Ch 6 5 Molecular Geometry VSEPR Theory Use











- Slides: 20

Ch 6. 5 Molecular Geometry

VSEPR Theory ¡ ¡ ¡ Use VSEPR theory l Valence-Shell-Electron-Pair Repulsion l =the repulsion between electron pairs causes molecular shapes to adjust so that the valence-electron pairs stay as far apart as possible. Determined by number of valence electrons of the central atom 3 -D shape is a result of bonded pairs and lone pairs of electrons

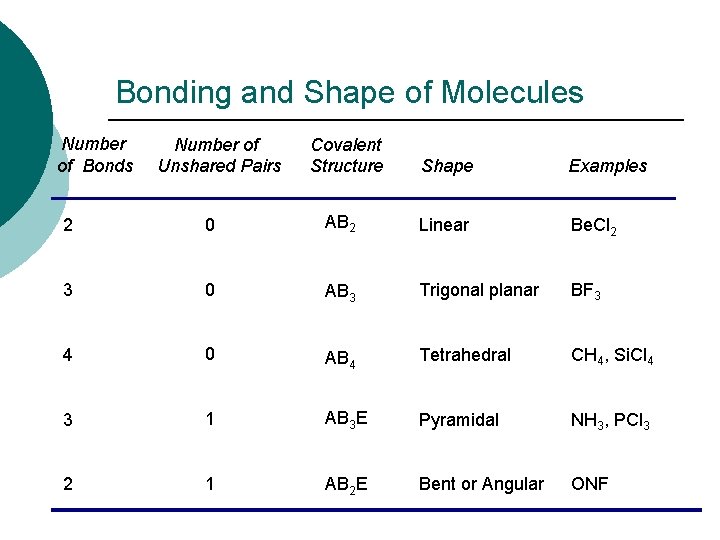
Bonding and Shape of Molecules Number of Bonds Number of Unshared Pairs Covalent Structure Shape Examples 2 0 AB 2 Linear Be. Cl 2 3 0 AB 3 Trigonal planar BF 3 4 0 AB 4 Tetrahedral CH 4, Si. Cl 4 3 1 AB 3 E Pyramidal NH 3, PCl 3 2 1 AB 2 E Bent or Angular ONF

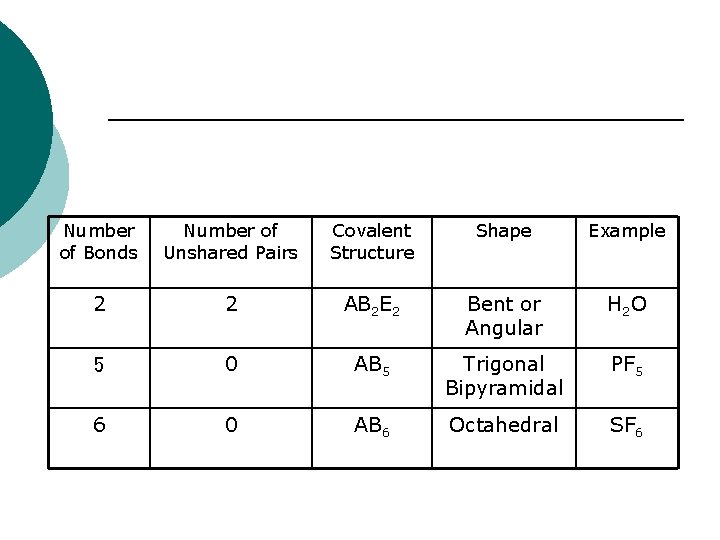
Number of Bonds Number of Unshared Pairs Covalent Structure Shape Example 2 2 AB 2 E 2 Bent or Angular H 2 O 5 0 AB 5 Trigonal Bipyramidal PF 5 6 0 AB 6 Octahedral SF 6

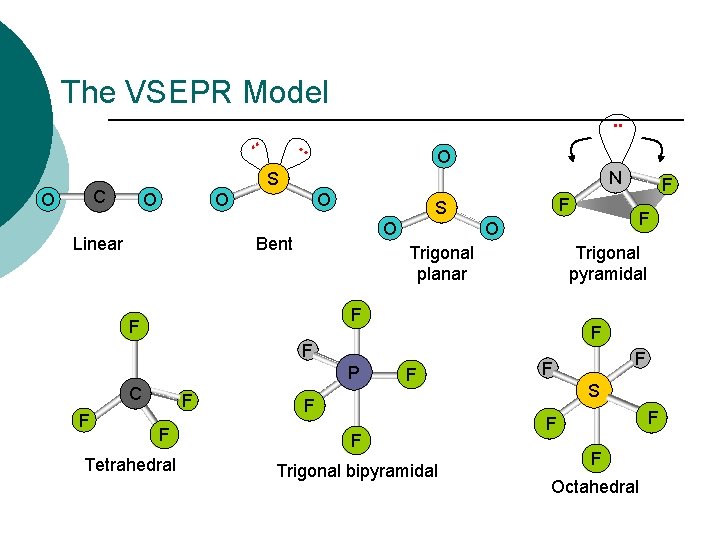
The VSEPR Model. . SO 2 O N S C O . . O O Linear O Bent Trigonal pyramidal AB 6 F F F P C F F Tetrahedral F O Trigonal planar F F F S O F F Trigonal bipyramidal F F S F F F Octahedral

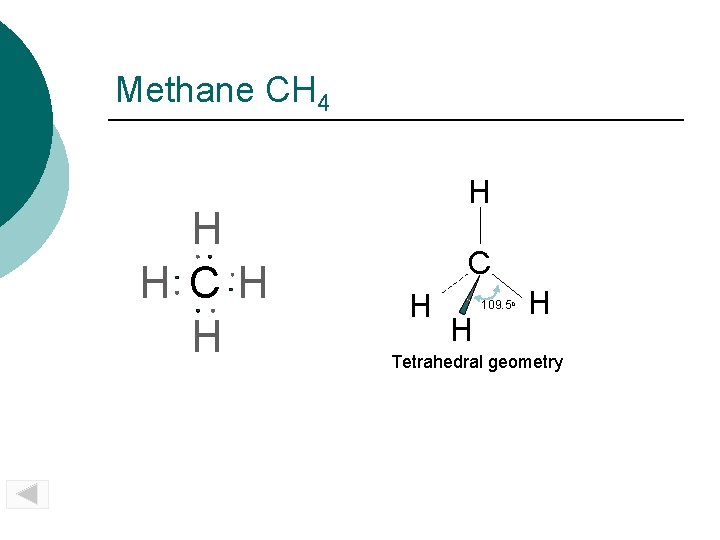
Methane CH 4 H H CH H H C H H 109. 5 o H Tetrahedral geometry

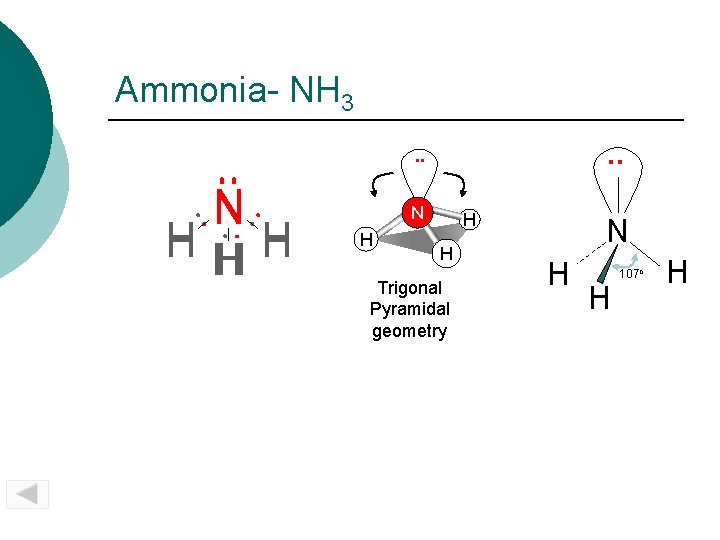
Ammonia- NH 3. . N HH H N H H H Trigonal Pyramidal geometry N H H 107 o H

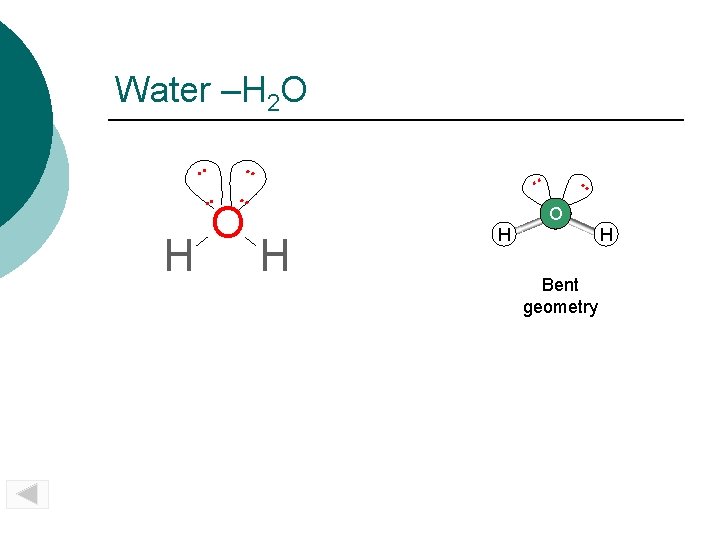
Water –H 2 O. . . H . . O . . SO 2 O H H H Bent geometry

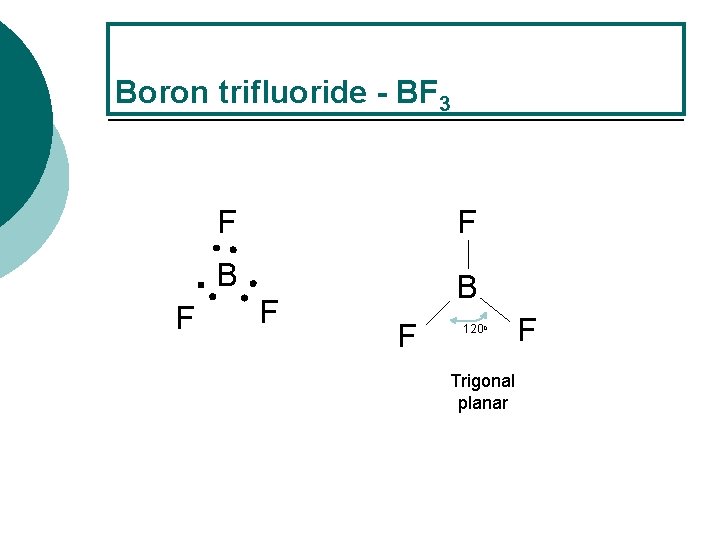
Boron trifluoride - BF 3 F F F B B F F 120 o Trigonal planar F

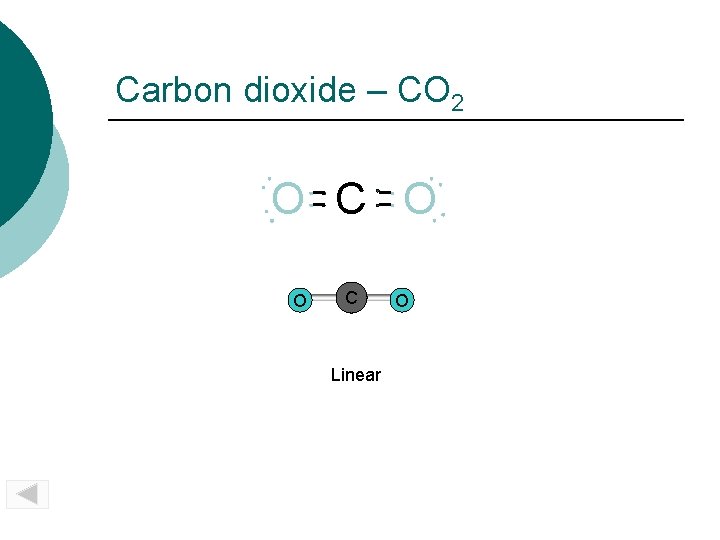
Carbon dioxide – CO 2 O C O O C Linear O

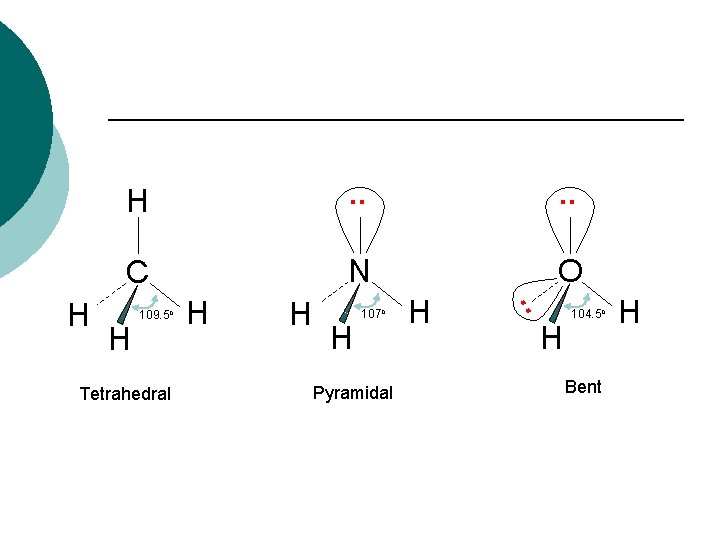
. . C N O H 109. 5 o Tetrahedral H H H 107 o Pyramidal H . . H H H 104. 5 o Bent H

Learning Check Determine the molecular geometry: A. CCl 4 B. H 2 CO

Learning Check Determine the molecular geometry: A. CCl 4 B. H 2 CO Tetrahedral

Learning Check Determine the molecular geometry: A. CCl 4 Tetrahedral B. H 2 CO Trigonal Planar

Intermolecular Forces of attraction between molecules ¡ Very weak compared to ionic and metallic bonding. ¡ The strongest occur between polar molecules. ¡

Intermolecular Forces ¡ Dipole: One end slightly positive and one end slightly negative. Can show the polar nature with an arrow pointing to the more electronegative atom. O H H H Cl ¡

Just because a molecule has a polar bond does not make it a polar molecule. ¡ The bond polarities cancel if they are in opposite directions. ¡ Example: CO 2 ¡

Attractions Between Molecules The two weakest attractions between molecules. ¡ Dipole Interactions ¡ l ¡ When polar molecules are attracted to one another. Dispersions Forces l l Caused by the motion of electrons Weakest of all interactions.

Hydrogen Bonds A strong attraction found in hydrogencontaining molecules. ¡ A hydrogen covalently bonded to a very electronegative atom is also weakly bonded to an unshared electron pair of another electronegative atom. ¡ l Example ¡ Water

Homework ¡ 6. 5 page 210 #33 -34, 37 -38 NOT 37 -42!!