over the years. • Dmitri Mendeleev, a Russian chemist developed the first Periodic Table of elements in 1869. • There were about 60 known Elements at this time.

“I began to look about and write down the elements with their atomic weights and typical properties, analogous elements and like atomic weights on separate cards, and this soon convinced me that the properties of elements are in periodic dependence upon their atomic weights. ” --Mendeleev, Principles of Chemistry, 1905, Vol. II

Mendeleev’s Predictions • When creating his periodic table, Mendeleev left blank spaces for elements that had not yet been discovered. • Using periodic properties of the other elements, he correctly predicted the properties of scandium, gallium, and germanium.

Mendeleev’s Predictions… • Mendeleev predicted 5 elements. • Within 15 yrs, 3 of the 5 elements had been discovered!!!

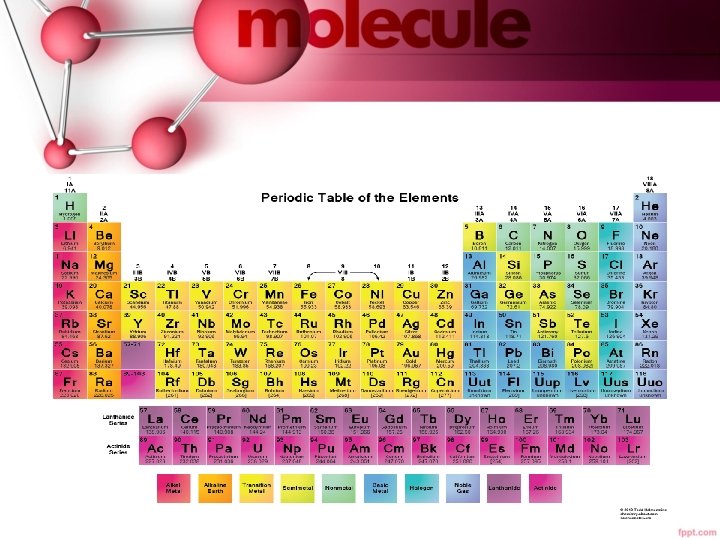
Our Modern Periodic Table… • Moseley rearranged the table by increasing atomic number, and resulted in a clear periodic pattern – our modern periodic table. • Periodic repetition of chemical and physical properties of the elements when they are arranged by increasing atomic number is called periodic law. • Interactive Periodic Table

• Elements in groups 1, 2, and 13 -18 possess a wide variety of chemical and physical properties and are called the representative elements. • Elements in groups 3 -12 are known as the transition metals. • Elements are classified as metals, non-metals, and metalloids.

Metals… • Metals are elements that are generally shiny when smooth and clean, solid at room temperature, good conductors of heat and electricity, malleable, and ductile. • Alkali metals are all the elements in group 1 except hydrogen, and are very reactive. • Alkaline earth metals are in group 2, and are also highly reactive.

Metals… • The transition elements are divided into transition metals and inner transition metals. • The two sets of inner transition metals are called the lanthanide series and actinide series and are located at the bottom of the periodic table.

Nonmetals… • Non-metals are elements that are generally gases or brittle, dull-looking solids, and poor conductors of heat and electricity. • Group 17 is composed of highly reactive elements called halogens. • Group 18 gases are extremely unreactive and commonly called noble gases.

Metalloids… • Metalloids have physical and chemical properties of both metals and nonmetals, such as silicon and germanium. • The metalloids form a staircase that separates the metals from the nonmetals, with the exceptions of aluminum and polonium which are metals.

Section 6. 2 Classification of the Elements • Recall electrons in the highest principal energy level are called valence electrons. • Groups can help determine how many valence electrons an atom has.

Remember… • Group 1 elements have 1 valence e- and group 2 elements have 2 valence e-. • The number of valence electrons for elements in groups 13 -18 is ten less than their group number. • Remember, the maximum number of valence e- is 8. • The Noble Gases are the only elements that have completely filled orbitals. He has 2 valence e- and the other noble gases all have 8 valence e-.
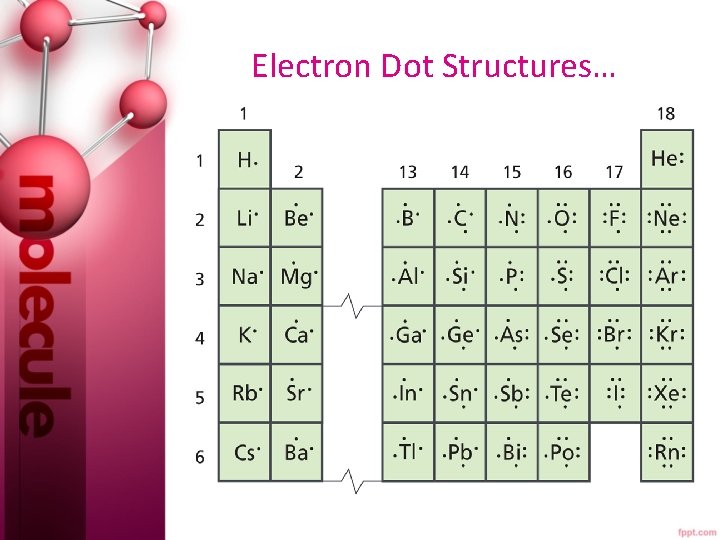
Electron Dot Structures…

Section 6. 3 Periodic Trends Atomic Radius… • Atomic size is a periodic trend influenced by electron configuration. • For metals, atomic radius is half the distance between adjacent nuclei in a crystal of the element or between the nuclei of identical elements in a molecule.

Atomic Radius Trends… • There is a general decrease in atomic radius from left to right, caused by increasing positive charge in the nucleus – the nucleus pulls the electrons in tighter. • Atomic radius generally increases as you move down a group, since an energy level is being added.

Ionic Radius… An ion is an atom or bonded group of atoms with a positive or negative charge. • When atoms lose electrons and form positively charged ions, they always become smaller for two reasons: 1. Losing valence e- leaves empty orbitals. 2. Repulsion decreases between remaining e-. • When atoms gain electrons, they can become larger due to an increase of repulsions between electrons.

Ionic Radius Trends… • The ionic radii of positive ions generally decrease from left to right. • The ionic radii of negative ions generally decrease from left to right, beginning with group 15 or 16.
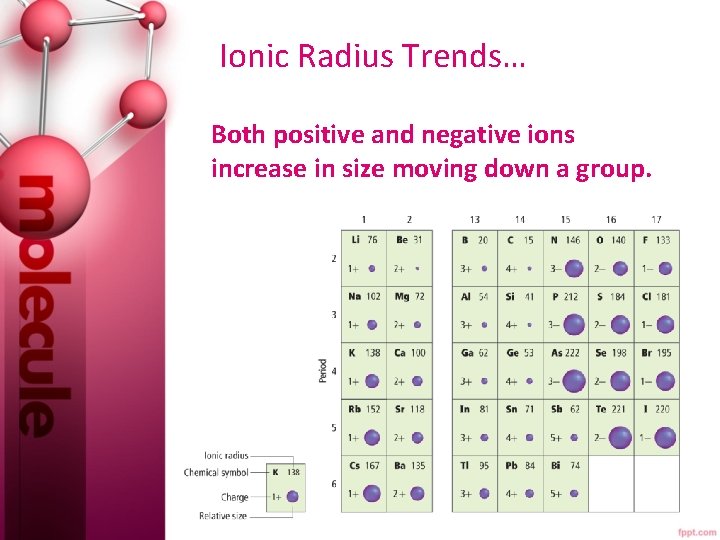
Ionic Radius Trends… Both positive and negative ions increase in size moving down a group.

Ionization Energy… Ionization energy is defined as the energy required to remove an electron from a gaseous atom. In terms of IE, electrons are removed one at a time. The energy required to remove the first electron is called the first ionization energy.
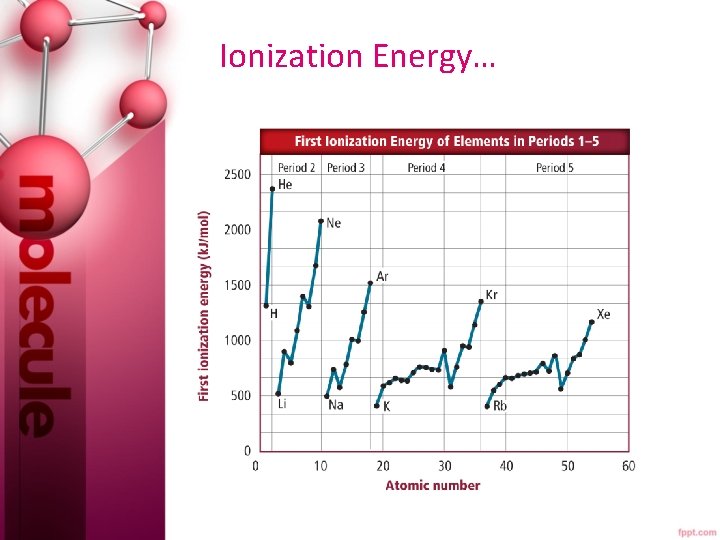
Ionization Energy…
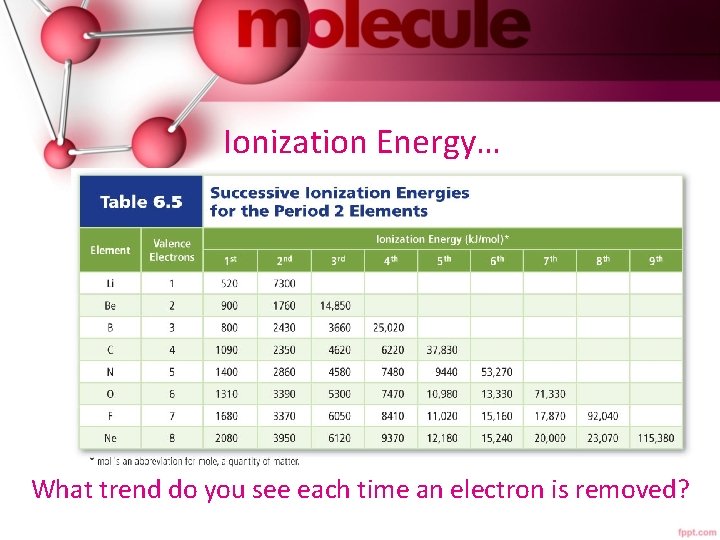
Ionization Energy… What trend do you see each time an electron is removed?

Ionization Energy… • The ionization at which the large increase in energy occurs is related to the number of valence electrons. • First ionization energy increases from left to right across a period, since metals tend to lose e- and nonmetals tend to gain e-. • First ionization energy decreases down a group because atomic size increases and less energy is required to remove an electron farther from the nucleus.
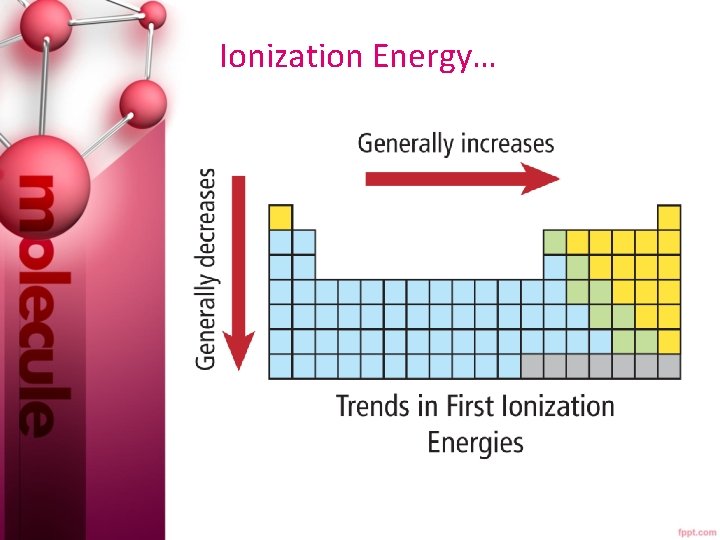
Ionization Energy…

Ionization Energy… • The octet rule states that atoms tend to gain, lose or share electrons in order to acquire a full set of eight valence electrons. • The octet rule is useful for predicting what types of ions an element is likely to form.

Electronegativity… • The electronegativity of an element indicates its relative ability to attract electrons in a chemical bond. • Electronegativity decreases down a group and increases left to right across a period. • Metals have low electronegativites and nonmetals have high electronegativites.

Electronegativity… Why don’t the noble gases have EN values listed?

- Slides: 29