Ch 6 7 Chemical Bonding I Introduction to

Ch. 6 & 7 - Chemical Bonding I. Introduction to Bonding (p. 161 – 163) I II IV
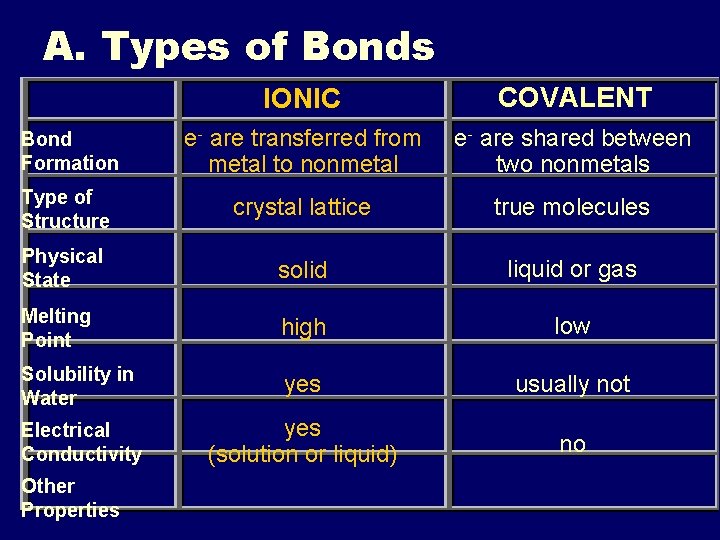
A. Types of Bonds IONIC COVALENT Bond Formation e- are transferred from metal to nonmetal e- are shared between two nonmetals Type of Structure crystal lattice true molecules Physical State solid liquid or gas Melting Point high low Solubility in Water yes usually not Electrical Conductivity yes (solution or liquid) no Other Properties
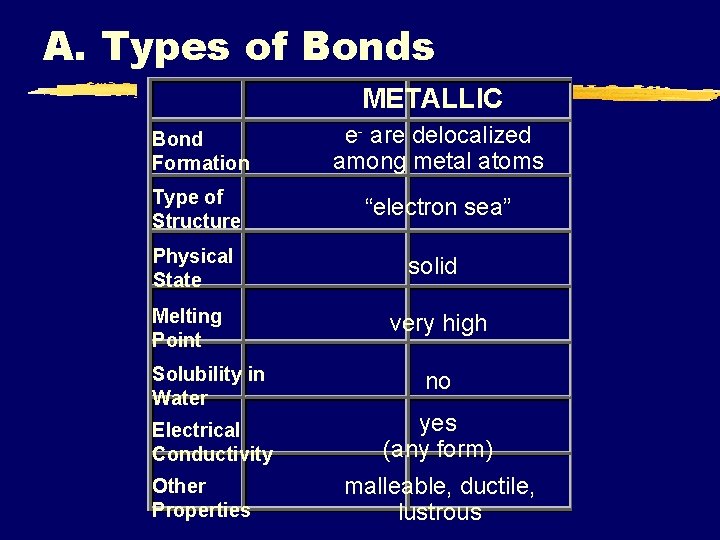
A. Types of Bonds METALLIC Bond Formation e- are delocalized among metal atoms Type of Structure “electron sea” Physical State solid Melting Point very high Solubility in Water no Electrical Conductivity yes (any form) Other Properties malleable, ductile, lustrous
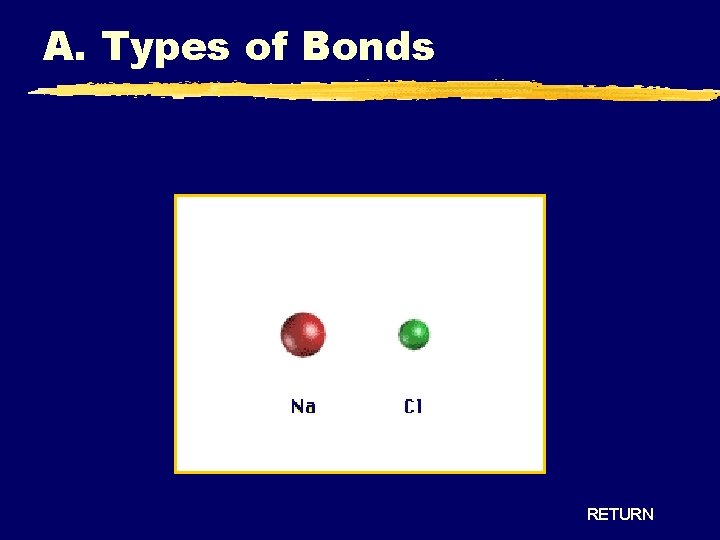
A. Types of Bonds RETURN
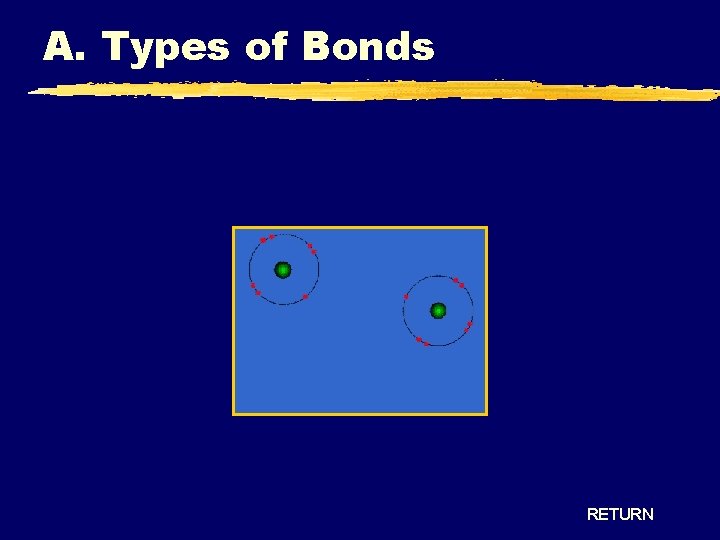
A. Types of Bonds RETURN
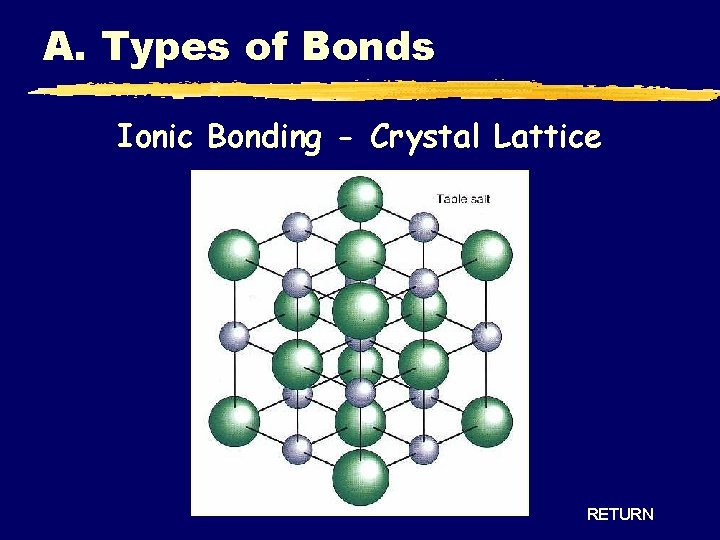
A. Types of Bonds Ionic Bonding - Crystal Lattice RETURN
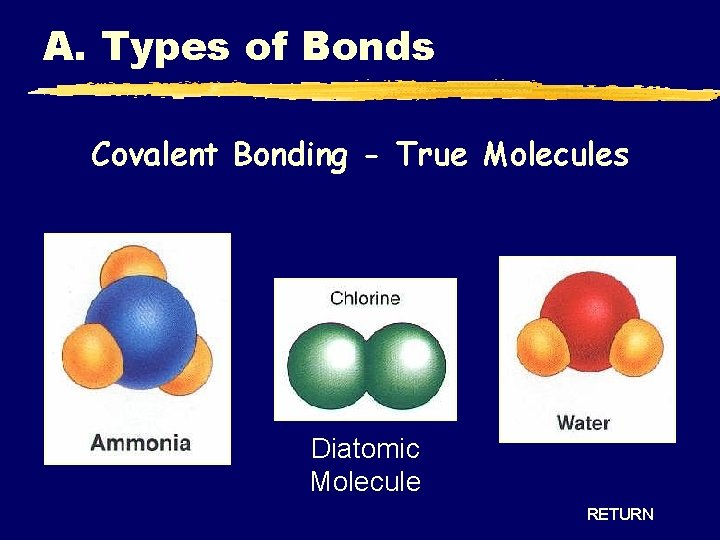
A. Types of Bonds Covalent Bonding - True Molecules Diatomic Molecule RETURN
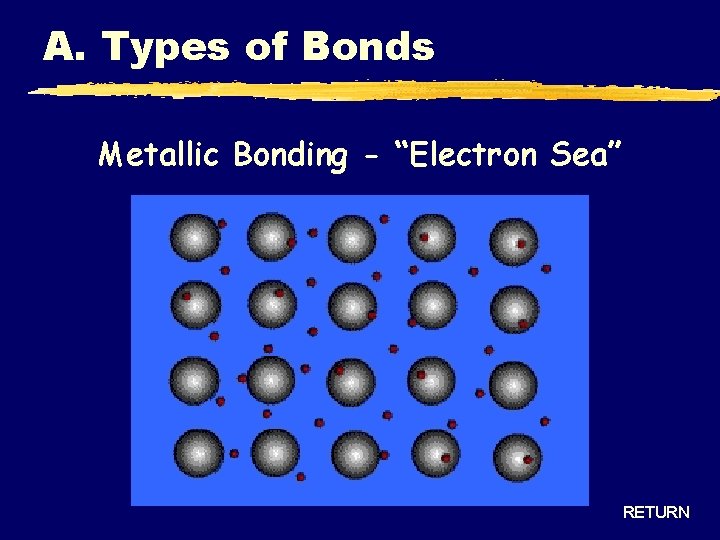
A. Types of Bonds Metallic Bonding - “Electron Sea” RETURN

B. Vocabulary z. Chemical Bond yelectrical attraction between nuclei and valence e- of neighboring atoms that binds the atoms together ybonds form in order to… xdecrease PE xincrease stability
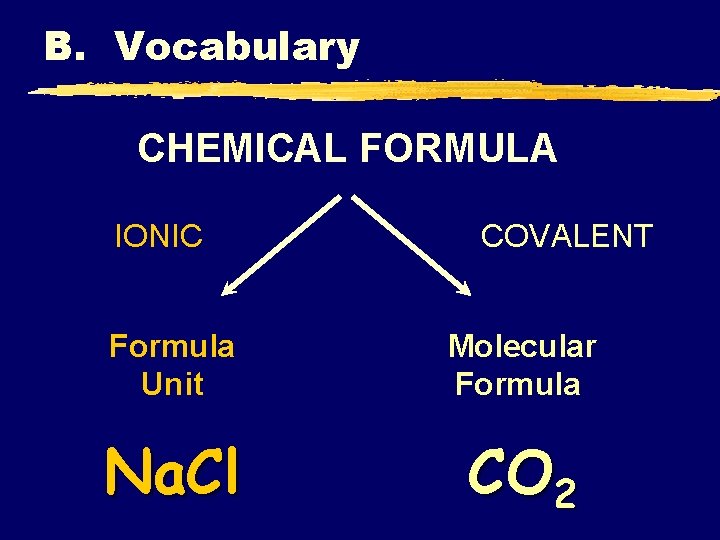
B. Vocabulary CHEMICAL FORMULA IONIC COVALENT Formula Unit Molecular Formula Na. Cl CO 2

B. Vocabulary COMPOUND 2 elements Binary Compound Na. Cl more than 2 elements Ternary Compound Na. NO 3
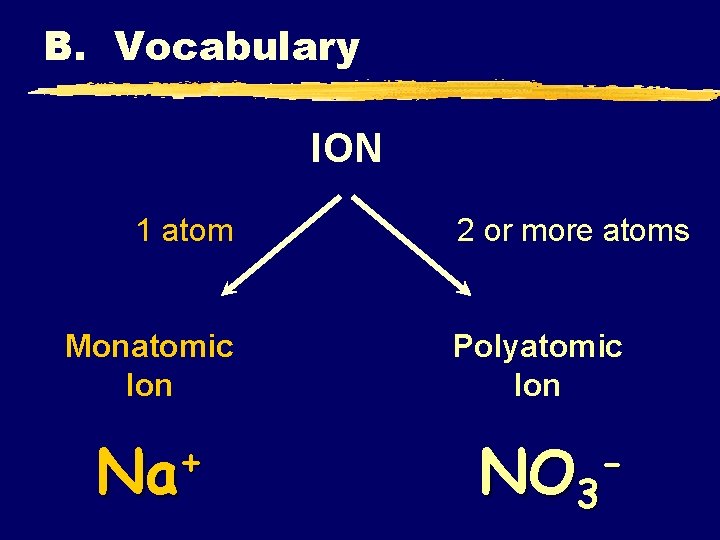
B. Vocabulary ION 1 atom Monatomic Ion + Na 2 or more atoms Polyatomic Ion NO 3
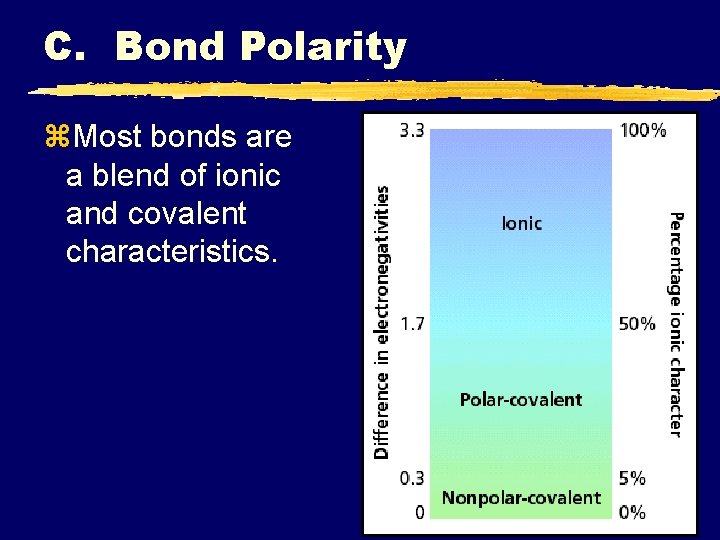
C. Bond Polarity z. Most bonds are a blend of ionic and covalent characteristics.
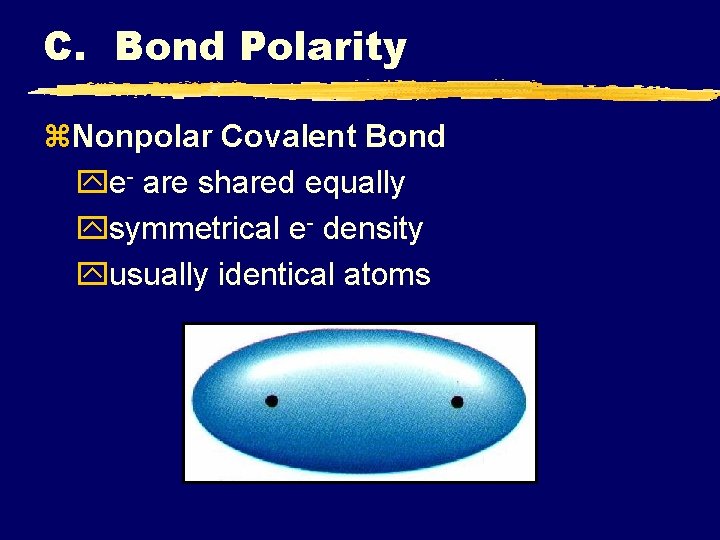
C. Bond Polarity z. Nonpolar Covalent Bond ye- are shared equally ysymmetrical e- density yusually identical atoms
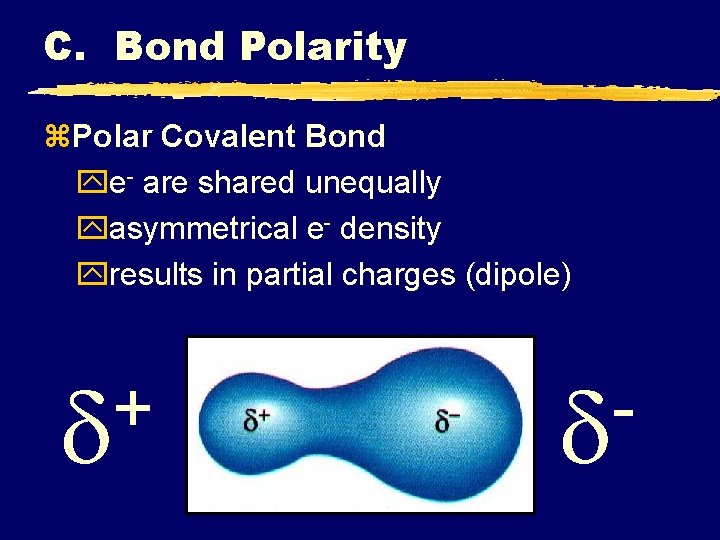
C. Bond Polarity z. Polar Covalent Bond ye- are shared unequally yasymmetrical e- density yresults in partial charges (dipole) +
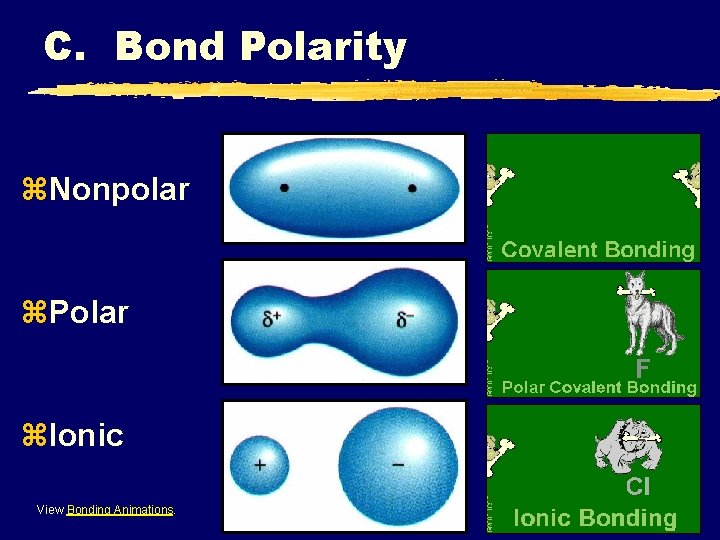
C. Bond Polarity z. Nonpolar z. Polar z. Ionic View Bonding Animations.

C. Bond Polarity z. Electronegativity y. Attraction an atom has for a shared pair of electrons. yhigher e-neg atom ylower e-neg atom +
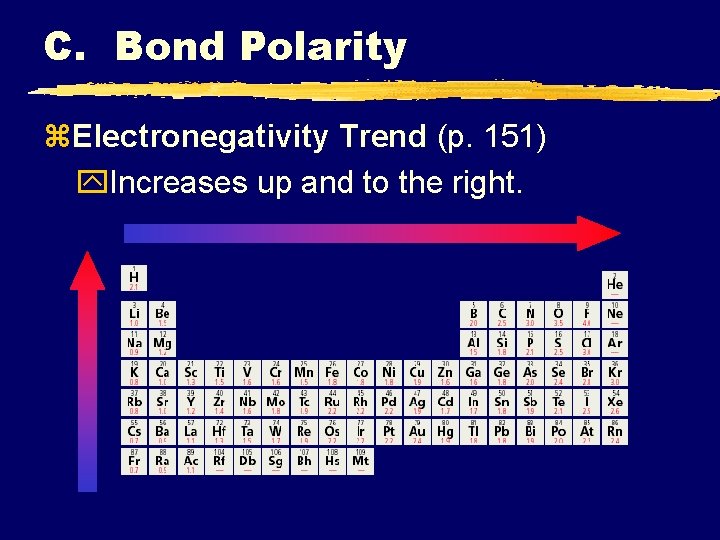
C. Bond Polarity z. Electronegativity Trend (p. 151) y. Increases up and to the right.
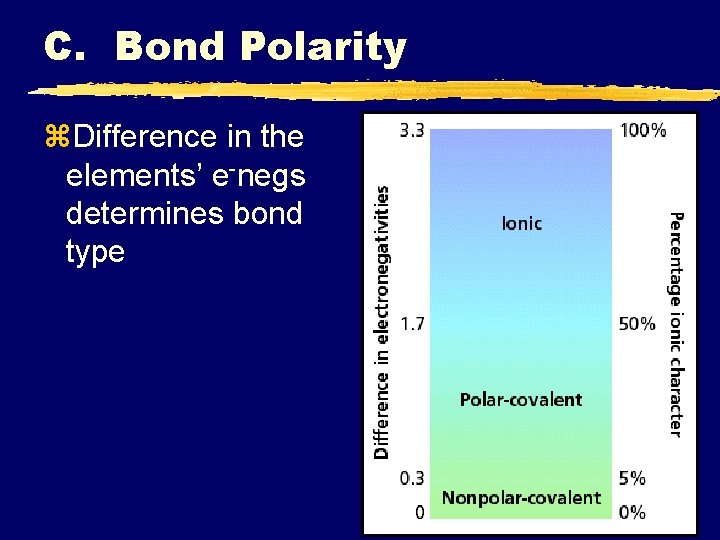
C. Bond Polarity z. Difference in the elements’ e-negs determines bond type
Ch. 6 & 7 - Chemical Bonding II. Molecular Compounds (p. 164 – 172, 211 – 213) I II IV
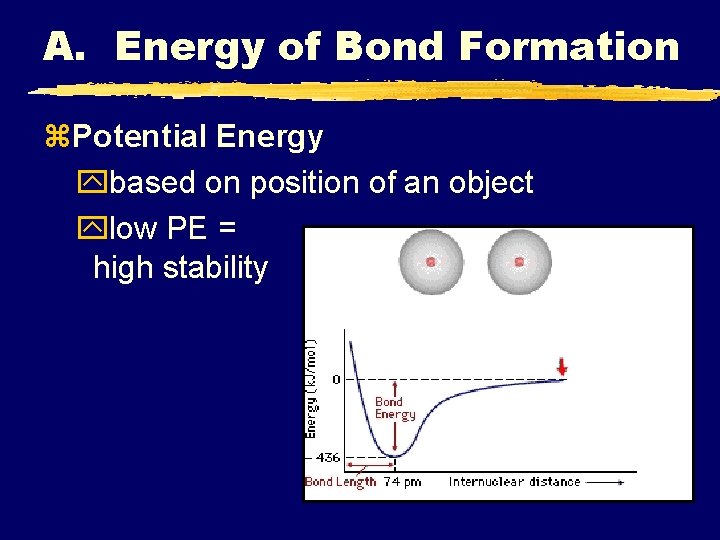
A. Energy of Bond Formation z. Potential Energy ybased on position of an object ylow PE = high stability
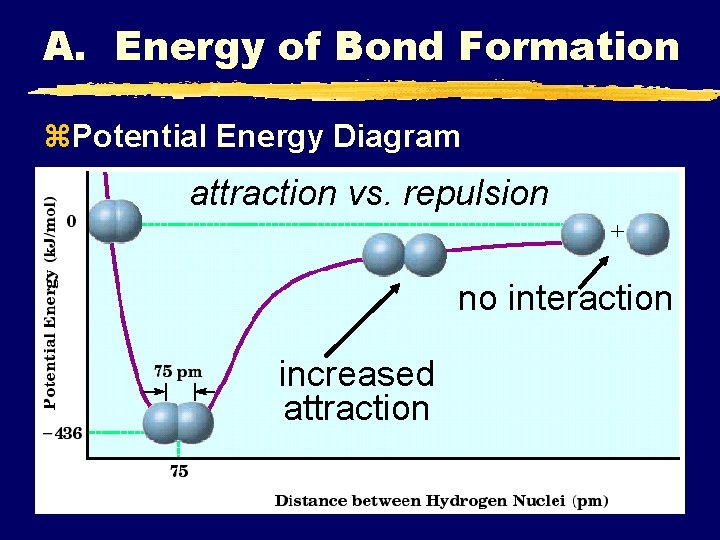
A. Energy of Bond Formation z. Potential Energy Diagram attraction vs. repulsion no interaction increased attraction
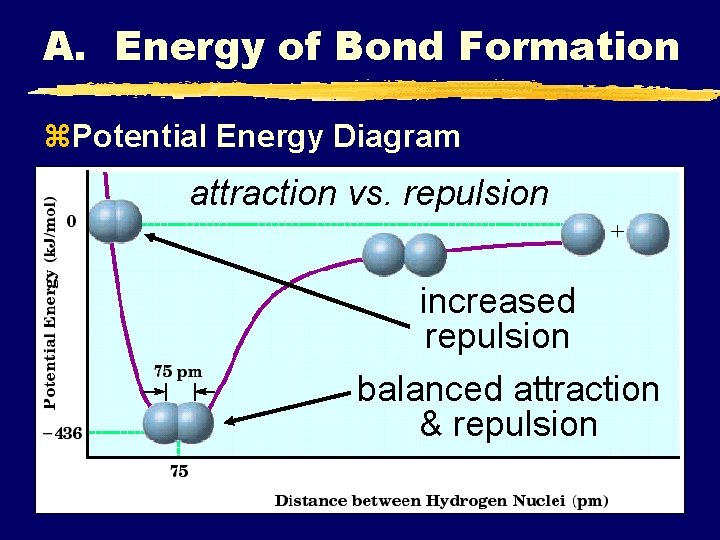
A. Energy of Bond Formation z. Potential Energy Diagram attraction vs. repulsion increased repulsion balanced attraction & repulsion
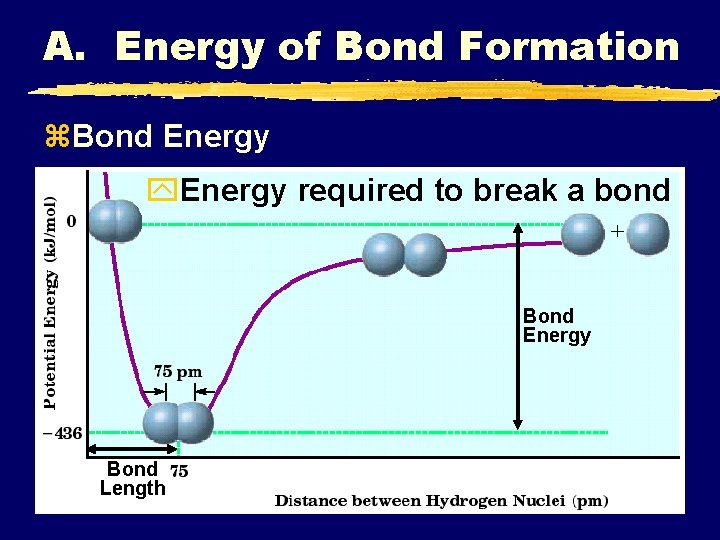
A. Energy of Bond Formation z. Bond Energy y. Energy required to break a bond Bond Energy Bond Length
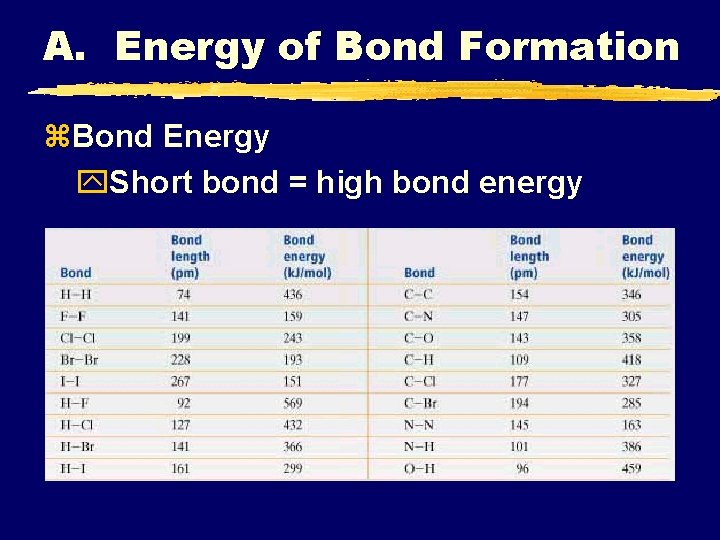
A. Energy of Bond Formation z. Bond Energy y. Short bond = high bond energy
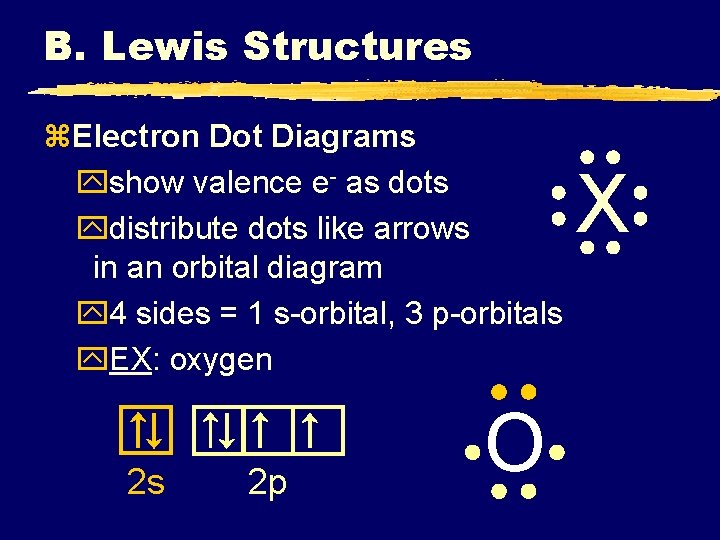
B. Lewis Structures z. Electron Dot Diagrams yshow valence e- as dots ydistribute dots like arrows in an orbital diagram y 4 sides = 1 s-orbital, 3 p-orbitals y. EX: oxygen 2 s 2 p O X

B. Lewis Structures z. Octet Rule y. Most atoms form bonds in order to obtain 8 valence ey. Full energy level stability ~ Noble Gases Ne
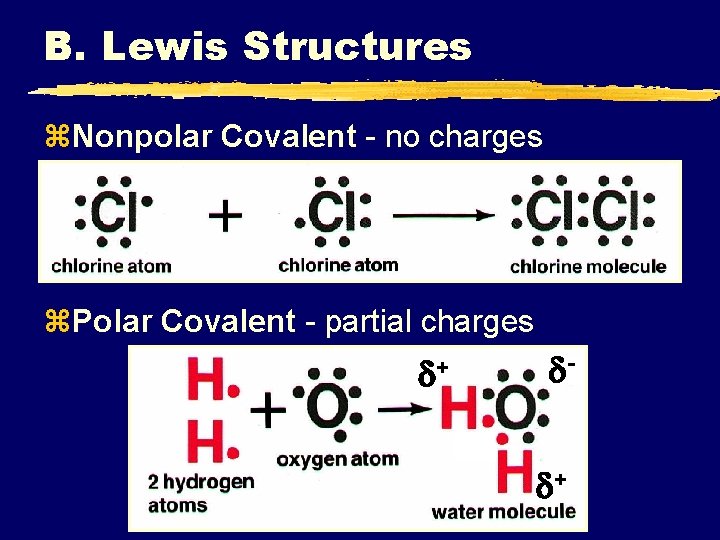
B. Lewis Structures z. Nonpolar Covalent - no charges z. Polar Covalent - partial charges + +
C. Molecular Nomenclature z. Prefix System (binary compounds) 1. Less e-neg atom comes first. 2. Add prefixes to indicate # of atoms. Omit mono- prefix on first element. 3. Change the ending of the second element to -ide.
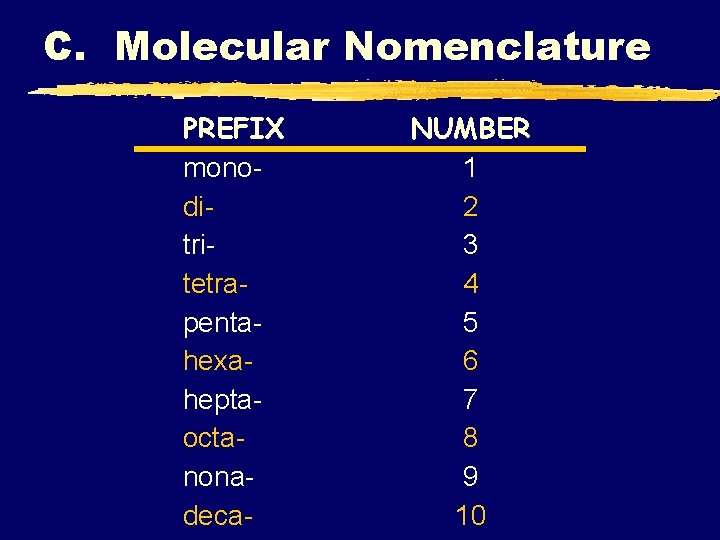
C. Molecular Nomenclature PREFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10

C. Molecular Nomenclature z. CCl 4 ycarbon tetrachloride z. N 2 O ydinitrogen monoxide z. SF 6 ysulfur hexafluoride

C. Molecular Nomenclature zarsenic trichloride y. As. Cl 3 zdinitrogen pentoxide y. N 2 O 5 ztetraphosphorus decoxide y. P 4 O 10
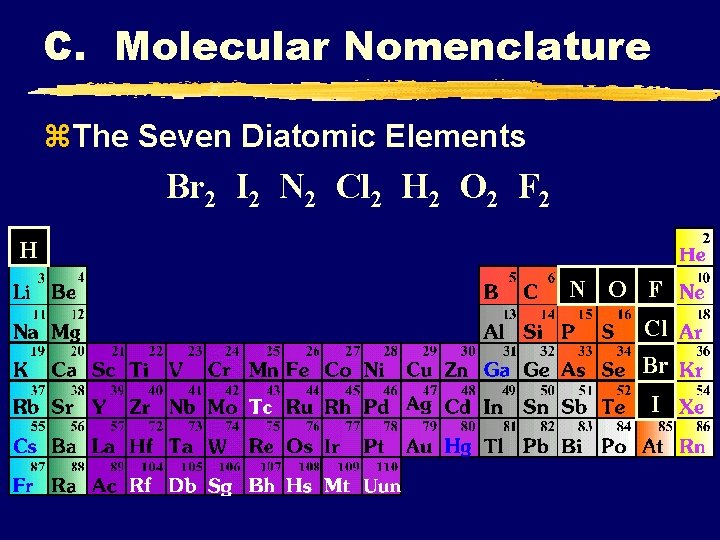
C. Molecular Nomenclature z. The Seven Diatomic Elements Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2 H N O F Cl Br I

Ch. 6 & 7 - Chemical Bonding III. Ionic Compounds (p. 176 – 180, 203 – 211) I II IV

A. Energy of Bond Formation z. Lattice Energy y. Energy released when one mole of an ionic crystalline compound is formed from gaseous ions
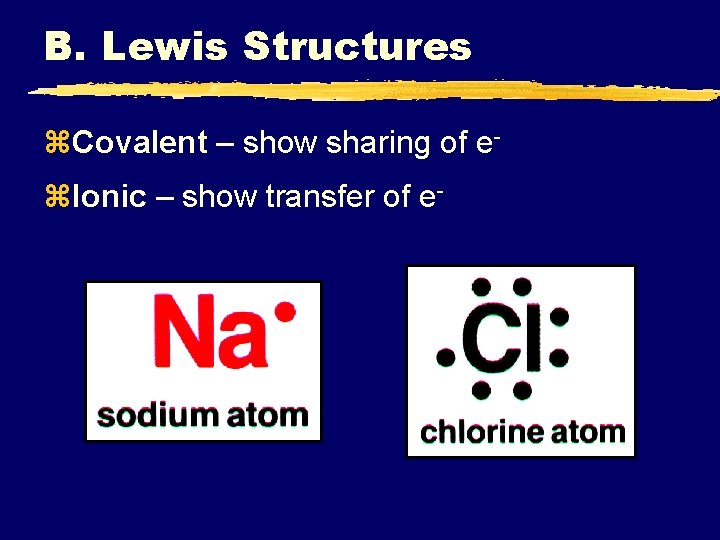
B. Lewis Structures z. Covalent – show sharing of ez. Ionic – show transfer of e-
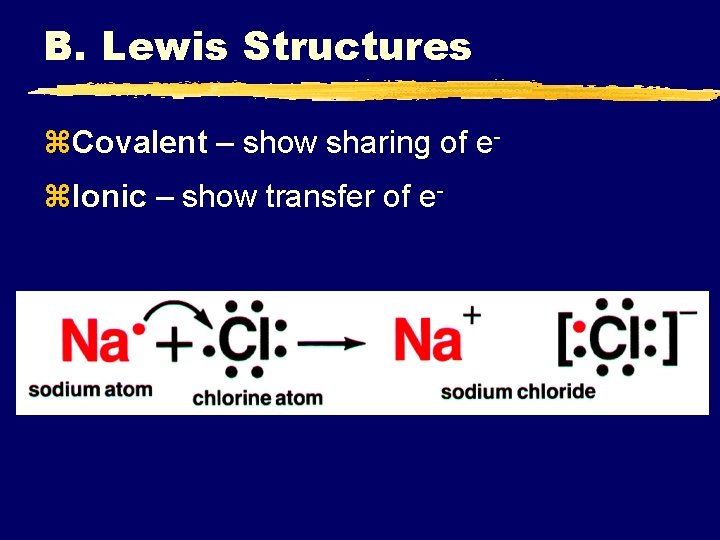
B. Lewis Structures z. Covalent – show sharing of ez. Ionic – show transfer of e-

C. Ionic Nomenclature Ionic Formulas z. Write each ion, cation first. Don’t show charges in the final formula. z. Overall charge must equal zero. y. If charges cancel, just write symbols. y. If not, use subscripts to balance charges. z. Use parentheses to show more than one polyatomic ion. z. Stock System - Roman numerals indicate the ion’s charge.

C. Ionic Nomenclature Ionic Names z. Write the names of both ions, cation first. z. Change ending of monatomic ions to -ide. z. Polyatomic ions have special names. z. Stock System - Use Roman numerals to show the ion’s charge if more than one is possible. Overall charge must equal zero.

C. Ionic Nomenclature z. Consider the following: y. Does it contain a polyatomic ion? x-ide, 2 elements no x-ate, -ite, 3+ elements yes y. Does it contain a Roman numeral? x. Check the table for metals not in Groups 1 or 2. y. No prefixes!

C. Ionic Nomenclature Common Ion Charges 1+ 0 2+ 3+ NA 3 - 2 - 1 -

C. Ionic Nomenclature zpotassium chloride y. K+ Cl- KCl zmagnesium nitrate y. Mg 2+ NO 3 - Mg(NO 3)2 zcopper(II) chloride y. Cu 2+ Cl- Cu. Cl 2

C. Ionic Nomenclature z. Na. Br ysodium bromide z. Na 2 CO 3 ysodium carbonate z. Fe. Cl 3 yiron(III) chloride
- Slides: 43