Ch 5 Atoms Molecule Formulas Subatomic Particles Atoms

Ch # 5 Atoms, Molecule, Formulas, Subatomic Particles.

Atoms § An atom is the smallest particle of an element that can exist and still have properties of the element.

Atomic Theory of Matter § All matter is made up of small particles called atoms. 113 types. § Atoms of same element are similar to one another. § The relative number and arrangement of different types of atoms contained in a pure substance determined its identity.

Atomic Theory of Atoms. § These atoms rearrange, separate or unite to form a new substance. § Atoms can participate in or result from any chemical change, atoms cannot be destructed.

An atom is very Small

The diameter of an atom is 0. 1 to 0. 5 nm. This is 1 to 5 ten billionths of a meter. If the diameter of this dot is 1 mm, then 10 million hydrogen atoms would form a line across the dot.

The Molecule § A group of two or more atoms that is strongly bound to each other.

The Molecule § Diatomic molecule: contains 2 atoms. H, N, O, F, Cl, Br, I, § Triatomic molecule: Contains 3 atoms. § P-4 atoms, S-8 atoms. § Homoatomic molecule: All atoms are of same kind. § Heteroatomic molecule: Two or more different kinds of atoms are present.



Molecular compounds § Compounds that have heteroatomic molecule as basic structural unit. § Ex: table sugar-sucrose. § Molecule is a limit of physical subdivision. Atom is a limit of chemical sub-division.

Molecular Compounds § Two kinds of atoms present, but, one kind of molecule. § Molecules have different properties than that of the atom.

Ionic compounds § Compounds that contain ions. Ex: Na. Cl



Natural and Synthetic compounds § The compounds could be naturally or artificially made.

Chemical Formulas § Made up of symbols. § The subscripts indicate the number of atoms. § Read the formula C 6 H 12 O 6

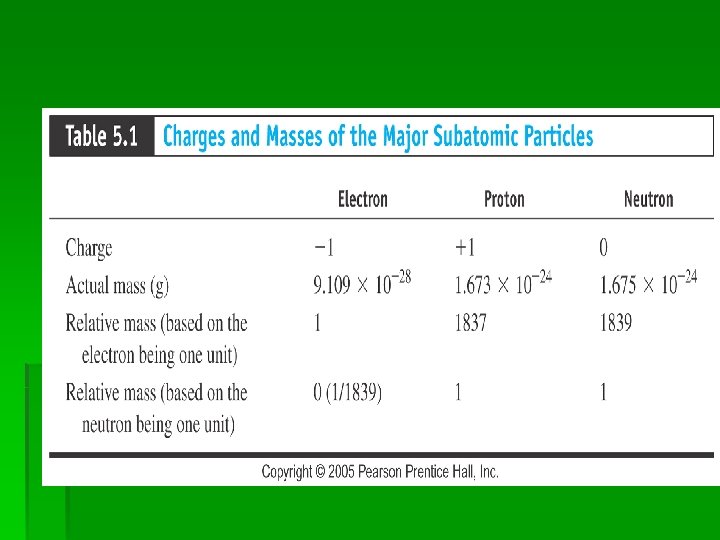
Protons

• Eugen Goldstein, a German physicist, first observed protons in 1886: § Thompson determined the proton’s characteristics. § Thompson showed that atoms contained both positive and negative charges. § This disproved the Dalton model of the atom which held that atoms were indivisible.

Subatomic Particles. § Protons: +ve charge. -Goldstein. Charge: +1 § Electron: -ve charge. - Thomson. Smallest mass. Charge: -1 § Neutron: No charge. -Chadwick. § Opposite charges attract, like charges repel.

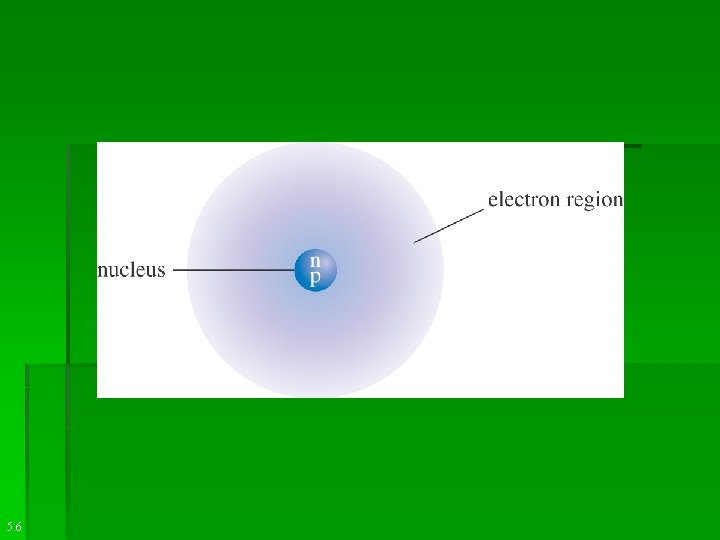
Nucleus: § Small, dense, positively charged center of an atom. It contains protons and neutrons. An atoms entire mass is concentrated to the center.

Nucleon § Any subatomic particle found in the nucleus of an atom. Ex; protons and neutrons. § Electrons revolve around the nucleus in a very large region. It is mostly empty space. § Volume occupied by electrons is called electron cloud. It is negatively charged.

An atom as a whole is neutral. Number of protons=Number of electrons in any atom. The +ve and-ve charges cancel each other out.

Diameter of nucleus= 10 -15 m. Entire mass is concentrated in the center of the atom.

Protons and neutrons are made of Leptons, Mesons, Baryons.

5. 6

Atomic Numbers of the Elements

• The atomic number of an element is equal to the number of protons in the nucleus of that element. § The atomic number of an atom determines which element the atom is.

atomic number of 1 is a hydrogen atom. Every hydrogen atom contains 1 proton in its nucleus.

atomic number Every atom with an atomic number of 6 is a carbon atom. 6 protons in the nucleus C 6

atomic number Every atom with an atomic number of 1 is a hydrogen atom. 1 proton in the nucleus H 1
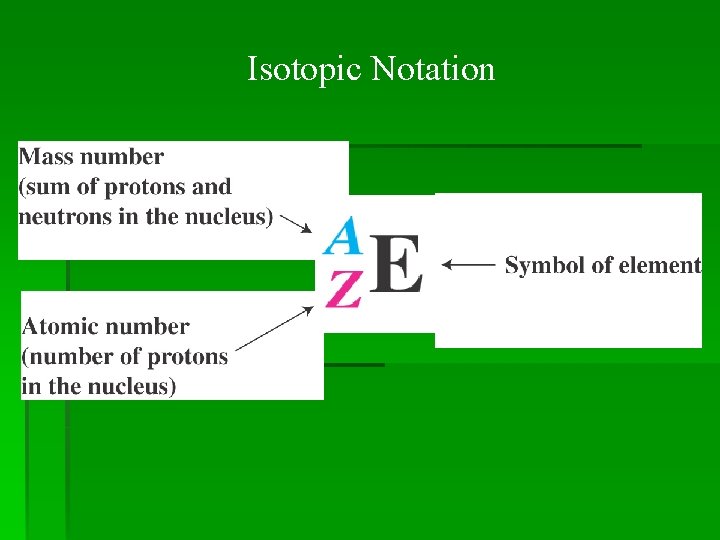
Isotopic Notation

Isotopes of the Elements

• Atoms of the same element can have different masses. § They always have the same number of protons, but they can have different numbers of neutrons in their nuclei. § The difference in the number of neutrons accounts for the difference in mass. § These are isotopes of the same

Isotopes of the Same Element Have Equal numbers of protons Different numbers of neutrons

Hydrogen has three isotopes 1 proton 0 neutrons 1 neutron 2 neutrons
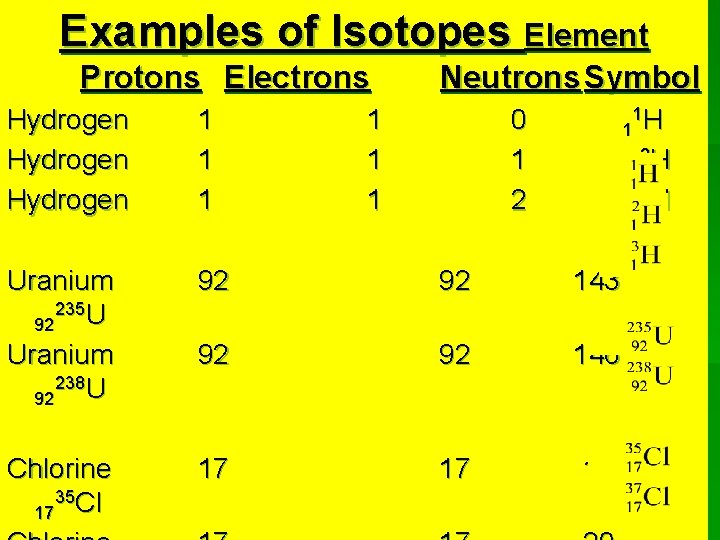
Examples of Isotopes Element Protons Electrons Neutrons Symbol Hydrogen 1 1 1 0 1 2 1 1 H 2 H 1 3 H 1 Uranium 235 U 92 Uranium 238 U 92 92 92 143 92 92 146 Chlorine 35 Cl 17 17 17 18

Atomic Mass

• The mass of a single atom is too small to measure on a balance. § Using a mass spectrometer, the mass of the hydrogen atom was determined.

Using a mass spectrometer, the mass of one hydrogen atom was determined to be 1. 673 x 10 -24 g.

This number is very small small small small small small
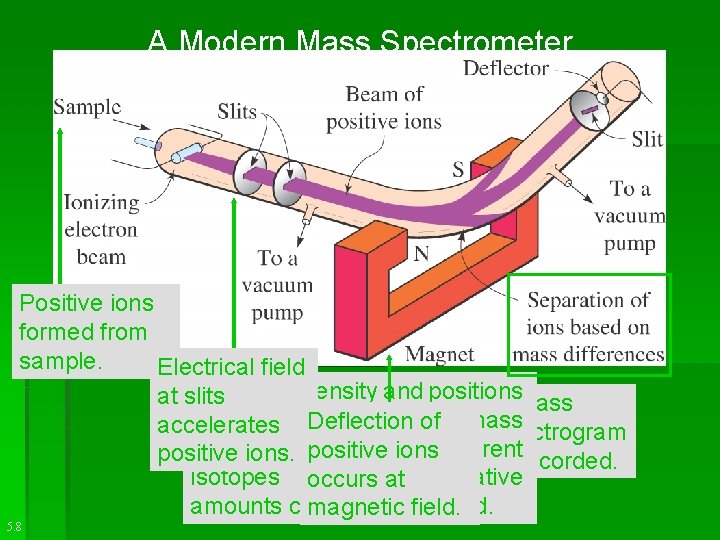
A Modern Mass Spectrometer Positive ions formed from sample. Electrical field From the intensity and positions at slits A mass of the lines on the Deflection of mass accelerates spectrogram, theionsdifferent positive ions. positive is recorded. isotopes and relative occurstheir at amounts can be determined. magnetic field. 5. 8

The standard to which the masses of all other atoms are compared to was chosen to be the most abundant isotope of carbon.
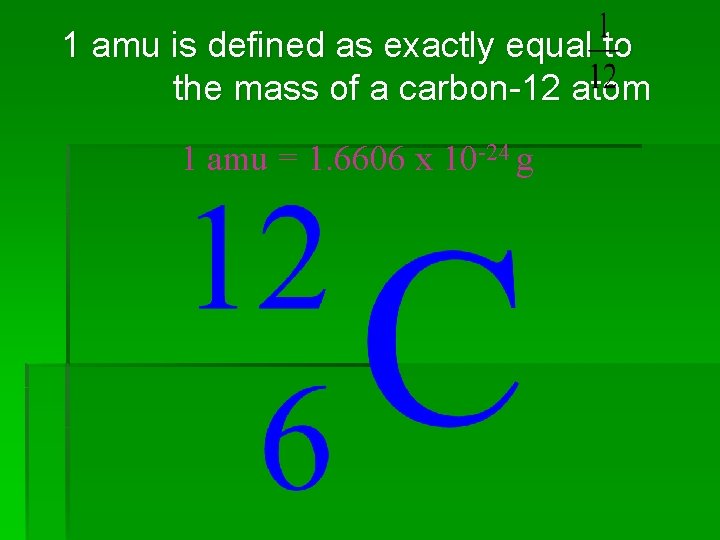
1 amu is defined as exactly equal to the mass of a carbon-12 atom 1 amu = 1. 6606 x 10 -24 g

Average atomic mass 1. 00797 amu.

Average atomic mass 248. 029 amu.

Average Relative Atomic Mass

• Most elements occur as mixtures of isotopes. § Isotopes of the same element have different masses. § The listed atomic mass of an element is the average relative mass of the isotopes of that element compared to the mass of carbon-12 (exactly 12. 0000…amu).

Relationship Between Mass Number and Atomic Number

The mass number minus the atomic number equals the number of neutrons in the nucleus. mass number atomic mass number 109 47 = = number of neutrons 62

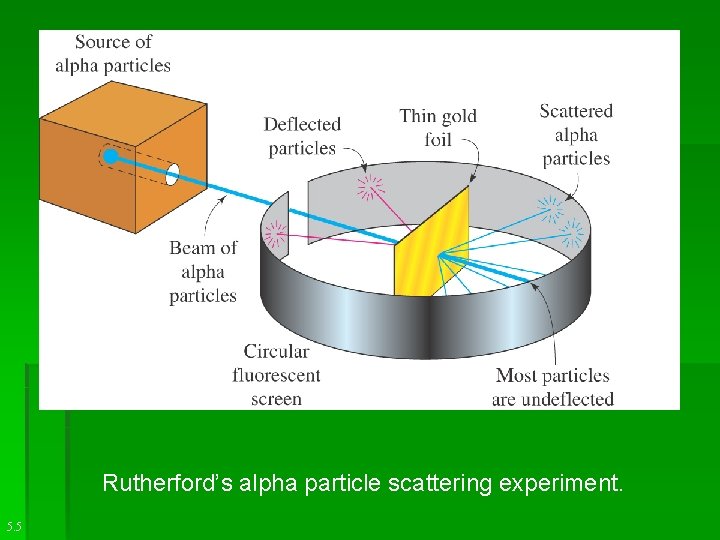
Rutherford’s alpha particle scattering experiment. 5. 5

- Slides: 55