Ch 6 Acids Bases and Salts ACIDS An

Ch # 6 Acids, Bases and Salts

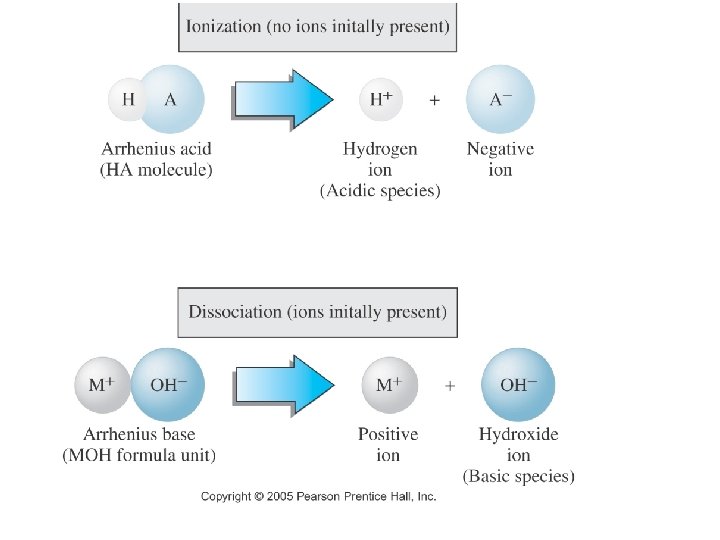
ACIDS • An acid “is a hydrogen-containing substance that produces hydrogen ions” in water. (Hydronium ion: H 3 O+) • An aqueous solution that contains more H+ ions than OH- ions is ‘acidic’

ACID PROPERTIES • Sour taste (think lemons and sour candies) • Change the color of litmus from blue to red.

Acid Properties (cont. ) • React with a base to produce water and salt in a double displacement neutralization reaction. • React with metals such as zinc and magnesium to produce hydrogen gas in a single replacement reaction.

BASES • An base is a hydroxide-containing substance that dissociates to produce hydroxide ions in aqueous solution. • An aqueous solution that contains more OH- ions than H+ ions is ‘basic’

BASE PROPERTIES • Bitter or caustic taste. • A slippery, soapy feeling. • The ability to change litmus red to blue. • The ability to interact with acids in a neutralization reaction to produce water and salt.

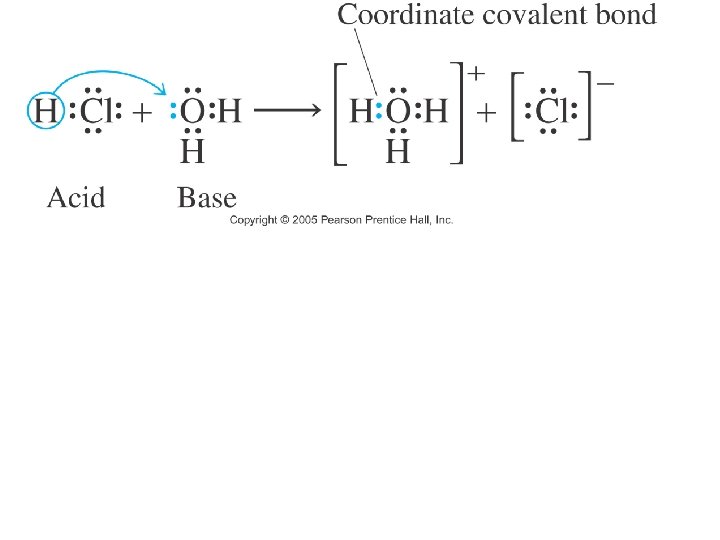
Ionization/Dissociation. • Ionization: A process in which ions are produced from a molecular compound when dissolved in a solvent. ACIDS • Dissociation: A process in which already existing ions in an ionic compound separate when an ionic compound is dissolved in a solvent. BASES

Amphoteric substances • Glycine • Hydrogen carbonate
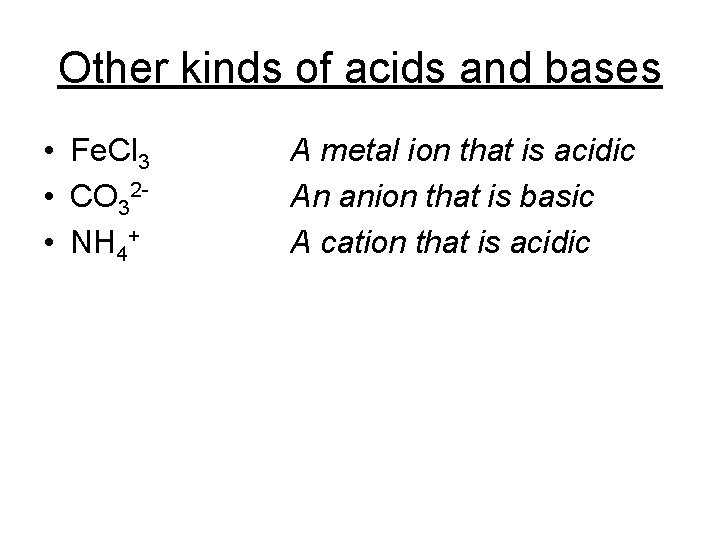
Other kinds of acids and bases • Fe. Cl 3 • CO 32 - • NH 4+ A metal ion that is acidic An anion that is basic A cation that is acidic
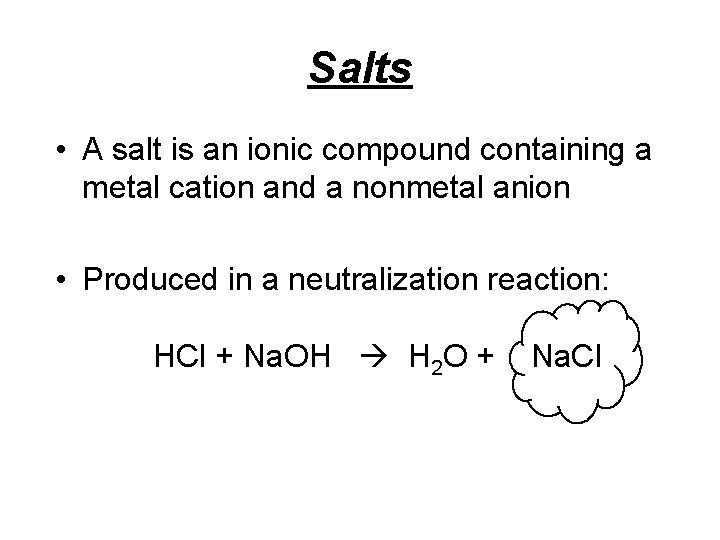
Salts • A salt is an ionic compound containing a metal cation and a nonmetal anion • Produced in a neutralization reaction: HCl + Na. OH H 2 O + Na. Cl

6. 3 Conductance of A, B, S Solutions • Strong electrolyte • Weak electrolyte • Non-electrolyte

6. 4 Dissociation of A & B • Strong acids & bases – strong electrolyte • Weak acids & bases – weak electrolyte
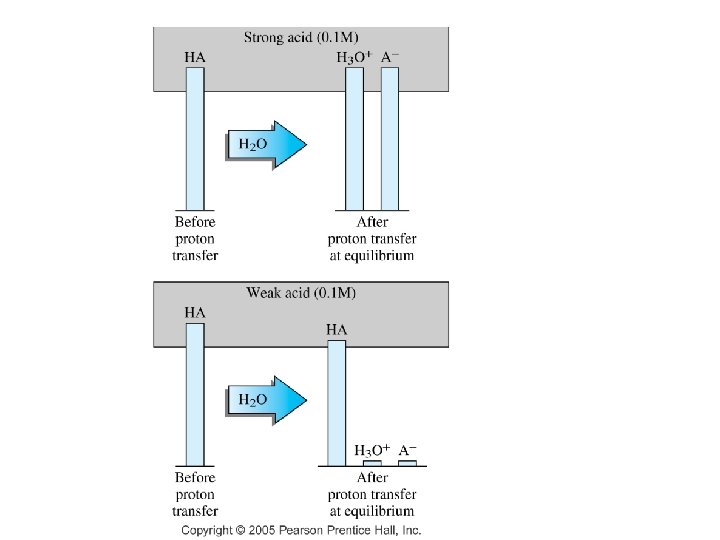


6. 5 Buffers • A solution that resists major changes in p. H when small amounts of acid or base is added to it. 1) A substance to react with and remove added base. • 2) A substance to react with and remove added acid. • 3) weak acid-salt.

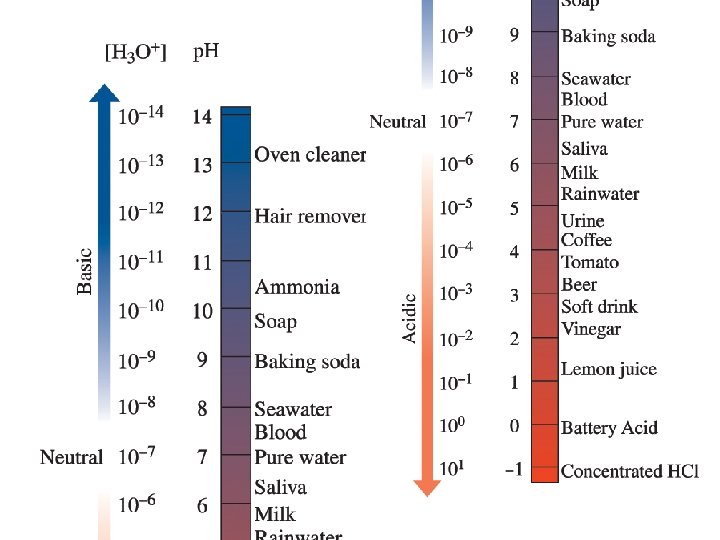
6. 6 p. H and Acidic / Basic Solutions
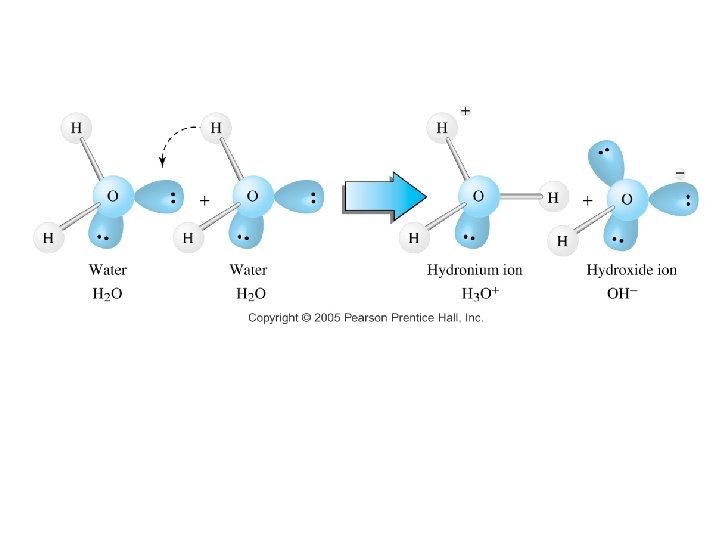

Self ionization of water: • • Ion product constant for water: Kw = [H 3 O+] [OH-] = 1. 00 x 10 -14 • If [H 3 O+] = 7. 50 x 10 -5 M What is the [OH-] in this solution?
![+ O ] p. H = -log [H 3 + = -log [H ] + O ] p. H = -log [H 3 + = -log [H ]](http://slidetodoc.com/presentation_image_h/073be6ca6be0037f201fbf2c4d628c1a/image-21.jpg)
+ O ] p. H = -log [H 3 + = -log [H ]
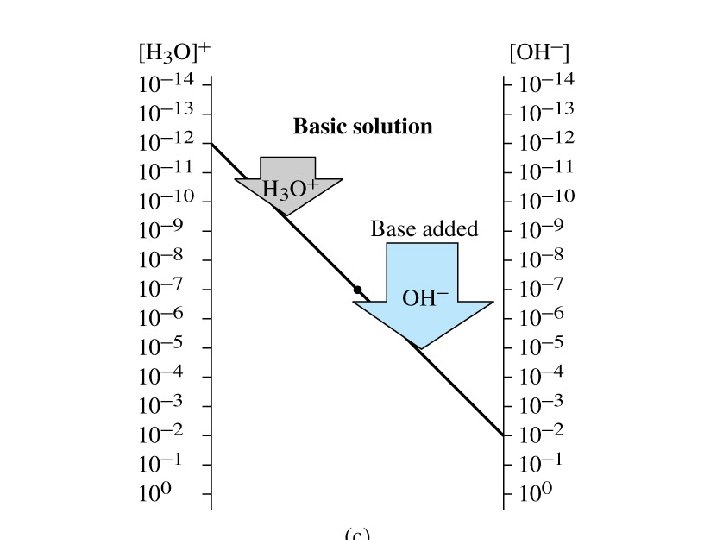
H 3 O +
![• Acidic solution: [H 3 O+]> [OH-] p. H= 0 -6 • Basic • Acidic solution: [H 3 O+]> [OH-] p. H= 0 -6 • Basic](http://slidetodoc.com/presentation_image_h/073be6ca6be0037f201fbf2c4d628c1a/image-26.jpg)
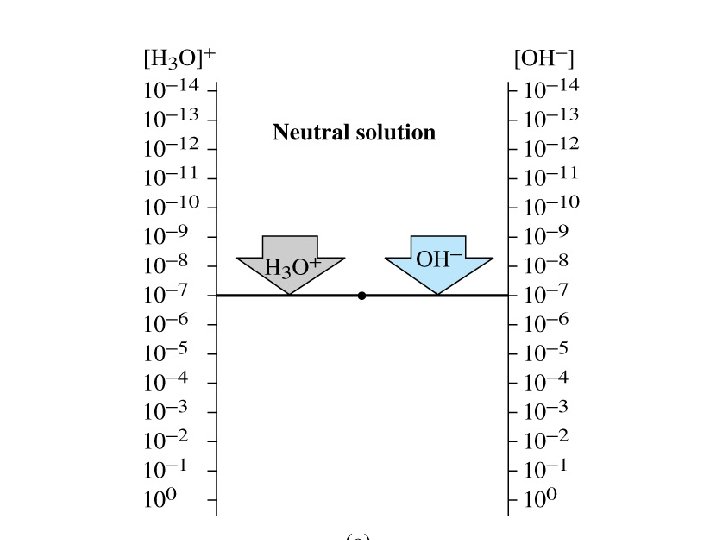
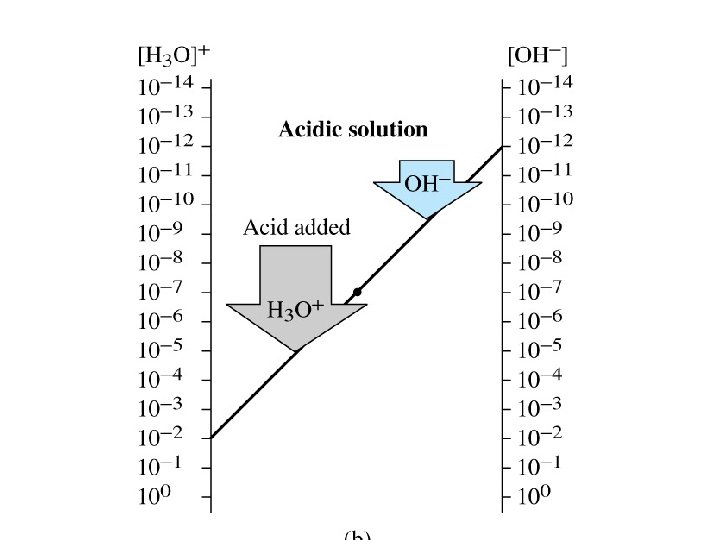
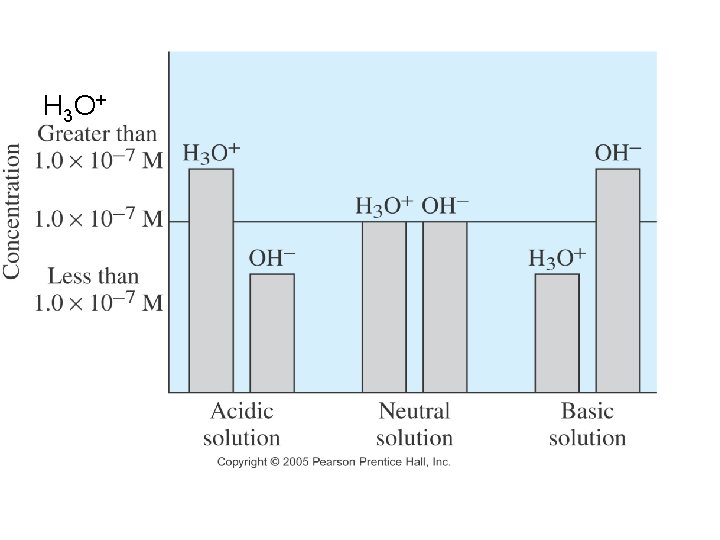
• Acidic solution: [H 3 O+]> [OH-] p. H= 0 -6 • Basic solution: [H 3 O+]< [OH-] p. H =8 -14 • Neutral solution: [H 3 O+]= [OH-] p. H =7 • p. H scale: Scale that is used to specify molar hydronium ion concentration in an aqueous solution.

Problems Calculate p. H for the following and identify as acidic or basic: [H 3 O+] = 1 X 10 -3 [H 3 O+] = 1 X 10 -9 [OH-] = 1 X 10 -4 [H 3 O+] = 3. 9 X 10 -5 [H 3 O+] = 7. 9 X 10 -11 The number of decimal places of a logarithm is equal to the number of significant figures in the original number.

Problem The p. H of a solution is 5. 70. What is the molar hydronium ion concentration for this solution? [H 3 O+] = 10 -p. H
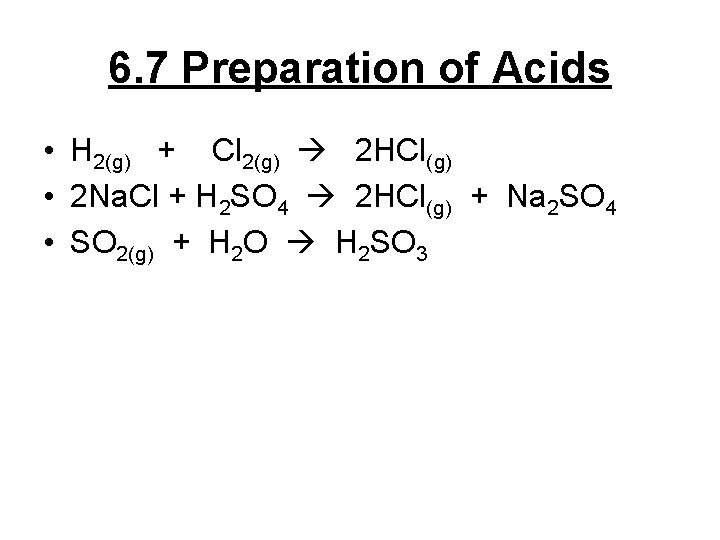
6. 7 Preparation of Acids • H 2(g) + Cl 2(g) 2 HCl(g) • 2 Na. Cl + H 2 SO 4 2 HCl(g) + Na 2 SO 4 • SO 2(g) + H 2 O H 2 SO 3

6. 8 Preparation of bases: • • • Alkali metal and water to produce hydrogen and a hydroxide salt. Ammonia in water Carbonate in water

6. 9 Preparation of Salts • The reaction of an acid with a base is called a neutralization reaction. In an aqueous solution the products are a salt and water. • Solvay process for making Na. HCO 3 and Na 2 CO 3 (p 226)

Reactions of salts: Reaction with metals: Single replacement reaction according to activity series. • Reaction with acids: Double displacement reaction. A new weaker acid, new insoluble salt, gaseous compound is one of the products. • Reaction with bases: Insoluble precipitate forms, or weaker base. • Reaction of salts with each other: Double displacement reaction. Insoluble salt is formed.

6. 10 Acidic and Basic Salts • • Ions that contain H+ Bases: See Page 213 Hydroxyapatite Ca 5 OH(PO 4)3 Reactions: Na. HCO 3 + Na. OH Na 2 CO 3 + H 2 O

6. 11 Water of Hydration • Water binds to some salts: • These are hydrates. They are used as desiccants (drying agents). • A desiccant is a hygroscopic substance that induces or sustains a state of dryness (desiccation) in its vicinity. • Anhydrous (without water of hydration)
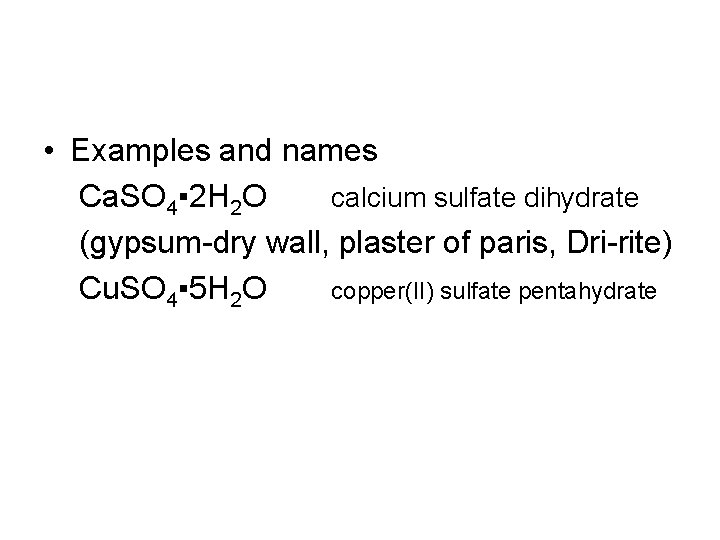
• Examples and names Ca. SO 4▪ 2 H 2 O calcium sulfate dihydrate (gypsum-dry wall, plaster of paris, Dri-rite) Cu. SO 4▪ 5 H 2 O copper(II) sulfate pentahydrate
- Slides: 36