CH 4 Acids and Bases 1 BronstedLowry definitions

CH 4. Acids and Bases 1
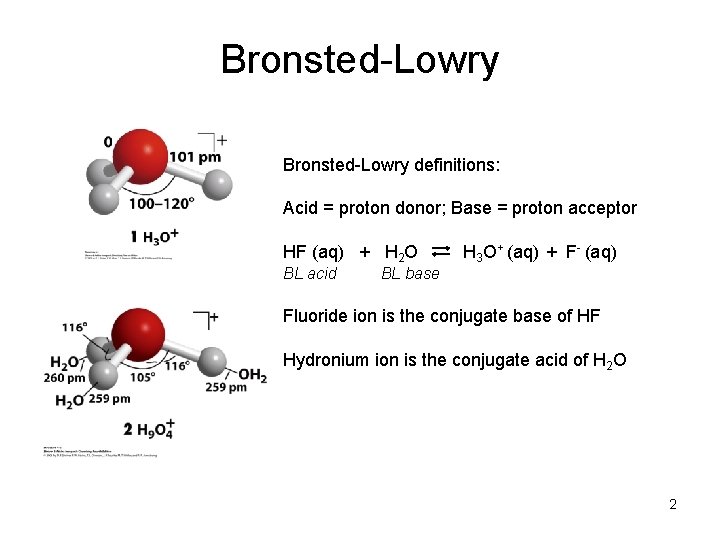
Bronsted-Lowry definitions: Acid = proton donor; Base = proton acceptor HF (aq) + H 2 O BL acid H 3 O+ (aq) + F- (aq) BL base Fluoride ion is the conjugate base of HF Hydronium ion is the conjugate acid of H 2 O 2

Amphiprotic species Amphiprotic – species that can act as BL acid or base NH 3 (aq) + BL base H 2 O BL acid NH 4+ (aqu) + OH (aqu) hydroxide Kb = base dissociation constant = [NH 4+] [OH ] / [NH 3] H 2 O is amphiprotic - it’s a base with HF, but an acid with NH 3 3

BL acid/base strength Ka, the acidity constant, measures acid strength as: Ka = [H 3 O+] [A-] / [HA] p. Ka = - log Ka When p. H = p. Ka, then [HA] = [A-] For strong acids p. Ka < 0 p. Ka(HCl) ≈ -7 4

BL acid/base strengths 5

Kw Kw = water autodissociation (autoionization) constant 2 H 2 O H 3 O+ (aqu) + OH- (aqu) Kw = [H 3 O+] [OH-] = 1 x 10 -14 (at 25°C) Using the above, you should prove that for any conjugate acid-base pair: p. Ka + p. Kb = p. Kw = 14 6
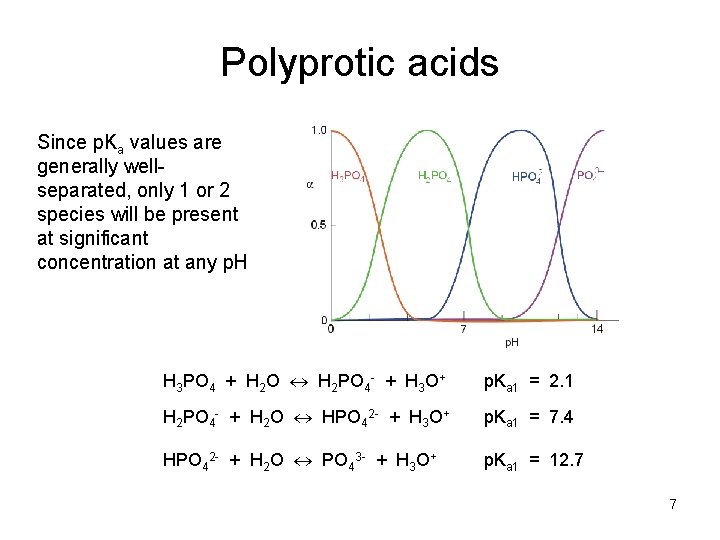
Polyprotic acids Since p. Ka values are generally wellseparated, only 1 or 2 species will be present at significant concentration at any p. H H 3 PO 4 + H 2 O H 2 PO 4 - + H 3 O+ p. Ka 1 = 2. 1 H 2 PO 4 - + H 2 O HPO 42 - + H 3 O+ p. Ka 1 = 7. 4 HPO 42 - + H 2 O PO 43 - + H 3 O+ p. Ka 1 = 12. 7 7

Solvent leveling The strongest acid possible in aqueous solution is H 3 O+ Ex: HCl + H 2 O H 3 O+ (aq) + Cl- (aq) there is no appreciable equilibrium, this reaction goes quantitatively; the acid form of HCl does not exist in aqueous solution Ex: KNH 2 + H 2 O K+ (aq) + OH- (aq) + NH 3 (aq) this is solvent leveling, the stable acid and base species are the BL acid-base pair of the solvent NH 2 - = imide anion NR 2 - , some substituted imide ions are less basic and can exist in aq soln 8
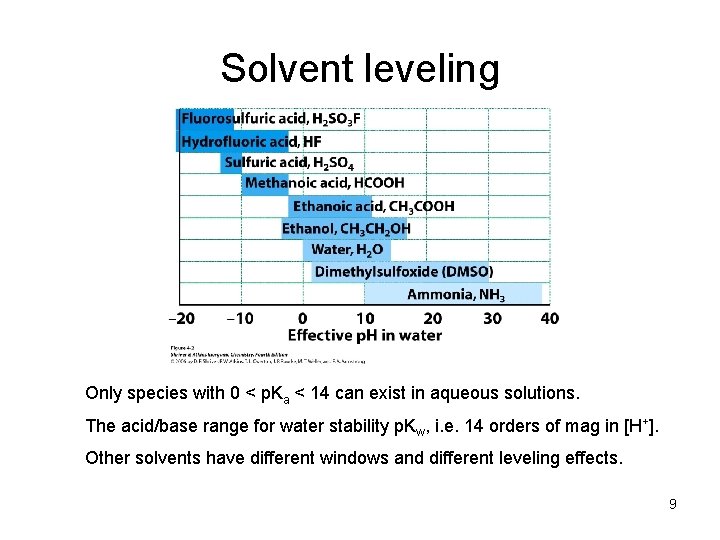
Solvent leveling Only species with 0 < p. Ka < 14 can exist in aqueous solutions. The acid/base range for water stability p. Kw, i. e. 14 orders of mag in [H+]. Other solvents have different windows and different leveling effects. 9
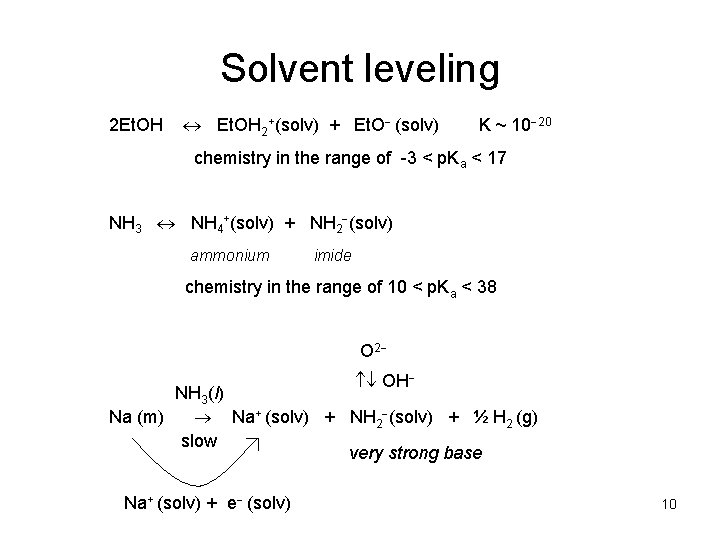
Solvent leveling 2 Et. OH 2+(solv) + Et. O (solv) K ~ 10 20 chemistry in the range of -3 < p. Ka < 17 NH 3 NH 4+(solv) + NH 2 (solv) ammonium imide chemistry in the range of 10 < p. Ka < 38 O 2 OH NH 3(l) Na (m) Na+ (solv) + NH 2 (solv) + ½ H 2 (g) slow very strong base Na+ (solv) + e (solv) 10
![Acid/base chemistry of complexes Aqueous chemistry: H 2 O Fe(NO 3)3 [Fe(OH 2)6]3+(aq) + Acid/base chemistry of complexes Aqueous chemistry: H 2 O Fe(NO 3)3 [Fe(OH 2)6]3+(aq) +](http://slidetodoc.com/presentation_image/45a4414909e3de4f67aab23e3e91634b/image-11.jpg)
Acid/base chemistry of complexes Aqueous chemistry: H 2 O Fe(NO 3)3 [Fe(OH 2)6]3+(aq) + 3 NO 3 (aq) 2 [Fe(OH 2)6]3+ (aq) Hexaaquairon(III), p. Ka ~ 3 [Fe 2(OH 2)10 OH]5+ (aq) + H 3 O+(aq) dimer 11
![Aqua, hydroxo, oxoacids aqua acid M(OH 2)xn+ ex: [Cu(OH 2)6]2+ hydroxoacid M(OH)x ex: B(OH)3 Aqua, hydroxo, oxoacids aqua acid M(OH 2)xn+ ex: [Cu(OH 2)6]2+ hydroxoacid M(OH)x ex: B(OH)3](http://slidetodoc.com/presentation_image/45a4414909e3de4f67aab23e3e91634b/image-12.jpg)
Aqua, hydroxo, oxoacids aqua acid M(OH 2)xn+ ex: [Cu(OH 2)6]2+ hydroxoacid M(OH)x ex: B(OH)3 , Si(OH)4 p. Ka ~ 10 oxoacid MOp(OH)q p and q designate oxo and hydroxo ligands hexaaquacopper(II) cation ex: H 2 CO 3 (aq) + H 2 O HCO 3 (aq) + H 3 O+(aq) carbonic acid CO 2 (g) + H 2 O bicarbonate p. K a ~ 3. 6 12
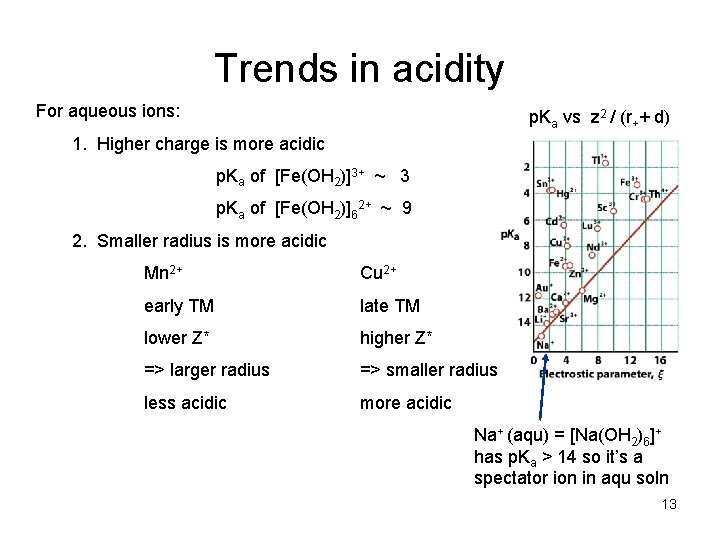
Trends in acidity For aqueous ions: p. Ka vs z 2 / (r++ d) 1. Higher charge is more acidic p. Ka of [Fe(OH 2)]3+ ~ 3 p. Ka of [Fe(OH 2)]62+ ~ 9 2. Smaller radius is more acidic Mn 2+ Cu 2+ early TM late TM lower Z* higher Z* => larger radius => smaller radius less acidic more acidic Na+ (aqu) = [Na(OH 2)6]+ has p. Ka > 14 so it’s a spectator ion in aqu soln 13

Anhydrides Ex: H 2 O + SO 3 anhydride H 2 SO 4 acid form Acidic SO 3 / H 2 SO 4 “P 2 O 5” / H 3 PO 4 CO 2/H 2 CO 3 Basic Na 2 O / Na. OH Amphoteric Al 2 O 3 / Al(OH)3 14
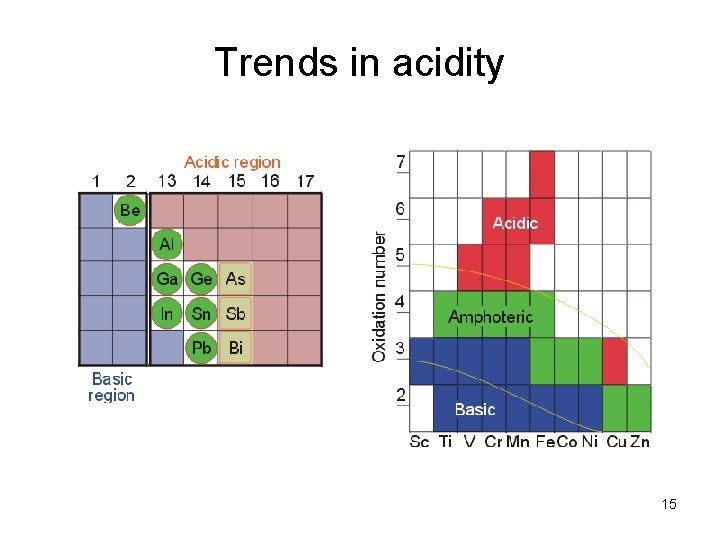
Trends in acidity 15

Common acids HNO 3 (D 3 h) Nitric acid Nitrate HNO 2 (C 2 v) Nitrous acid Nitrite H 3 PO 43 (Td) Phosphoric acid Phosphate H 3 PO 3 HPO 32 (C 3 v) Phosphorous acid Phosphite You should know these! 16

Common acids H 2 SO 4 (Td) SO 42 Sulfuric acid Sulfate H 2 SO 3 (C 3 v) SO 32 Sulfurous acid Sulfite You should know these! 17

Common acids HCl. O 4 (Td) Perchloric acid Perchlorate HCl. O 3 (C 3 v) Chloric acid Chlorate HCl. O 2 (C 2 v) Chlorous acid Chlorite HOCl Hypochlorous acid Hypochlorite You should know these! 18

Pauling’s rules for p. Ka‘s of oxoacids 1. Write formula as MOp(OH)q 2. p. Ka 8 – 5 p 3. Each succeeding deprotonation increases the p. Ka by 5 Ex: rewrite HNO 3 as NO 2(OH) p = 2; p. Ka 8 – 5(2) 2 (exptl value is 1. 4) Ex: rewrite H 3 PO 4 as PO(OH)3 p = 1; p. Ka 1 8 – 5(1) 3 (exptl value is 2. 1) p. Ka 2 8 (exptl value is 7. 4) p. Ka 3 13 (exptl value is 12. 7) 19
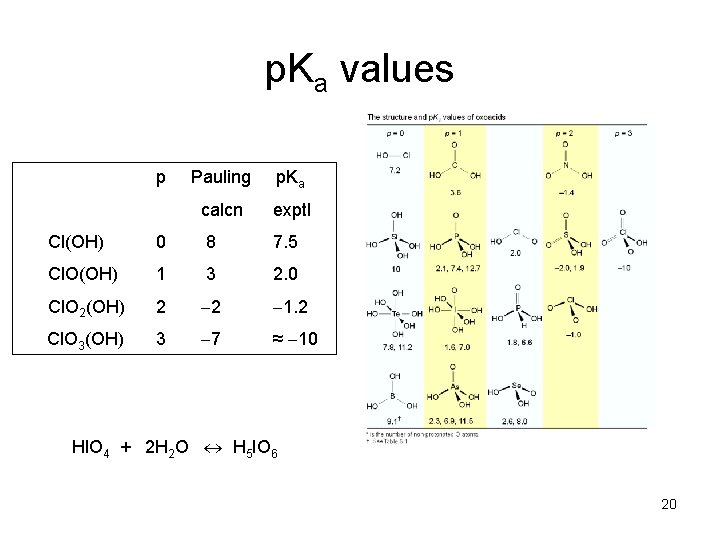
p. Ka values p Pauling p. Ka calcn exptl Cl(OH) 0 8 7. 5 Cl. O(OH) 1 3 2. 0 Cl. O 2(OH) 2 2 1. 2 Cl. O 3(OH) 3 7 ≈ 10 Hl. O 4 + 2 H 2 O H 5 IO 6 20
![Amphoteric oxides [Al(OH 2)6]3+ Oh Al 2 O 3 / Al(OH)3 [Al(OH)4] H 3 Amphoteric oxides [Al(OH 2)6]3+ Oh Al 2 O 3 / Al(OH)3 [Al(OH)4] H 3](http://slidetodoc.com/presentation_image/45a4414909e3de4f67aab23e3e91634b/image-21.jpg)
Amphoteric oxides [Al(OH 2)6]3+ Oh Al 2 O 3 / Al(OH)3 [Al(OH)4] H 3 O + OH Td 2 [Al(OH 2)6]3+(aq) p. Ka ~ 2 [Al 2(OH 2)10(OH)]5+(aq) + H 3 O+(aq) dimer 21
![polyoxocations linear trimer is [Al 3(OH 2)14(OH)2]7+ Keggin ion [Al. O 4(Al(OH)2)12]7+ p. H polyoxocations linear trimer is [Al 3(OH 2)14(OH)2]7+ Keggin ion [Al. O 4(Al(OH)2)12]7+ p. H](http://slidetodoc.com/presentation_image/45a4414909e3de4f67aab23e3e91634b/image-22.jpg)
polyoxocations linear trimer is [Al 3(OH 2)14(OH)2]7+ Keggin ion [Al. O 4(Al(OH)2)12]7+ p. H ≈ 4 charge/volume ratios Al(OH 2)63+ 3+ / Oh > dimer 5+ / 2 Oh > trimer 7+ / 3 Oh --- > Al(OH)3 neutral 22
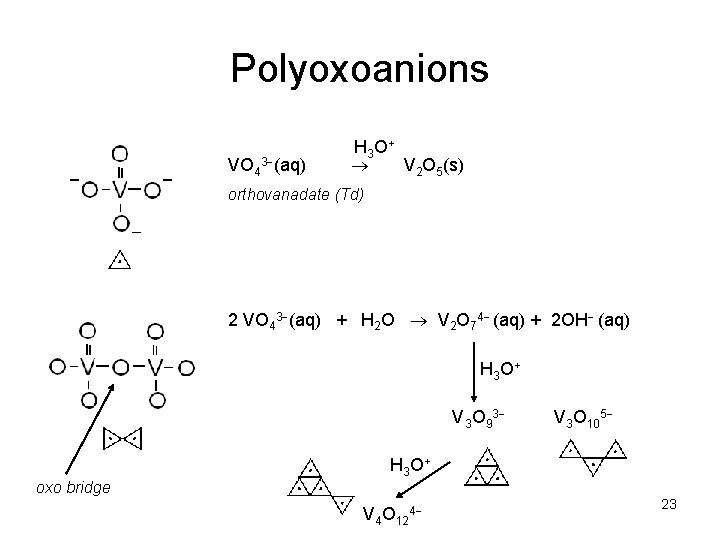
Polyoxoanions VO 43 (aq) H 3 O + V 2 O 5(s) orthovanadate (Td) 2 VO 43 (aq) + H 2 O V 2 O 74 (aq) + 2 OH (aq) H 3 O + V 3 O 93 V 3 O 105 H 3 O + oxo bridge V 4 O 124 23

Lewis acids and bases A LA + B: LB A: B complex LA = electron pair acceptor; LB = electron pair donor Lewis definition is more general than BL definition, does not require aqueous or protic solvent Ex: W + 6 : CO [W(CO)6] BCl 3 + : OEt 2 BCl 3: OEt 2 D 3 h Fe 3+(g) + 6 : OH 2 → [Fe(OH 2)6]3+ 24

LA/LB strengths LA strength is based on reaction Kf LA/LB strengths depend on specific acid base combination Ex: BCl 3 + : NR 3 Kf: NH 3 < Me. NH 2 < Me 2 NH < Me 3 N BMe 3 + : NR 3 Kf: Cl 3 B: NR 3 Me 3 B: NR 3 NH 3 < Me. NH 2 < Me 2 NH > Me 3 N Hrxn 58 74 inductive effect 81 inductive + steric 74 k. J/mol 25
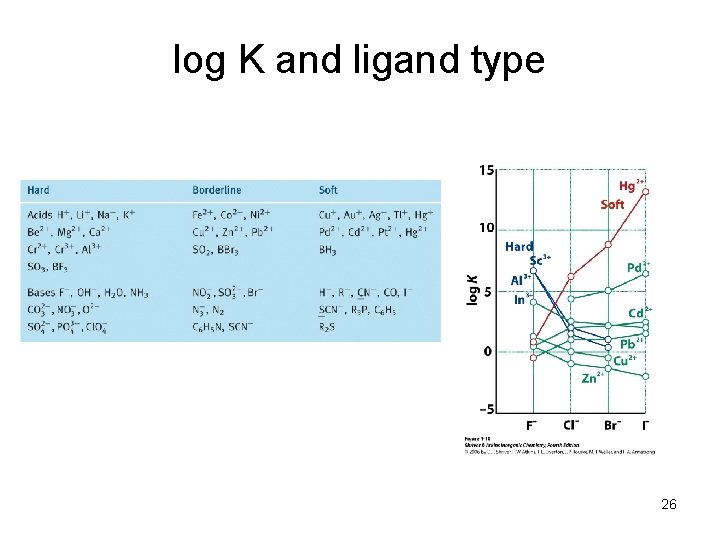
log K and ligand type 26

Drago-Wayland equation A (g) + : B (g) A: B (g) Gas phase reactions (omits solvation effects) - H rxn = EA EB + CA CB look up E, C values for reactants (Table 4. 4) 27

Donor/Acceptor numbers Commonly used to choose appropriate solvents (Table 4. 5) Donor Number (DN) is derived from Hrxn (Sb. Cl 5 + : B Cl 5 Sb: B) higher DN corresponds to stronger LB Acceptor Number (AN) is derived from stability of Et 3 P=O: A complex higher AN corresponds to stronger LA Ex: THF (tetrahydrofuran) C 4 H 8 O DN AN ε dielectric constant THF 20 8 7 H 2 O 18 55 82 Some Li+ salts and BF 3 have similar solubilities in THF, H 2 O NH 3 is much more soluble in H 2 O Most salts are much more soluble in H 2 O 28

Descriptive chemistry - Group 13 Expect inductive effect ex: BF 3 > BCl 3 > BBr 3 but the opposite is true BF 3 is stable in H 2 O, R 2 O (ethers) BCl 3 rapidly hydrolyzes due to nucleophilic attack of : OH 2 the lower acidity of BF 3 is due to unusually favorable B–X bonding in the planar conformation due to interaction “Al. Cl 3“ is a dimer (Al 2 Cl 6) General trend larger central atom, tends to have higher CN Al 2 Me 6 is isostructural with Al 2 Cl 6 Friedel-Crafts C 6 H 6 C 6 H 5 C(O)R RC(O)-X: + “Al. Cl 3” RC(O) + Al. Cl 3 X 29

Descriptive chemistry - Group 14 CX 4 is not a Lewis Acidity Si. F 4 > Si. Cl 4 > Si. Br 4 > Si. I 4 (inductive effect) ex: 2 KF(s) + Si. F 4(g) K 2 Si. F 6(s) LB LA Si. F 62 Oh Sn. F 4 and Pb. F 4 have Oh not Td coordination (heavier congener, higher CN) each M has 2 unique axial F and 4 shared F 30

Descriptive chemistry - Group 15 MF 5 does not exist for nitrogen; it’s trigonal bipyramidal for M = P, As Sb. F 5: Sb has Oh coordination (oligomerizes to Sb 4 F 20 or Sb 6 F 30) LB LA K 2 Mn. F 6 (s) + 2 Sb. F 5 (l) KF, H 2 O 2 aqu HF KMn. O 4 Sb 2 O 3 transient “Mn. F 4” + 2 KSb. F 6 (s) F transfer Mn. F 3 + ½ F 2 (g) Dove (1980’s), chemical synthesis of F 2 gas 31
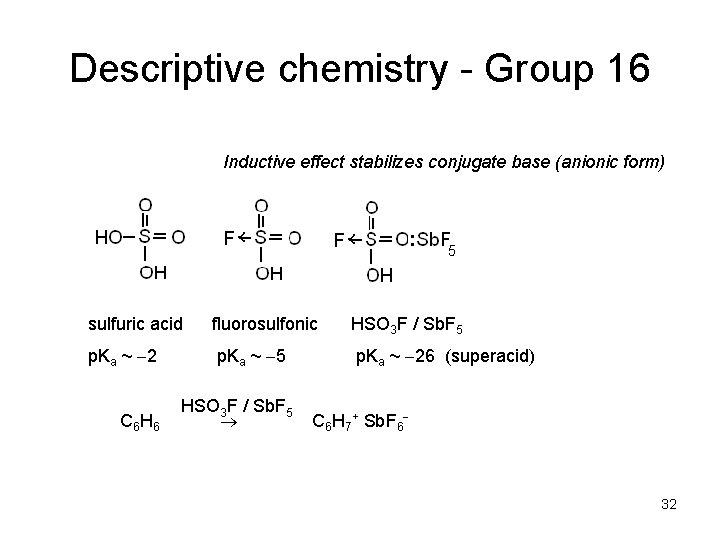
Descriptive chemistry - Group 16 Inductive effect stabilizes conjugate base (anionic form) sulfuric acid fluorosulfonic HSO 3 F / Sb. F 5 p. Ka ~ 2 p. Ka ~ 5 p. Ka ~ 26 (superacid) C 6 HSO 3 F / Sb. F 5 C 6 H 7+ Sb. F 6 32
- Slides: 32