Ch 6 Notes The Periodic Table elements are

Ch 6 Notes

The Periodic Table • elements are arranged in groups based on properties • Dmitri Mendeleev arranged the elements according to atomic mass and used the arrangements to predict the properties of missing elements.
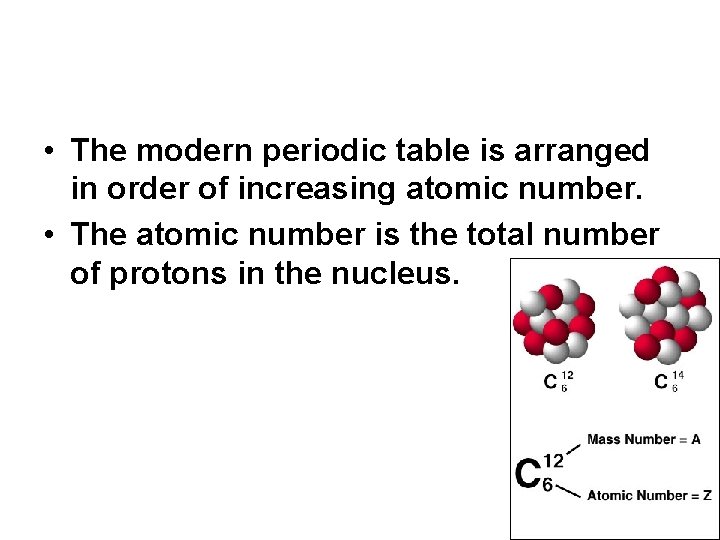
• The modern periodic table is arranged in order of increasing atomic number. • The atomic number is the total number of protons in the nucleus.

• The electron mostly determines the properties of an element.

• Indium: ______ protons ______ electrons • 49 49
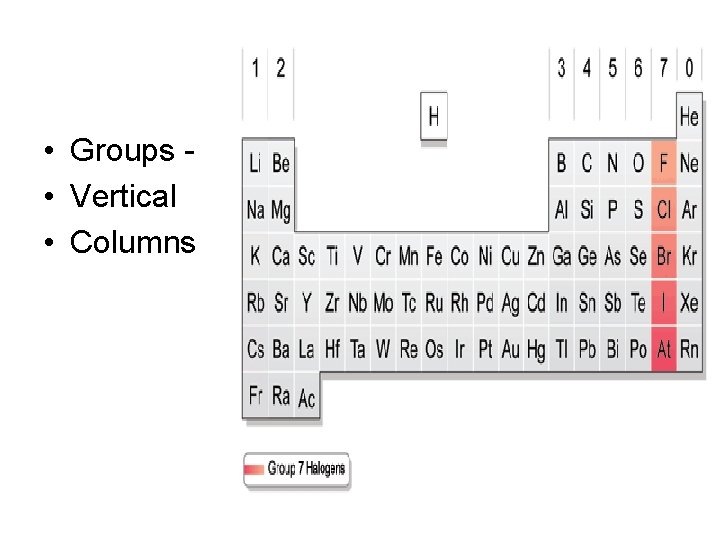
• Groups • Vertical • Columns
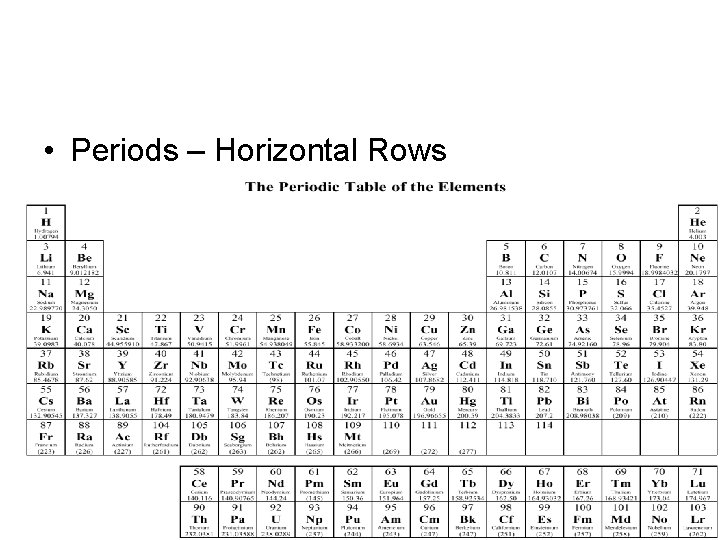
• Periods – Horizontal Rows

• Magnesium is in the same group as calcium.
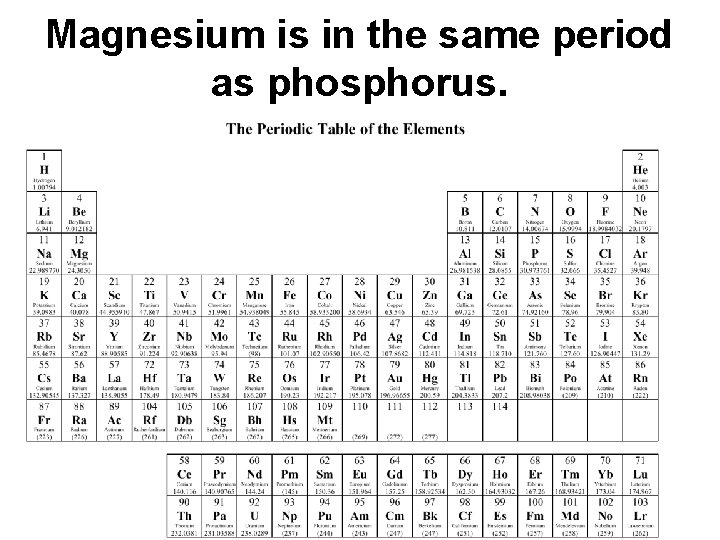
Magnesium is in the same period as phosphorus.
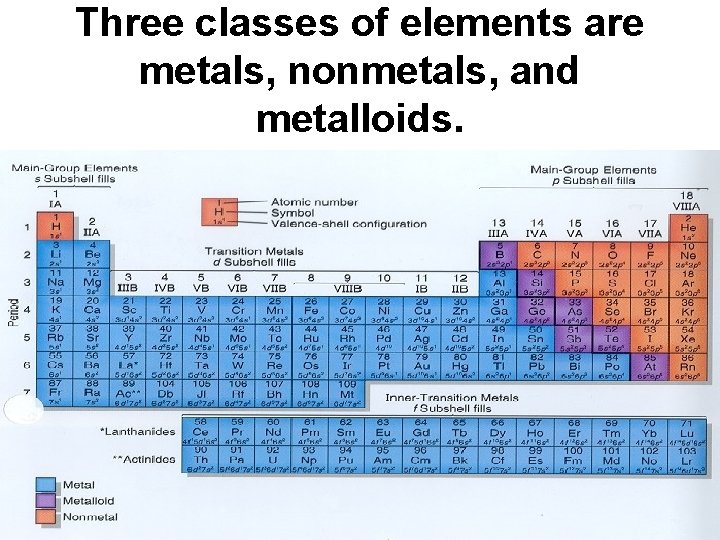
Three classes of elements are metals, nonmetals, and metalloids.
• Metals include the majority of the elements.
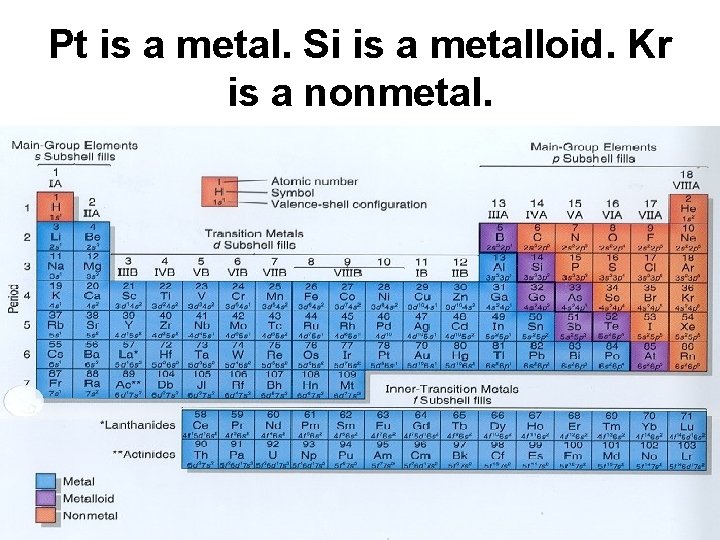
Pt is a metal. Si is a metalloid. Kr is a nonmetal.

Metals are shiny, malleable (hammered into sheets), ductile (drawn into wires), solid at room temperature, and good conductors of electricity.
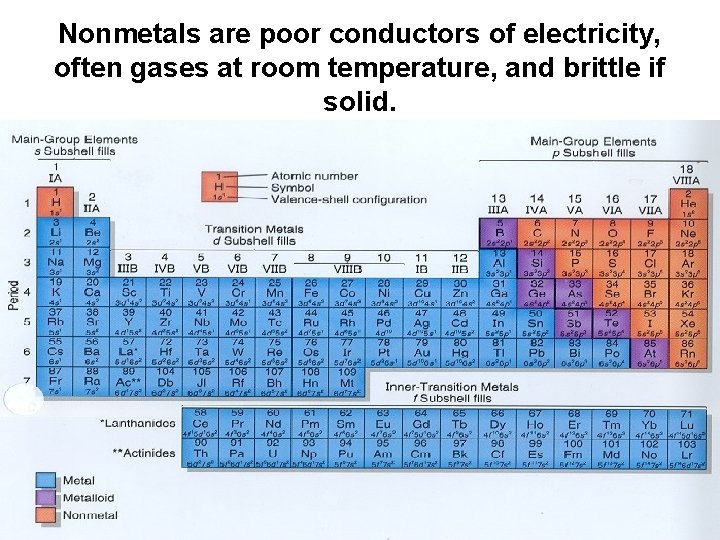
Nonmetals are poor conductors of electricity, often gases at room temperature, and brittle if solid.
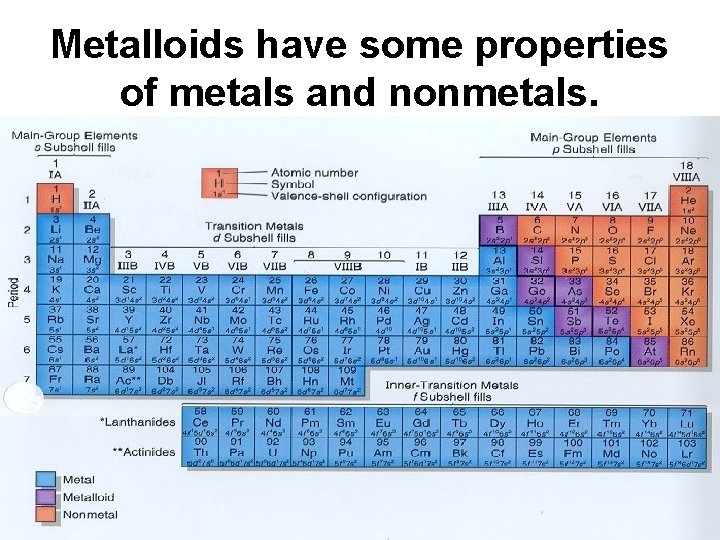
Metalloids have some properties of metals and nonmetals.

Arsenic Antimony Germanium Boron
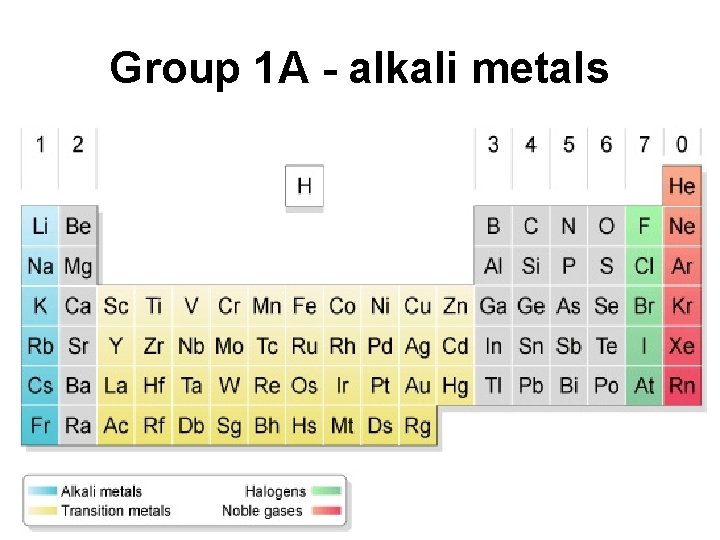
Group 1 A - alkali metals
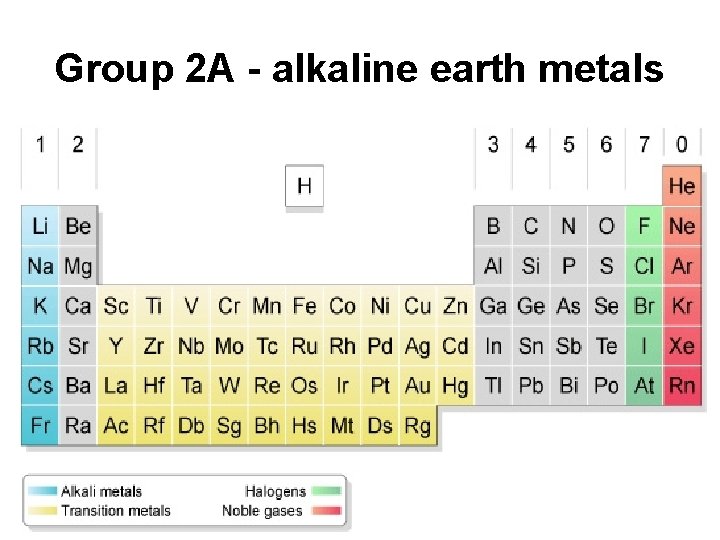
Group 2 A - alkaline earth metals
Group 7 A - halogens
Group 8 A - noble gases
Groups 1 A through 7 A representative elements Group B - transition metals
Aluminum is a representative element. Copper is a transition metal.
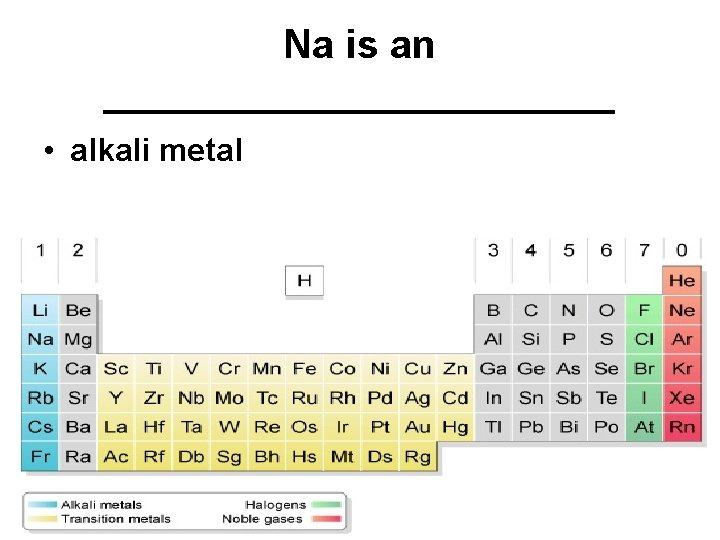
Na is an ____________ • alkali metal
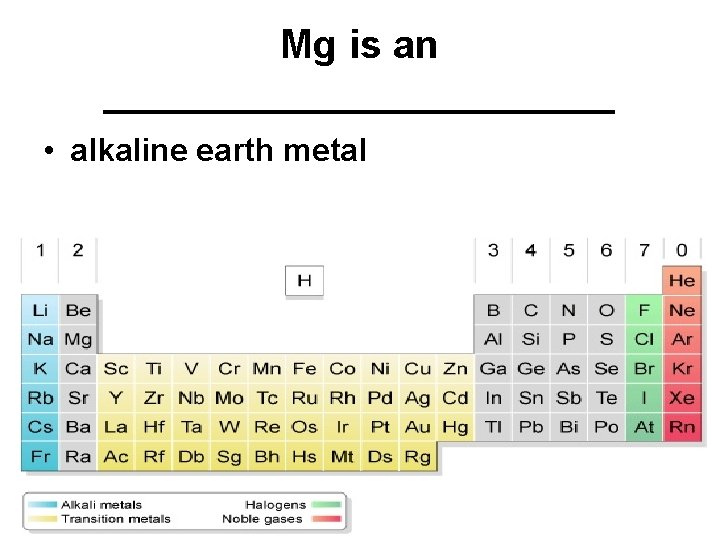
Mg is an ____________ • alkaline earth metal
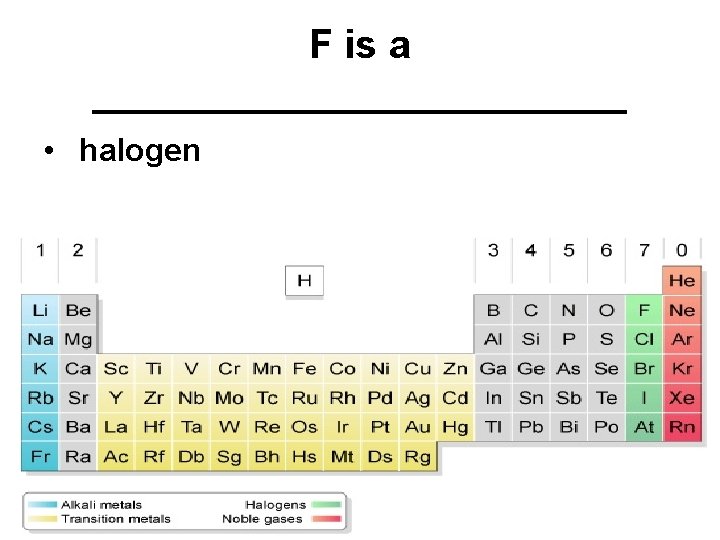
F is a ____________ • halogen
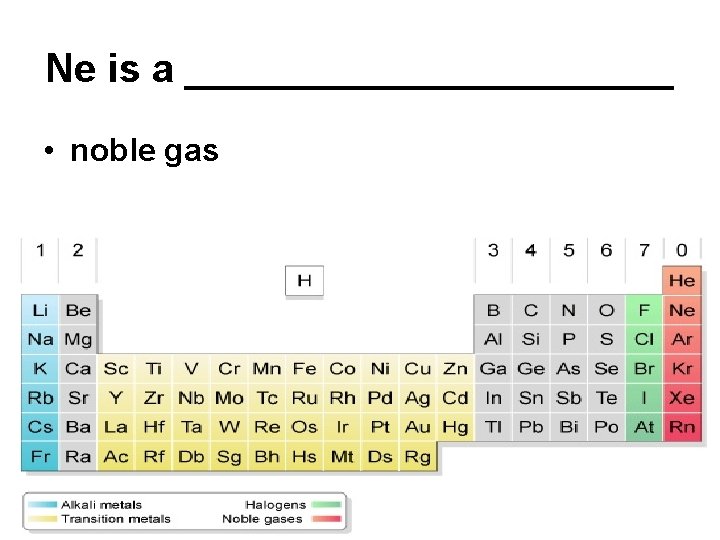
Ne is a ___________ • noble gas
Ag is a ___________ • transition metal
There are 5 electrons in the valence level of an element in Group 5 A.

N, P, As, and Sb have the same number of electrons in their valence levels.

The electron configuration for an element in the halogen group should always end with ns 2 np 5. The electron configuration of the element chlorine ends in 3 s 23 p 5.

Noble gases (inert gases) have their highest occupied s and p sublevels filled.
Fe contains an electron in a d sublevel.
- Slides: 33