Ch 5 MICROBIAL METABOLISM 2013 Pearson Education Inc

Ch 5 MICROBIAL METABOLISM © 2013 Pearson Education, Inc.
© 2013 Pearson Education, Inc.

What is metabolism? ? § Metabolism: is the sum of the chemical reactions in an organism § There are two types of metabolic reactions Catabolism: the breakdown of complex organic compounds into smaller ones Releases energy for use during anabolism Anabolism: the building of complex organic compounds from simpler ones uses energy © 2013 Pearson Education, Inc.

§ A metabolic pathway is a sequence of enzymatically catalyzed chemical reactions in a cell § Metabolic pathways are determined by enzymes § Enzymes - Biological catalysts § Catalyst- substances (proteins) that can speed up a chemical reaction by lower the activation energy § Specific for a chemical reaction; not used up in that reaction © 2013 Pearson Education, Inc.
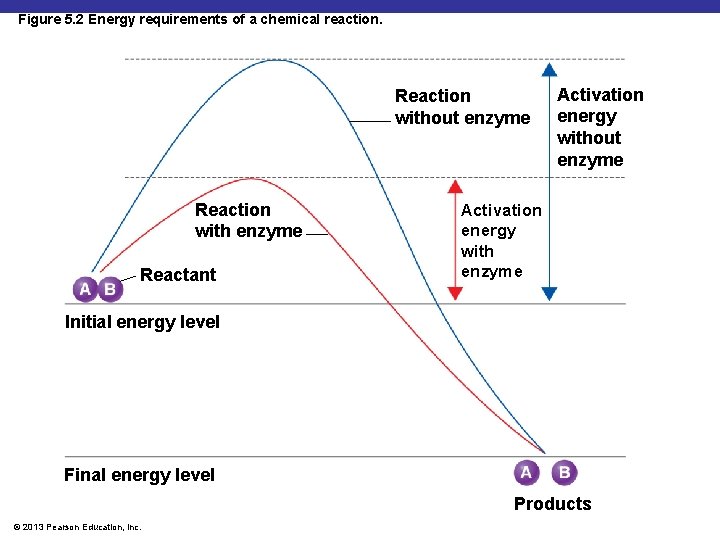
Figure 5. 2 Energy requirements of a chemical reaction. Reaction without enzyme Reaction with enzyme Reactant Activation energy without enzyme Activation energy with enzyme Initial energy level Final energy level Products © 2013 Pearson Education, Inc.

How enzymes work § Enzyme binds to a substrate in the active site forming the enzyme-substrate complex § Causes reaction to occur more quickly § Once product is formed enzyme is removed and ready to help in next reaction § Enzymes are very specific § Usually end in –ase Ex: Lactase © 2013 Pearson Education, Inc.
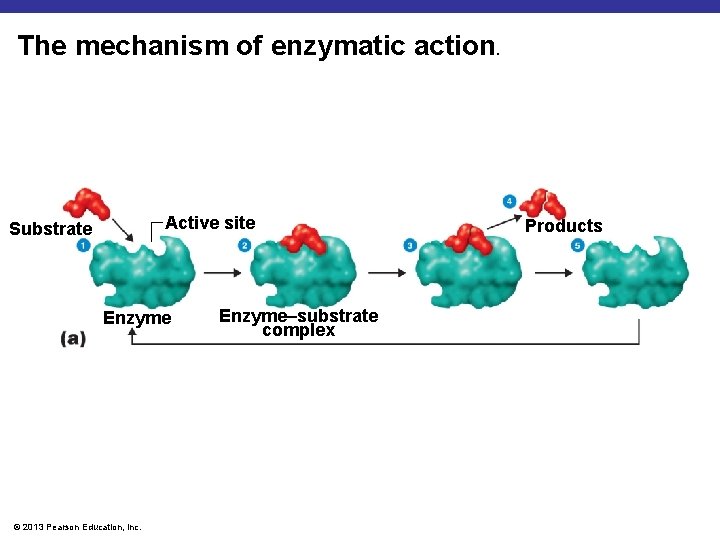
The mechanism of enzymatic action. Active site Substrate Enzyme © 2013 Pearson Education, Inc. Enzyme–substrate complex Products
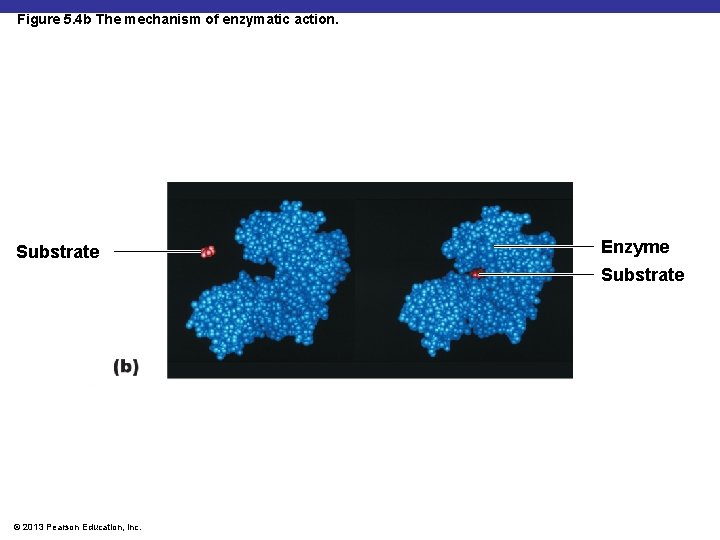
Figure 5. 4 b The mechanism of enzymatic action. Substrate Enzyme Substrate © 2013 Pearson Education, Inc.

Factors Influencing Enzyme Activity § § Temperature p. H Substrate concentration Inhibitors © 2013 Pearson Education, Inc.

Factors Influencing Enzyme Activity § Temperature and p. H denature proteins § Denature – changing the shape © 2013 Pearson Education, Inc.
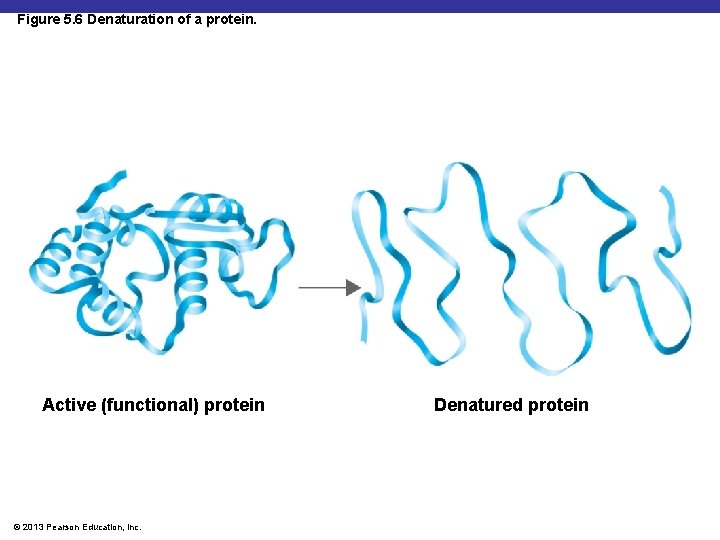
Figure 5. 6 Denaturation of a protein. Active (functional) protein © 2013 Pearson Education, Inc. Denatured protein
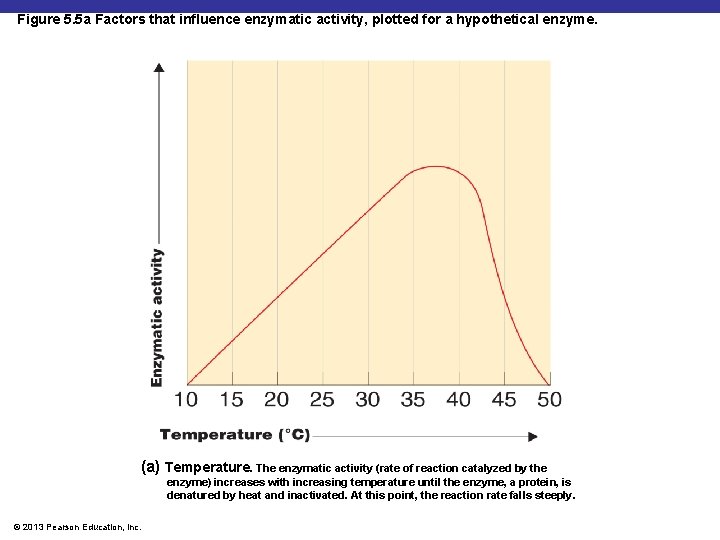
Figure 5. 5 a Factors that influence enzymatic activity, plotted for a hypothetical enzyme. (a) Temperature. The enzymatic activity (rate of reaction catalyzed by the enzyme) increases with increasing temperature until the enzyme, a protein, is denatured by heat and inactivated. At this point, the reaction rate falls steeply. © 2013 Pearson Education, Inc.
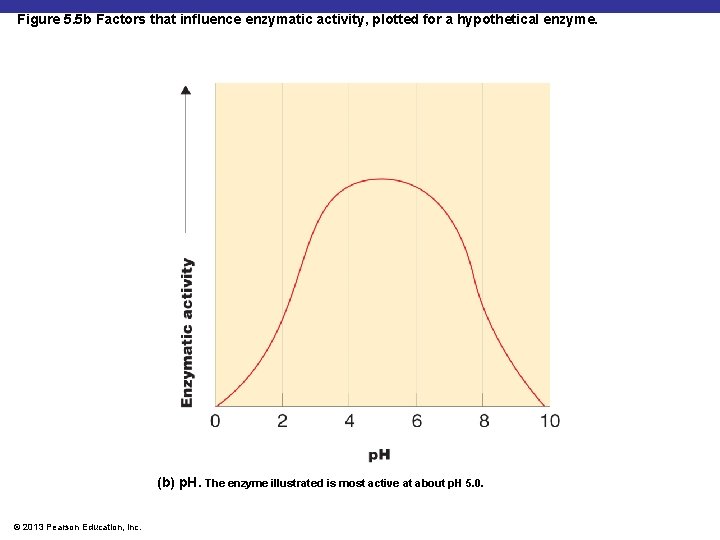
Figure 5. 5 b Factors that influence enzymatic activity, plotted for a hypothetical enzyme. (b) p. H. The enzyme illustrated is most active at about p. H 5. 0. © 2013 Pearson Education, Inc.
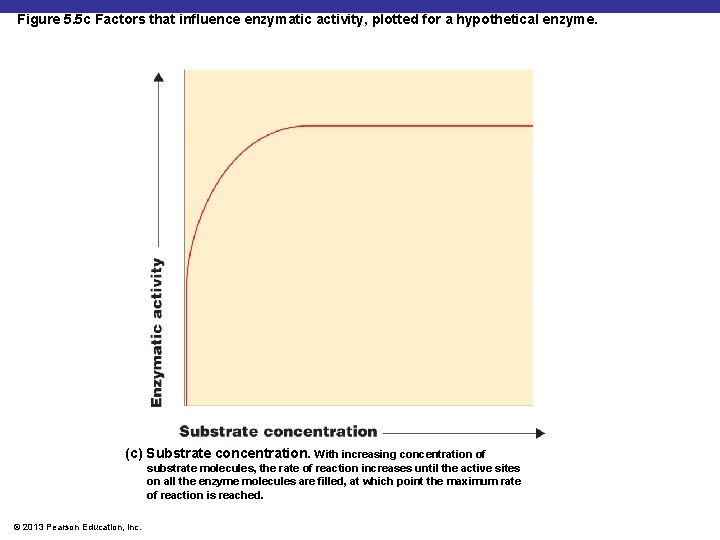
Figure 5. 5 c Factors that influence enzymatic activity, plotted for a hypothetical enzyme. (c) Substrate concentration. With increasing concentration of substrate molecules, the rate of reaction increases until the active sites on all the enzyme molecules are filled, at which point the maximum rate of reaction is reached. © 2013 Pearson Education, Inc.

Enzyme Inhibitors: Competitive Inhibition ANIMATION Enzymes: Competitive Inhibition © 2013 Pearson Education, Inc.

§ Competitive Inhibitors – substances that compete with a substrate to fill the active site of enzymes § Ex: Cyanide, arsenic, and mercury § Sulfa drugs – inhibits the enzyme from attaching to the PABA (an essential nutrient for a lot of Bacteria) © 2013 Pearson Education, Inc.
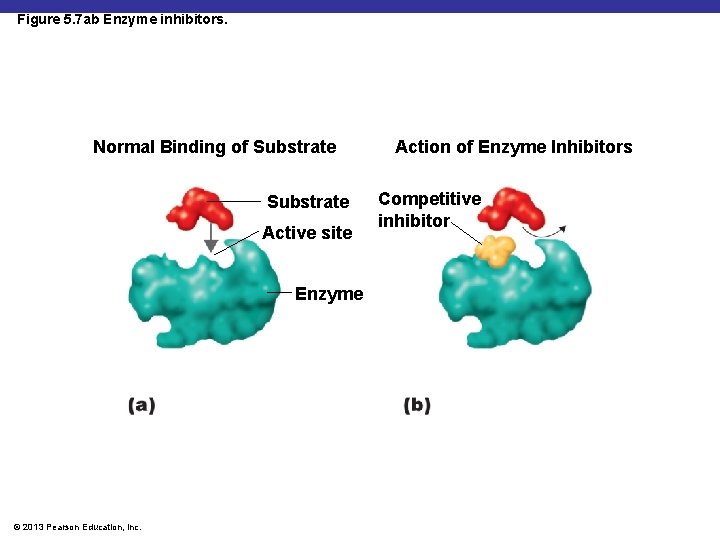
Figure 5. 7 ab Enzyme inhibitors. Normal Binding of Substrate Active site Enzyme © 2013 Pearson Education, Inc. Action of Enzyme Inhibitors Competitive inhibitor

Enzyme Inhibitors: Noncompetitive Inhibition ANIMATION Enzymes: Non-competitive Inhibition © 2013 Pearson Education, Inc.

Feedback inhibition § Inhibits a metabolic pathway by inhibiting a enzyme needed to make a product. § Usually the first enzyme is effected. © 2013 Pearson Education, Inc.
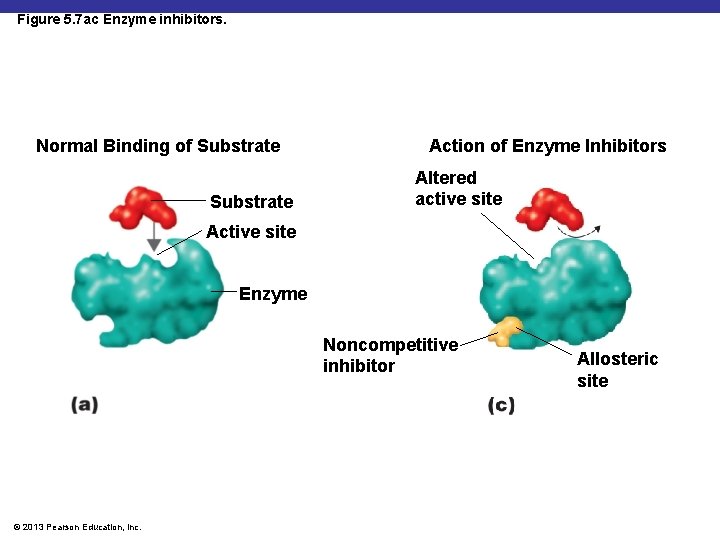
Figure 5. 7 ac Enzyme inhibitors. Normal Binding of Substrate Action of Enzyme Inhibitors Altered active site Active site Enzyme Noncompetitive inhibitor © 2013 Pearson Education, Inc. Allosteric site
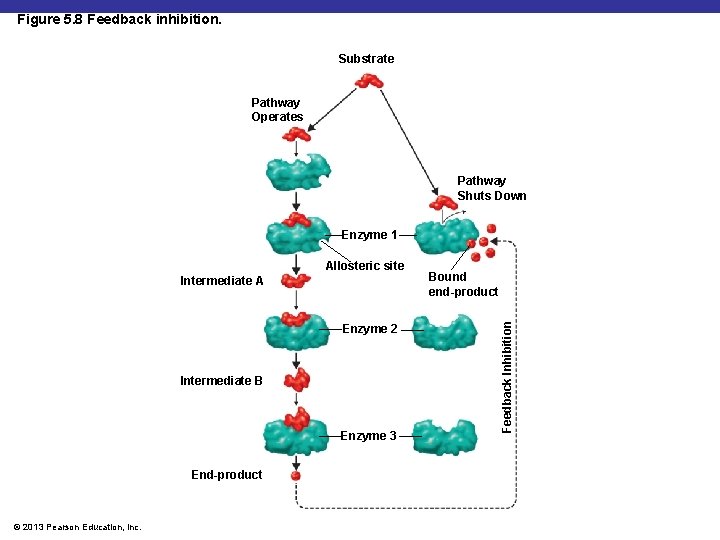
Figure 5. 8 Feedback inhibition. Substrate Pathway Operates Pathway Shuts Down Enzyme 1 Intermediate A Enzyme 2 Intermediate B Enzyme 3 End-product © 2013 Pearson Education, Inc. Bound end-product Feedback Inhibition Allosteric site

Ribozymes § RNA that cuts and splices RNA § (non protein enzyme) © 2013 Pearson Education, Inc.

Cellular respiration/Carbohydrate Catabolism § The breakdown of glucose into ATP § Cellular Respiration occurs in three steps Glycolysis (Anaerobic) Krebs cycle (Aerobic) Electron transport chain (Aerobic) © 2013 Pearson Education, Inc.

Glycolysis § The conversion of glucose into pyruvic acid. § End Products § Pyruvic acid § NADH § Net gain of 2 ATP © 2013 Pearson Education, Inc.

Glycolysis ANIMATION Glycolysis: Overview ANIMATION Glycolysis: Steps © 2013 Pearson Education, Inc.

Krebs Cycle § A series of reactions that release a large amount of potential energy to be transferred to the ETC. § End products 2 ATP NADH FADH § By products § Carbon Dioxide © 2013 Pearson Education, Inc.
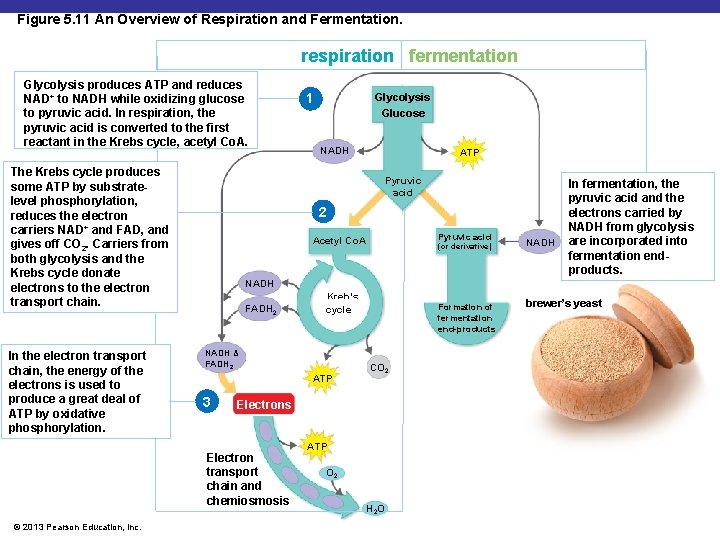
Figure 5. 11 An Overview of Respiration and Fermentation. respiration fermentation Glycolysis produces ATP and reduces NAD+ to NADH while oxidizing glucose to pyruvic acid. In respiration, the pyruvic acid is converted to the first reactant in the Krebs cycle, acetyl Co. A. The Krebs cycle produces some ATP by substratelevel phosphorylation, reduces the electron carriers NAD+ and FAD, and gives off CO 2. Carriers from both glycolysis and the Krebs cycle donate electrons to the electron transport chain. In the electron transport chain, the energy of the electrons is used to produce a great deal of ATP by oxidative phosphorylation. Glycolysis Glucose NADH ATP Pyruvic acid 2 Pyruvic acid Acetyl Co. A (or derivative) NADH In fermentation, the pyruvic acid and the electrons carried by NADH from glycolysis are incorporated into fermentation endproducts. NADH FADH 2 Kreb’s cycle NADH & FADH 2 ATP 3 Formation of fermentation end-products CO 2 Electrons Electron transport chain and chemiosmosis © 2013 Pearson Education, Inc. 1 ATP O 2 H 2 O brewer’s yeast

The Krebs Cycle ANIMATION Krebs Cycle: Overview ANIMATION Krebs Cycle: Steps © 2013 Pearson Education, Inc.
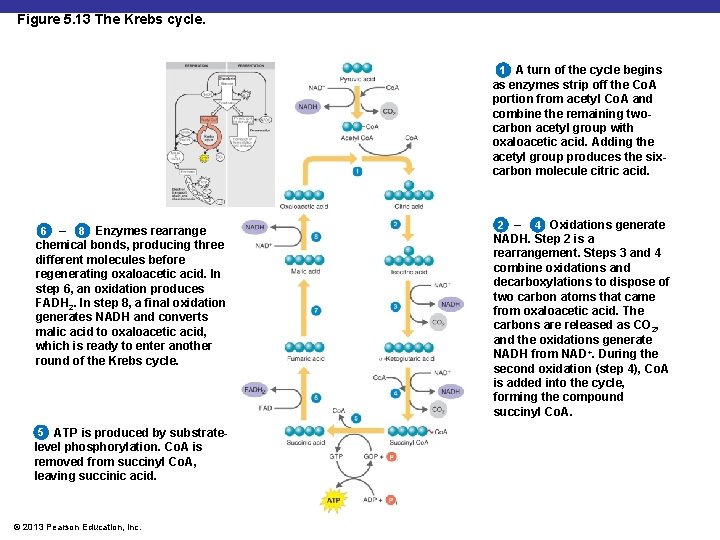
Figure 5. 13 The Krebs cycle. 1 A turn of the cycle begins as enzymes strip off the Co. A portion from acetyl Co. A and combine the remaining twocarbon acetyl group with oxaloacetic acid. Adding the acetyl group produces the sixcarbon molecule citric acid. – 8 Enzymes rearrange chemical bonds, producing three different molecules before regenerating oxaloacetic acid. In step 6, an oxidation produces FADH 2. In step 8, a final oxidation generates NADH and converts malic acid to oxaloacetic acid, which is ready to enter another round of the Krebs cycle. 6 5 ATP is produced by substratelevel phosphorylation. Co. A is removed from succinyl Co. A, leaving succinic acid. © 2013 Pearson Education, Inc. 2 – 4 Oxidations generate NADH. Step 2 is a rearrangement. Steps 3 and 4 combine oxidations and decarboxylations to dispose of two carbon atoms that came from oxaloacetic acid. The carbons are released as CO 2, and the oxidations generate NADH from NAD+. During the second oxidation (step 4), Co. A is added into the cycle, forming the compound succinyl Co. A.

The Electron Transport Chain § A series of reactions where the release of energy is used to produce ATP § 34 ATP produced ANIMATION Electron Transport Chain: Overview © 2013 Pearson Education, Inc.

Total energy production from Cellular Respiration § Glycolysis – 2 ATP § Krebs Cycle – 2 ATP § ETC – 34 ATP § TOTAL = 38 ATP © 2013 Pearson Education, Inc.
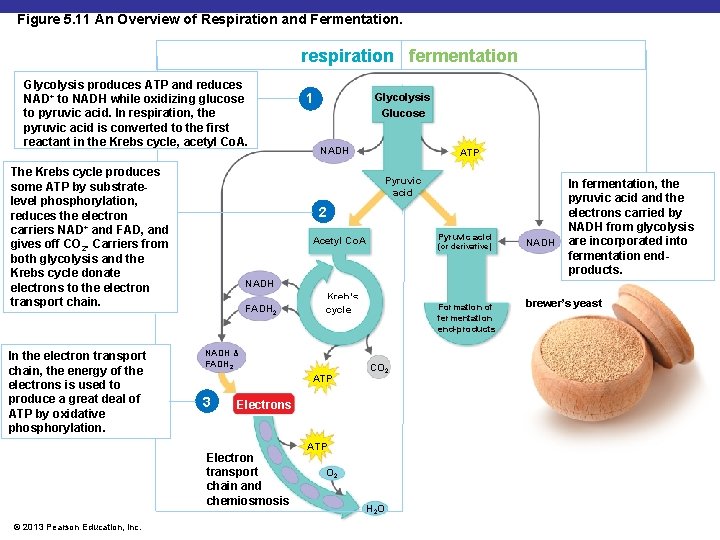
Figure 5. 11 An Overview of Respiration and Fermentation. respiration fermentation Glycolysis produces ATP and reduces NAD+ to NADH while oxidizing glucose to pyruvic acid. In respiration, the pyruvic acid is converted to the first reactant in the Krebs cycle, acetyl Co. A. The Krebs cycle produces some ATP by substratelevel phosphorylation, reduces the electron carriers NAD+ and FAD, and gives off CO 2. Carriers from both glycolysis and the Krebs cycle donate electrons to the electron transport chain. In the electron transport chain, the energy of the electrons is used to produce a great deal of ATP by oxidative phosphorylation. Glycolysis Glucose NADH ATP Pyruvic acid 2 Pyruvic acid Acetyl Co. A (or derivative) NADH In fermentation, the pyruvic acid and the electrons carried by NADH from glycolysis are incorporated into fermentation endproducts. NADH FADH 2 Kreb’s cycle NADH & FADH 2 ATP 3 Formation of fermentation end-products CO 2 Electrons Electron transport chain and chemiosmosis © 2013 Pearson Education, Inc. 1 ATP O 2 H 2 O brewer’s yeast

A Summary of Respiration § Aerobic respiration: the final electron acceptor in the electron transport chain is molecular oxygen (O 2) § Anaerobic respiration: the final electron acceptor in the electron transport chain is NOT O 2 § Yields less energy than aerobic respiration because only part of the Krebs cycle operates under anaerobic conditions © 2013 Pearson Education, Inc.

Respiration ANIMATION Electron Transport Chain: Factors Affecting ATP Yield © 2013 Pearson Education, Inc.

Carbohydrate Catabolism Pathway Eukaryote Prokaryote Glycolysis Cytoplasm Intermediate step Cytoplasm Krebs cycle Mitochondrial matrix Cytoplasm ETC Mitochondrial inner membrane Plasma membrane © 2013 Pearson Education, Inc.

Fermentation § Any spoilage of food by microorganisms (general use) § Any process that produces alcoholic beverages or acidic dairy products (general use) § Any large-scale microbial process occurring with or without air (common definition used in industry) © 2013 Pearson Education, Inc.

Fermentation § Scientific definition: § Does not require oxygen § Does not use the Krebs cycle or ETC § Produces only small amounts of ATP © 2013 Pearson Education, Inc.
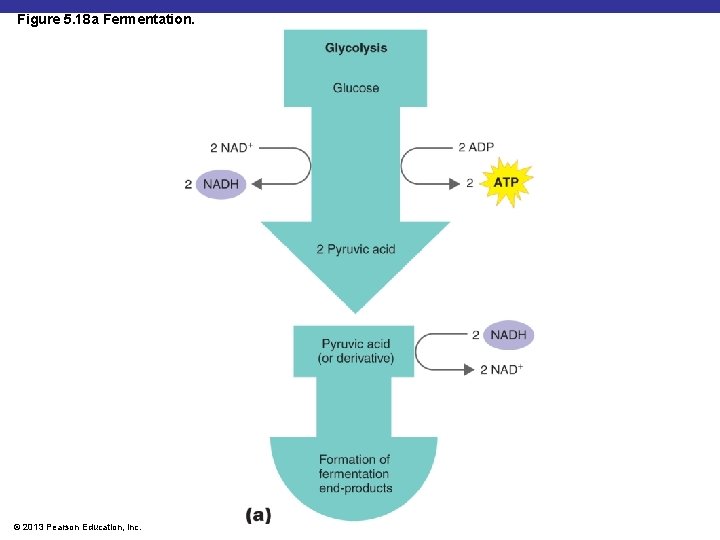
Figure 5. 18 a Fermentation. © 2013 Pearson Education, Inc.
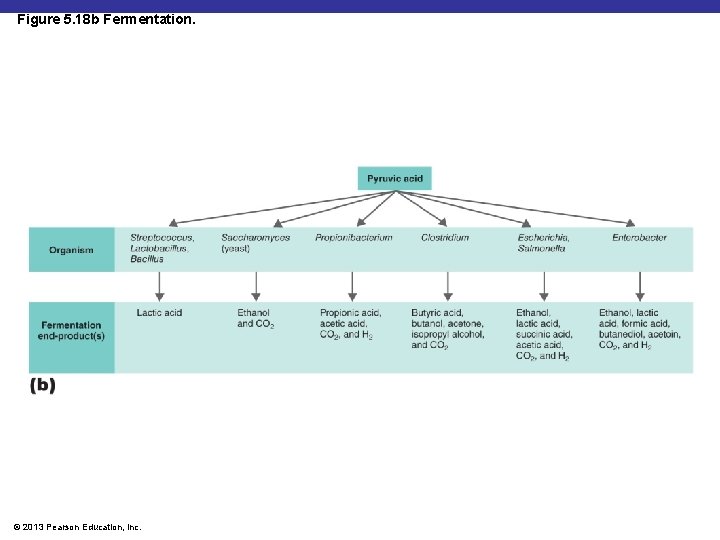
Figure 5. 18 b Fermentation. © 2013 Pearson Education, Inc.

Fermentation § Alcohol fermentation: produces ethanol + CO 2 § Lactic acid fermentation: produces lactic acid § Homolactic fermentation: produces lactic acid only § Heterolactic fermentation: produces lactic acid and other compounds © 2013 Pearson Education, Inc.
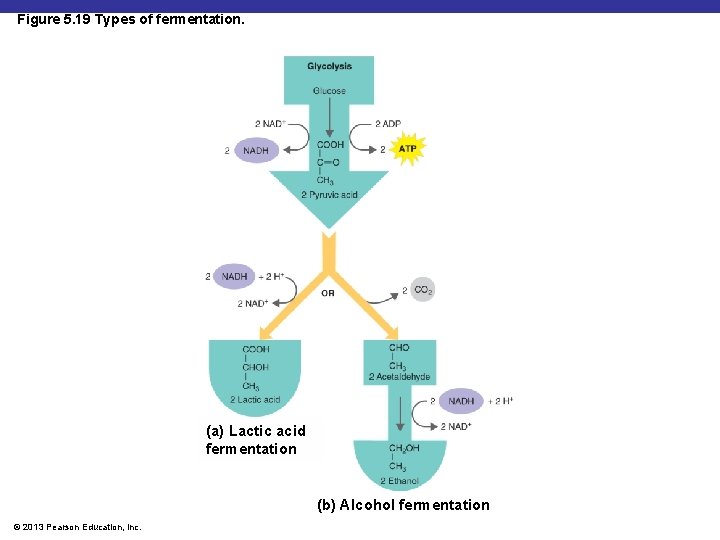
Figure 5. 19 Types of fermentation. (a) Lactic acid fermentation (b) Alcohol fermentation © 2013 Pearson Education, Inc.
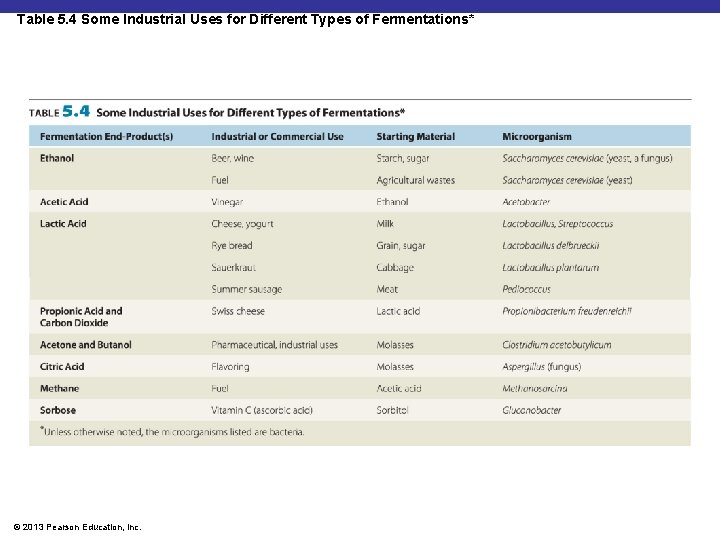
Table 5. 4 Some Industrial Uses for Different Types of Fermentations* © 2013 Pearson Education, Inc.

Biochemical Tests § Used to identify bacteria based on their ability to breakdown certain substances. § Ex: Urease test- In a positive test, bacterial urease hydrolyzes urea, this produces ammonia. Ammonia raises the ph. The media has a ph indicator in it and when the ph increases it turns the media pink. © 2013 Pearson Education, Inc.

Clinical Focus: Human Tuberculosis – Dallas, Texas Figure B The urease test. © 2013 Pearson Education, Inc.
- Slides: 44