Ch 5 The Periodic Table I History p
Ch. 5 - The Periodic Table I. History (p. 123 - 127)

A. Mendeleev z. Dmitri Mendeleev (1869, Russian) y. Organized elements by increasing atomic mass. y. Elements with similar properties were grouped together. y. There were some discrepancies. C. Johannesson

A. Mendeleev z. Dmitri Mendeleev (1869, Russian) y. Predicted properties of undiscovered elements.

B. Moseley z. Henry Mosely (1913, British) y. Organized elements by increasing atomic number. y. Resolved discrepancies in Mendeleev’s arrangement. C. Johannesson

Ch. 5 - The Periodic Table II. Organization of the Elements
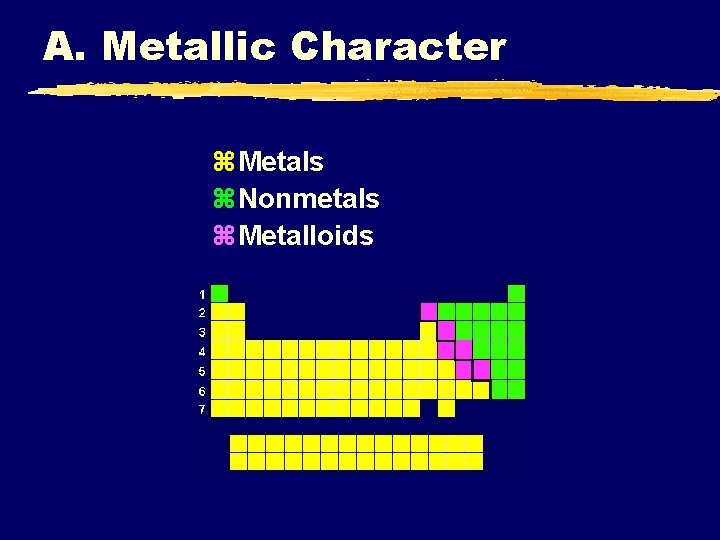
A. Metallic Character z. Metals z. Nonmetals z. Metalloids
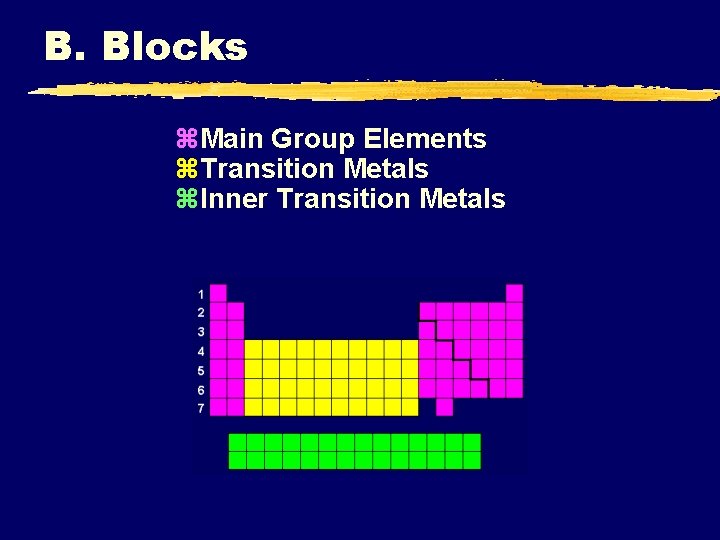
B. Blocks z. Main Group Elements z. Transition Metals z. Inner Transition Metals
C. Periods

D. Families Similar valence e- within a group result in similar chemical properties C. Johannesson

D. Families z. Alkali Metals z. Alkaline Earth Metals z. Transition Metals z. Halogens z. Noble Gases C. Johannesson
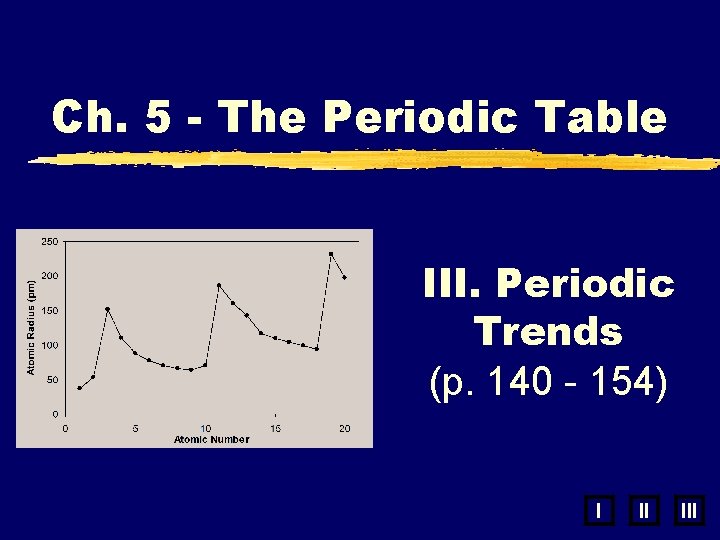
Ch. 5 - The Periodic Table III. Periodic Trends (p. 140 - 154) I II III
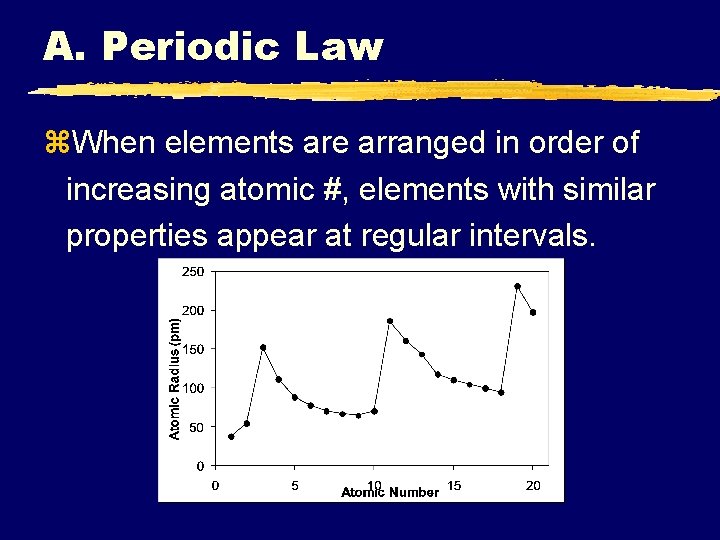
A. Periodic Law z. When elements are arranged in order of increasing atomic #, elements with similar properties appear at regular intervals.
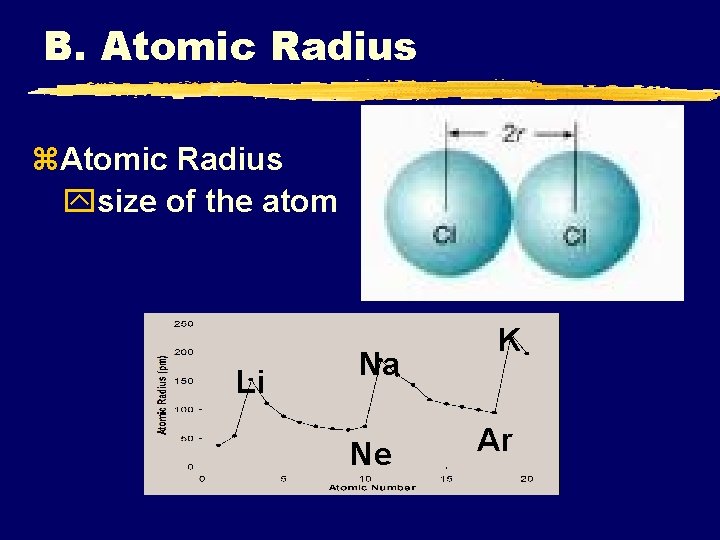
B. Atomic Radius z. Atomic Radius ysize of the atom © 1998 LOGAL Li Na Ne K Ar

B. Atomic Radius z. Atomic Radius y. Increases to the LEFT and DOWN C. Johannesson

B. Atomic Radius z. Why larger going down? y. Higher energy levels have larger orbitals y. Shielding - core e- block the attraction between the nucleus and the valence ez. Why smaller to the right? y. Increased nuclear charge without additional shielding pulls e- in tighter C. Johannesson
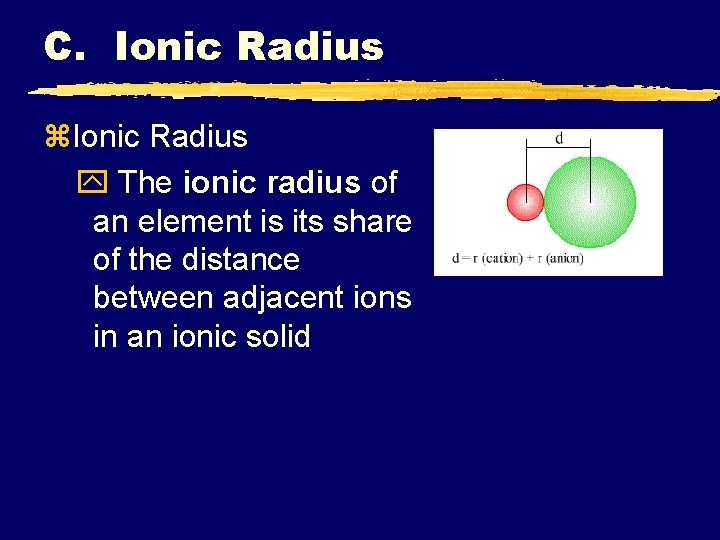
C. Ionic Radius z. Ionic Radius y The ionic radius of an element is its share of the distance between adjacent ions in an ionic solid
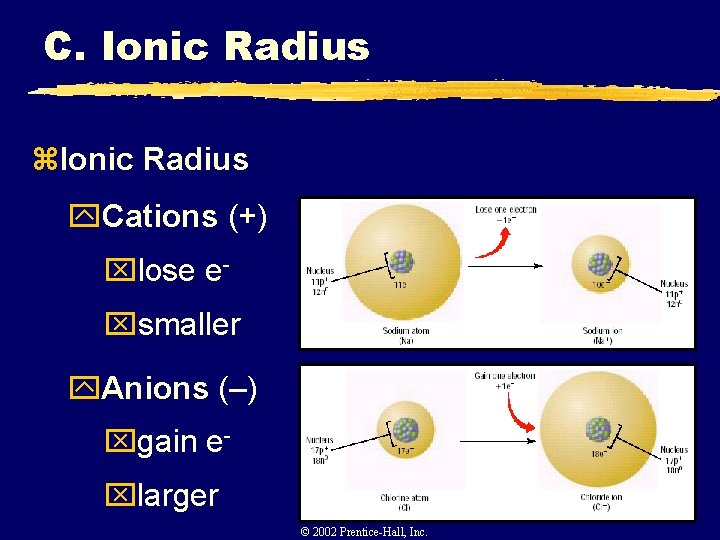
C. Ionic Radius z. Ionic Radius y. Cations (+) xlose exsmaller y. Anions (–) xgain exlarger C. Johannesson © 2002 Prentice-Hall, Inc.
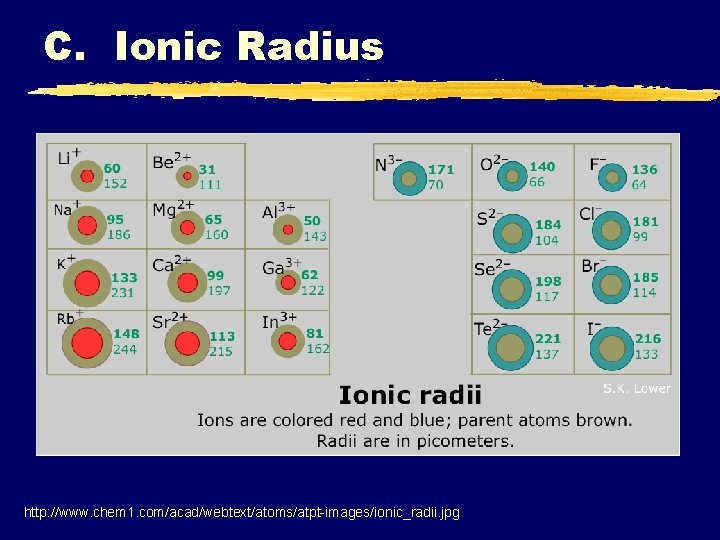
C. Ionic Radius http: //www. chem 1. com/acad/webtext/atoms/atpt-images/ionic_radii. jpg
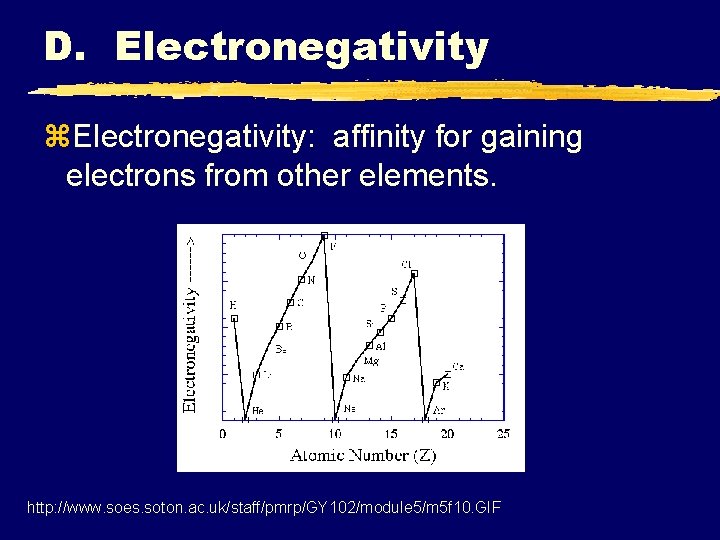
D. Electronegativity z. Electronegativity: affinity for gaining electrons from other elements. http: //www. soes. soton. ac. uk/staff/pmrp/GY 102/module 5/m 5 f 10. GIF

D. Electronegativity http: //online. uis. edu/spring 2000/che 367/m 1 -bonding/img 008. GIF

D. Electronegativity z. Why increase to the right? y. Closer to noble gas. z. Why decrease going down? y. Shielding
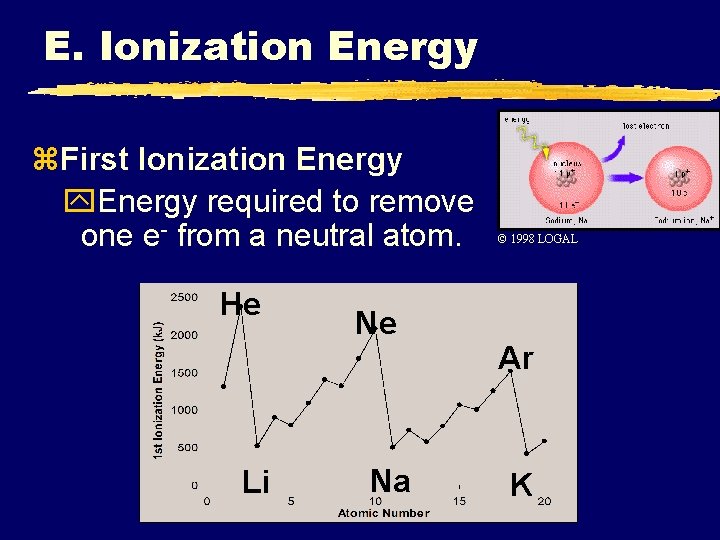
E. Ionization Energy z. First Ionization Energy y. Energy required to remove one e- from a neutral atom. He Li Ne Na C. Johannesson © 1998 LOGAL Ar K

E. Ionization Energy z. First Ionization Energy y. Increases UP and to the RIGHT C. Johannesson

E. Ionization Energy z. Why opposite of atomic radius? y. In small atoms, e- are close to the nucleus where the attraction is stronger z. Why small jumps within each group? y. Stable e- configurations don’t want to lose e. C. Johannesson

E. Ionization Energy z. Successive Ionization Energies y. Large jump in I. E. occurs when a CORE e- is removed. y. Mg Core e- 1 st I. E. 736 k. J 2 nd I. E. 1, 445 k. J 3 rd I. E. 7, 730 k. J C. Johannesson

E. Ionization Energy z. Successive Ionization Energies y. Large jump in I. E. occurs when a CORE e- is removed. y. Al Core e- 1 st I. E. 577 k. J 2 nd I. E. 1, 815 k. J 3 rd I. E. 2, 740 k. J 4 th I. E. 11, 600 k. J C. Johannesson

F. Electron Affinity z. Electron Affinity - The amount of energy required, or given off, when a neutral atom gains an electron. z. In a sense, it is the opposite of IE, since IE represents the energy change when an atom loses an electron. z. These values are usually negative (indicating energy is given off. )
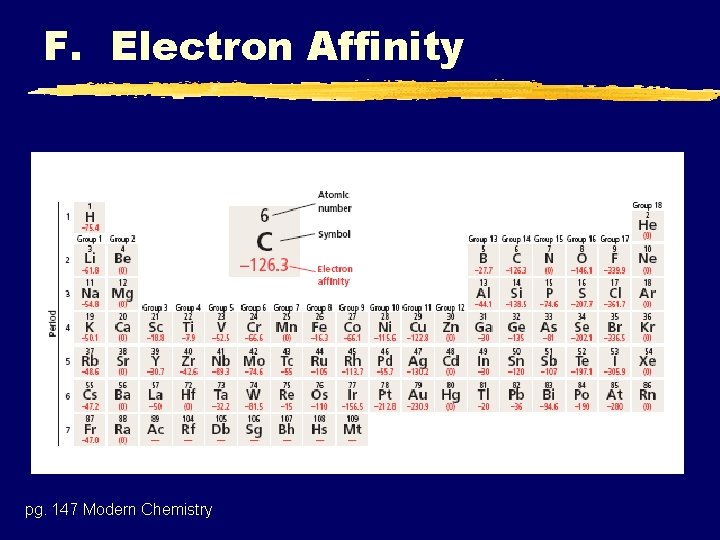
F. Electron Affinity pg. 147 Modern Chemistry

F. Electron Affinity z Why increase energy released (become more negative) to the right within blocks? y. Stability associated with full (and half full) valence shells. z. Group trends not regular due to competing affects. ynuclear charge y. Shielding (dominates)

G. Chemical Reactivity z. Chemical Reactivity refers to how likely or vigorously an atom is to react with other substances, usually determined by how easily electrons can be removed (ionization E) and how attracted they are to other atom’s electrons (electronegativity).
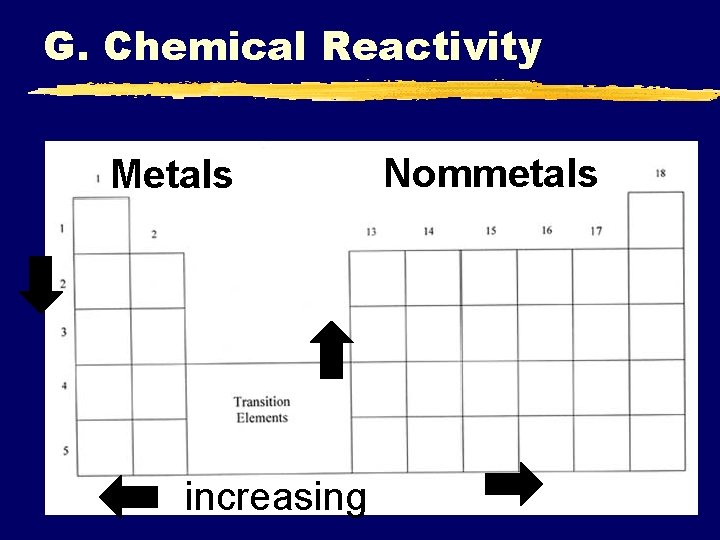
G. Chemical Reactivity Metals increasing Nommetals
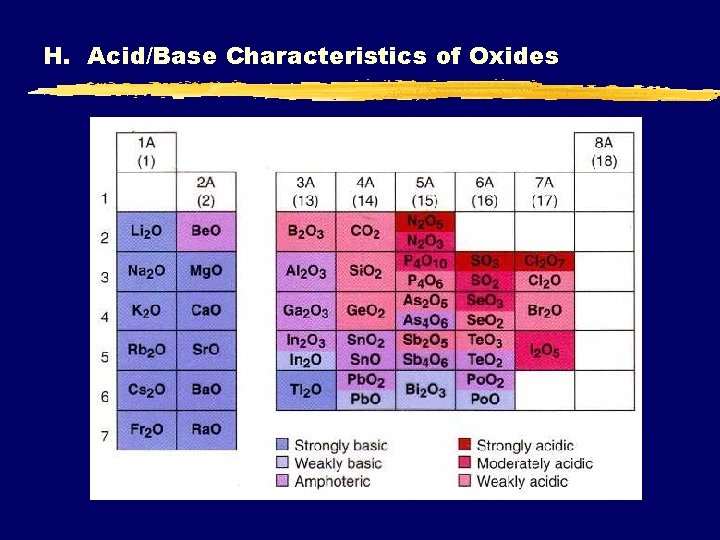
H. Acid/Base Characteristics of Oxides

F. Melting/Boiling Point z. Melting/Boiling Point y. Highest in the middle of a period. C. Johannesson
- Slides: 33