Ch 6 Chemistry of Life White Board Activity
Ch. 6 Chemistry of Life

White Board Activity- What are we made of? l What would it look like if we could see things that make up our skin? l Draw a diagram to show this and come up with an explanation of what you would see. l What would an ever CLOSER view look like? l Draw a diagram to show this and come up with an explanation of what you would see.

Different levels of organization l. Cells…. . l Cell l parts (organelles) Molecules l Atom Check this out! http: //scaleofuniverse. com/

A few things to think about……. l Where do the atoms come from that make us up? l If the atoms come from foods we eat, why don’t we look like the hamburgers, french fries, and apples we eat?
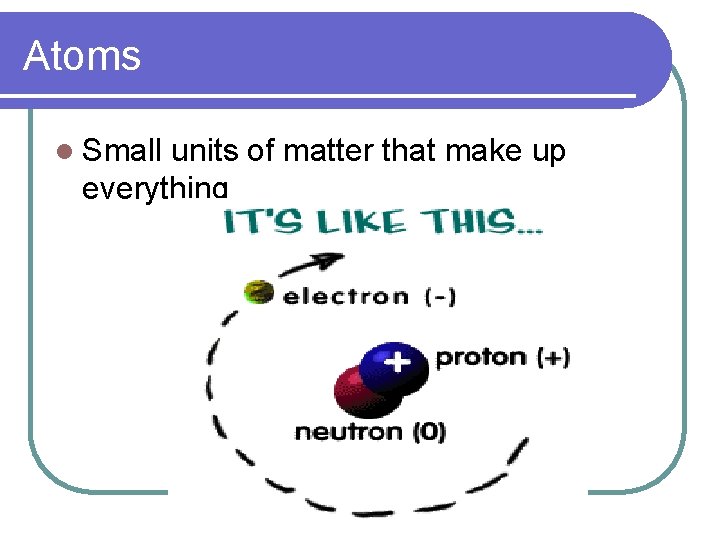
Atoms l Small units of matter that make up everything

What are the main atoms that make us up? l. C l. H l. O l. N l. P l. S

Elements l Elements are substances made up of ONE type of atom l Here are some elements found in living things

What are some of the main molecules (compounds) that make us up? l Examples of molecules in living things are: l l l Water H 2 O Carbohydrates (Lots of different kinds) Proteins (1000’s of different kinds) Fats (lots of different kinds) Nucleic acids

Organic Molecules (compounds) l Compounds that contain Carbon (and Hydrogen) l Carbon can form bonds that allow organic compounds to have these shapes: l Proteins, carbs, fats, DNA are organic molecules

WATER l Why is it soooooo important to living things? A person can die within hours if it is HOT and they are sweating l 3 -5 days normally l l What makes it different from other liquids? (you mean there are liquids that don’t contain water? What might they be? )

Importance of Water to Life l Cools us off l Transports materials in the body l Dissolves materials (in digestive system) l Helps chemical reactions happen in cells l Makes up about 85% of your brain, 80% of your blood and 70% of your muscles

Unique properties of water l. A water molecule has a negative end a positive end l Water molecules like to “stick” together

Water molecules “stick” to each other l Surface tension- a “film” occurs on the top of water – l Water forms BIG drops

Ice floats- less dense than liquid water l It is important so ice can float (lakes freeze from top to bottom) l Liquid Solid

Water resists temperature changes Water doesn’t evaporate as easy as other liquids l This is good for us- so lakes don’t evaporate in the summer.

Find these out as you read “the structure of carbohydrates, ”. p. 162 l What are carbohydrates made up of? l What do carbohydrates do for us (why do we need them? )? l What are the differences between monosaccharides, disaccharides, and polysaccharides?

Carbohydrates l Elements: Carbon, Hydrogen, Oxygen l All carbs are made of units called sugar units (glucose is a common one) bonded together l The formula for glucose is C 6 H 12 O 6 l The carbon bonding makes a sugar unit have a “stop sign” shape.

Uses of ALL carbohydrates l ENERGY for cells (and therefore us!)
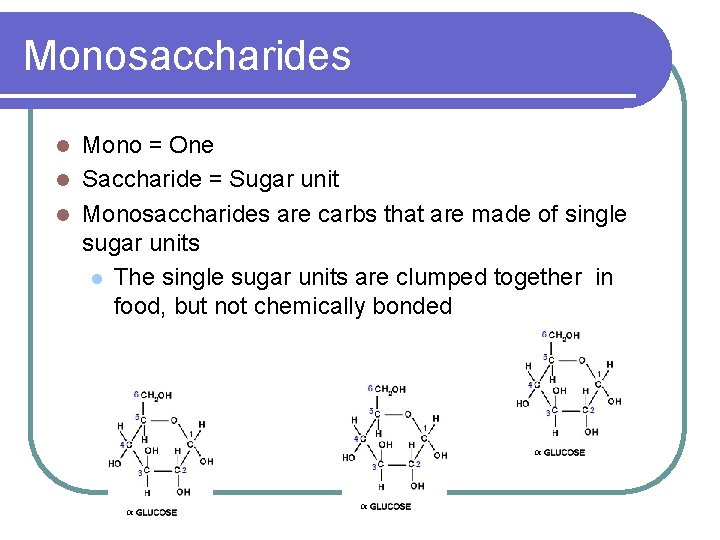
Monosaccharides Mono = One l Saccharide = Sugar unit l Monosaccharides are carbs that are made of single sugar units l The single sugar units are clumped together in food, but not chemically bonded l

Monosaccharides l Very FAST energy l Since monosaccharides are made of single sugar units they don’t need to be chemically digested- they need to be crushed up and then the sugar units can DIFFUSE into the blood stream
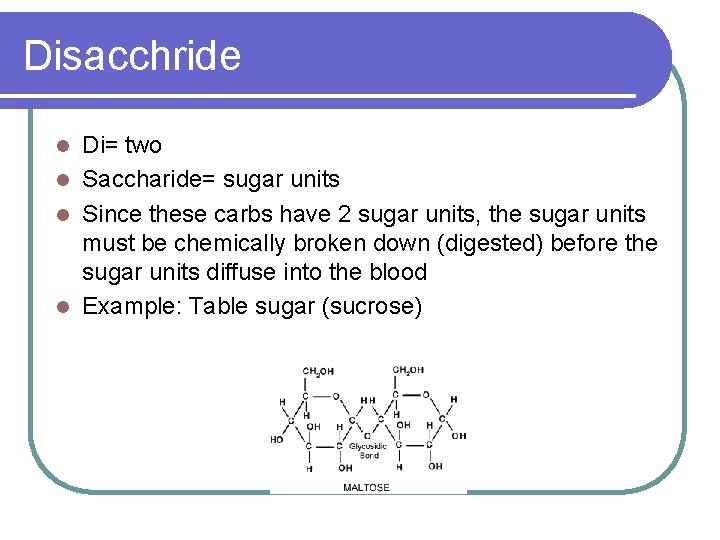
Disacchride Di= two l Saccharide= sugar units l Since these carbs have 2 sugar units, the sugar units must be chemically broken down (digested) before the sugar units diffuse into the blood l Example: Table sugar (sucrose) l
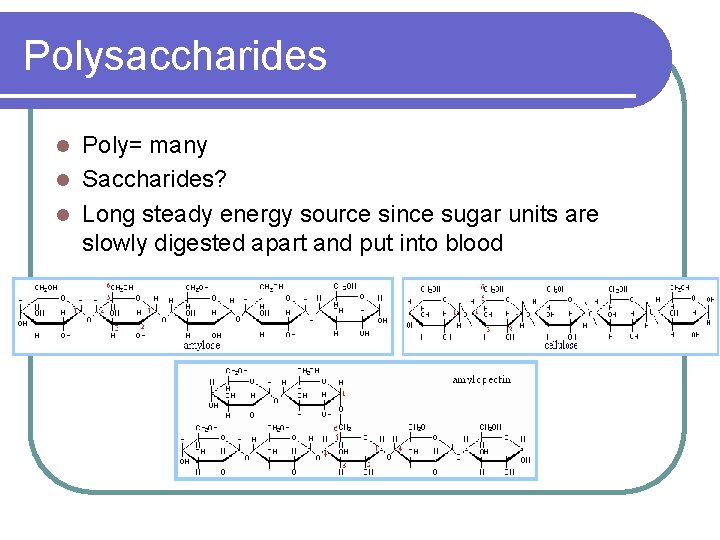
Polysaccharides Poly= many l Saccharides? l Long steady energy source since sugar units are slowly digested apart and put into blood l

Quiz- 6 pts Draw a diagram to show the difference between an atom and a molecule (or compound). Label parts of the diagram. 2. Explain 2 reasons why a living thing would die without water. Be specific and reference important jobs of water. 3. Describe 2 characteristics of water that another liquid, such as oil or rubbing alcohol, would not have. 1.

Make up quiz Describe the difference between a carbon atom and a molecule of carbon dioxide (CO 2). 2. What are 2 important jobs that water does for a living thing? 3. What are 2 characteristics of water that allow it to exist in so many places on earth in a liquid form? 1.

Review questions on Carbs What smaller unit are all carbs made of? 2. What are all carbs used for? 3. When and why would a person prefer eating a monosaccharide instead of a polysaccharide? 4. What is the main source (food) that contains disaccharides? 1.

Sources of Monosaccharides l Fruits, honey l Which would be a faster source of energy? l An apple or apple juice

Sources of Disaccharides l Any thing that contains table sugar

Sources of Polysaccharides l Any grain l Anything that contains flour l Potatoes and other “starchy” vegetables

FYI l The names of carbohydrates end in ---- ose l Examples: Glucose l Sucrose l Lactose l Amylose l

Lipids or fats Elements: C, H, O l Made of smaller units linked together- These units must be broken apart in order to get into the bloodstream l Functions: Stored energy (on the body), insulation, protect organs l

Sources of Lipids l Lard, butter, oil, and any food that contains these l Cholesterol- found in animal fat

Proteins l Elements: C, H, O, N, and sometimes S l Made of smaller units called amino acids l Functions: Cell growth l Makes up hair, nails, skin l Repairs cells l

Special proteins l Hemoglobin Found in red blood cells- attaches to oxygen l http: //www. youtube. com/watch? v=eor 6 EK_J P 40&feature=related l l Hormones l Proteins that are put into the blood stream and cause something to happen in another area of the body
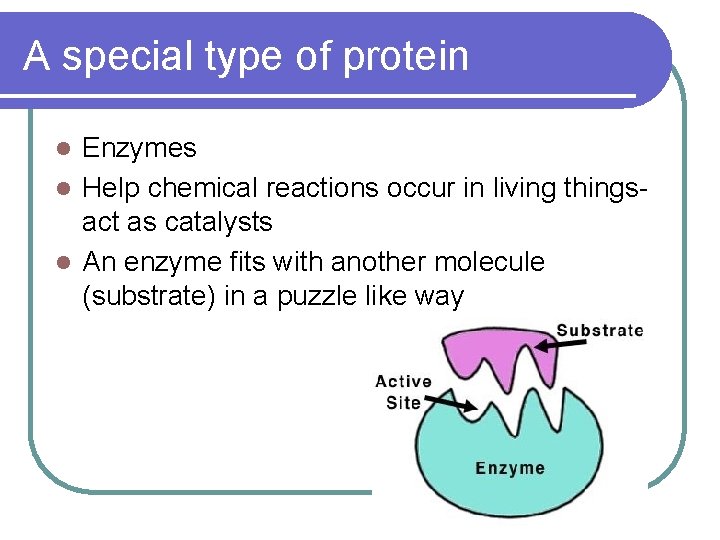
A special type of protein Enzymes l Help chemical reactions occur in living thingsact as catalysts l An enzyme fits with another molecule (substrate) in a puzzle like way l

FYI- Names of enzymes l Always end in ---ase l The beginning of the name is the same as the material it works on l Examples Sucrase breaks down sucrose (table sugar) l Lactase breaks down lactose l Amylase breaks down _______ l

l Animation on enzymes http: //www. youtube. com/watch? v=TLr 7_2 wn IXU- overview of enzymes l http: //www. youtube. com/watch? v=KED 6 BH VM 97 s - digestive enzymes l

Nucleic Acids l Elements: C, H, O, N, P l Nucleic acids are made of smaller units called nucleotides l DNA- codes for proteins cells make l RNA- helps make the proteins coded in DNA

Sources of DNA l Anything made of cells

Macromolecules l These are big (macro) molecules l Many of these are molecules made of smaller units that repeat over and over= POLYMER l What are some examples of macromolecules we have already learned?

Dehydration (condensation) reactions l. A chemical reaction in which small units are linked to make larger molecules (polymers) l Water is released l A dehydration reaction would occur in your cells when amino acids are connected to make proteins

Hydrolysis l. A hydrolysis reaction is a chemical reaction in which large polymers are broken down into smaller units l Water is used l A hydrolysis reaction occurs in the intestines when polysaccharides and proteins are broken apart into smaller units

Polymers l Polymers are large molecules made of smaller molecules that repeat over and are bonded together l The smaller molecules are sometimes called “subunits” or “building blocks”

Draw diagrams to represent each of these: l Atoms l What will you use to represent: nucleus (with protons and neutrons), electrons l Molecules l (or compounds) What will you use to represent: different atoms, bonds between atoms l Polymers l What will you use to represent: molecules that make up the polymer, bonds that hold the molecules together
- Slides: 43