Ch 6 1 Notes Covalent Bonds Review of

Ch. 6. 1 Notes Covalent Bonds

Review of Ionic Bonds • How are ionic bonds formed? • What causes the attraction between an ionic compound’s elements?

Covalent Compounds • We’ll learn… • • How they form How to draw How to name How to determine their shape

Covalent Compounds • Ionic compounds • Electrons are either lost or gained • Covalent compounds • Electrons are shared • Constituent elements are neutral • NOT ions

Covalent Compounds • How come they just share? • No element is strong enough to completely remove an e • Ex. Hydrogen • Diatomic exists as H 2 • You have 2 hydrogen atoms with same properties • Same electronegativities, ionization energies • Equal pull
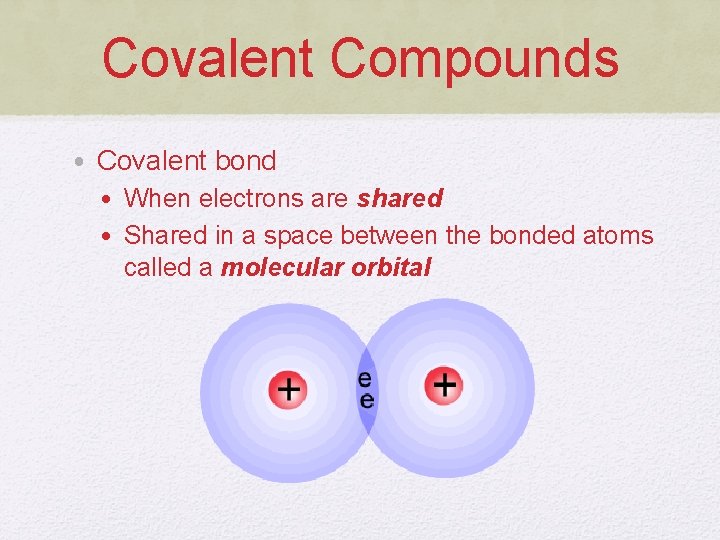
Covalent Compounds • Covalent bond • When electrons are shared • Shared in a space between the bonded atoms called a molecular orbital

Covalent Compounds • Not completely different from ionic compounds • Atoms still form bonds in way that helps them fill their octet • Trying to get 8 e • Just don’t get “full custody” of the added e • e- still belongs to the other atom, too
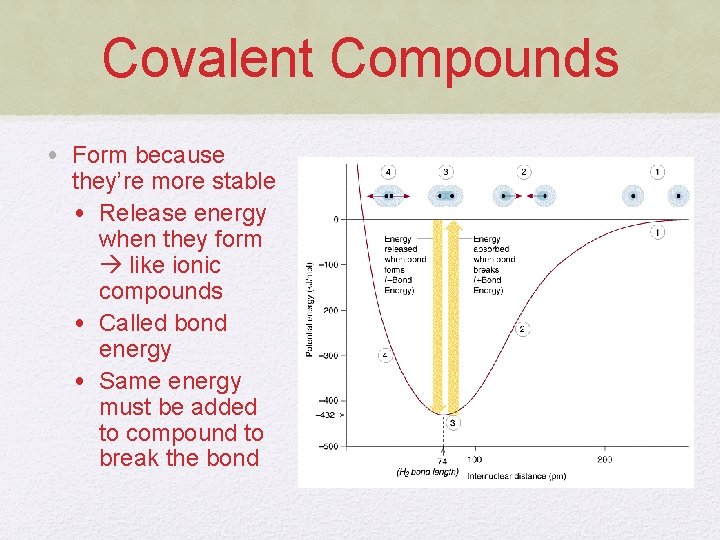
Covalent Compounds • Form because they’re more stable • Release energy when they form like ionic compounds • Called bond energy • Same energy must be added to compound to break the bond

Covalent Compounds • Bond length • Distance between the nuclei of the bonded atoms • Just an average • Covalent bonds are flexible, dynamic

Covalent Compounds • Correlation between bond energy and length • Stronger bonds generally have shorter bond lengths • Why is this?

Organize Your Information • Create a Venn diagram • Compare/contrast covalent and ionic compounds • What type of compound does a Venn diagram resemble? Why?

Covalent Bonding • Used example of hydrogen yesterday • H 2 is 2 hydrogen atoms covalently bonded together • Since both atoms were the same, had same electronegativities • Equal pull • e- shared equally • This is called a nonpolar covalent bond

Covalent Bonding • What about covalent bonds with different atoms? • Their electronegativities are different • Atom with higher electronegativity attracts emore • Creates uneven sharing • Called a polar covalent bond • e- more likely to be found closer to atom with higher electronegativity

Covalent Bonding • If one atom’s electronegativity is much higher than the other’s, it will completely take the e • Ionic bonding • Polar covalent bond is still sharing e-

Covalent Bonding • Electronegativity difference determines bond type • 0 -0. 5 – nonpolar covalent • 0. 5 -2. 1 – polar covalent • 2. 1 -3. 3 – ionic

Covalent Bonding • Electronegativity difference determines bond type • 0 -0. 5 – nonpolar covalent • 0. 5 -2. 1 – polar covalent • 2. 1 -3. 3 – ionic

Covalent Bonding • Trends • A larger difference between electronegativity values… • More polar bond… • And stronger bond

Covalent Bonding • Properties of compounds will vary based on their bond type • Stronger bonds have higher boiling, melting points
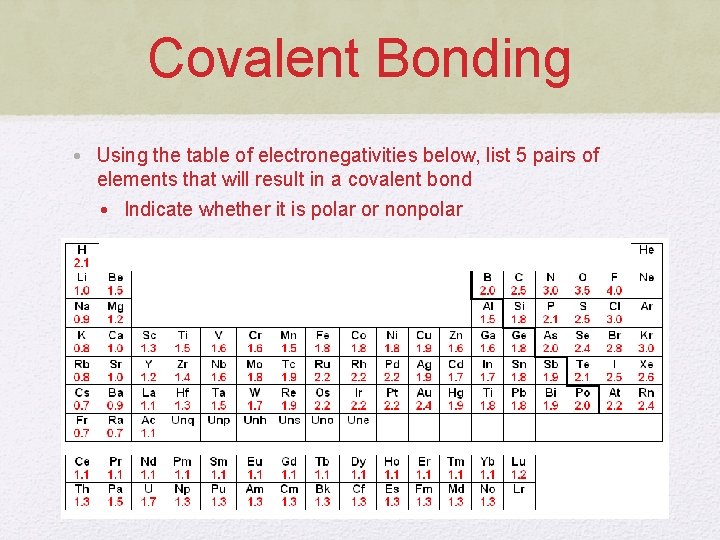
Covalent Bonding • Using the table of electronegativities below, list 5 pairs of elements that will result in a covalent bond • Indicate whether it is polar or nonpolar
- Slides: 19