CH 339 K Proteins Higher Order Structure Higher














- Slides: 74

CH 339 K Proteins: Higher Order Structure

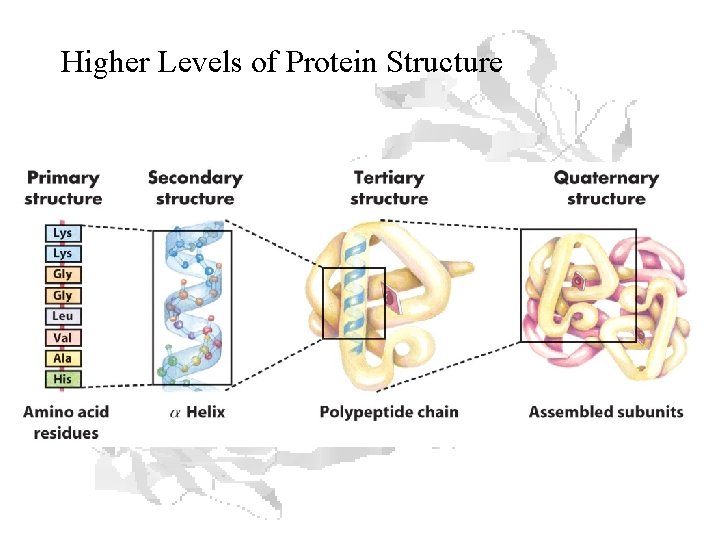
Higher Levels of Protein Structure

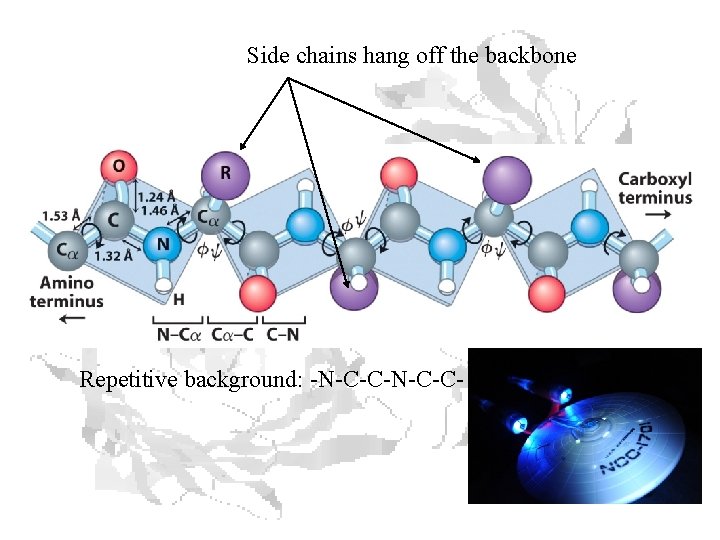
Side chains hang off the backbone Repetitive background: -N-C-C-

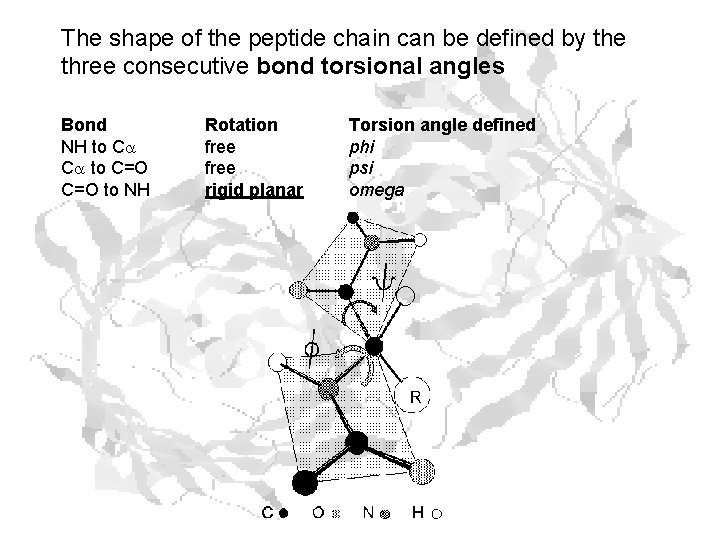
The shape of the peptide chain can be defined by the three consecutive bond torsional angles Bond NH to Ca Ca to C=O to NH Rotation free rigid planar Torsion angle defined phi psi omega

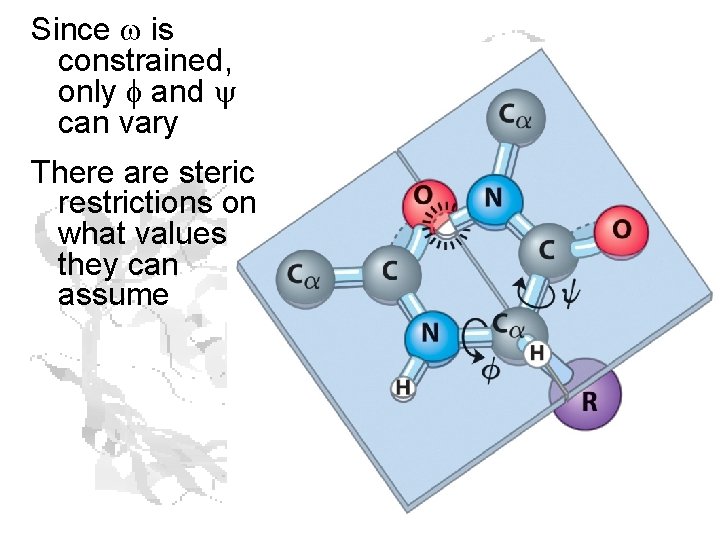
Since w is constrained, only f and y can vary There are steric restrictions on what values they can assume

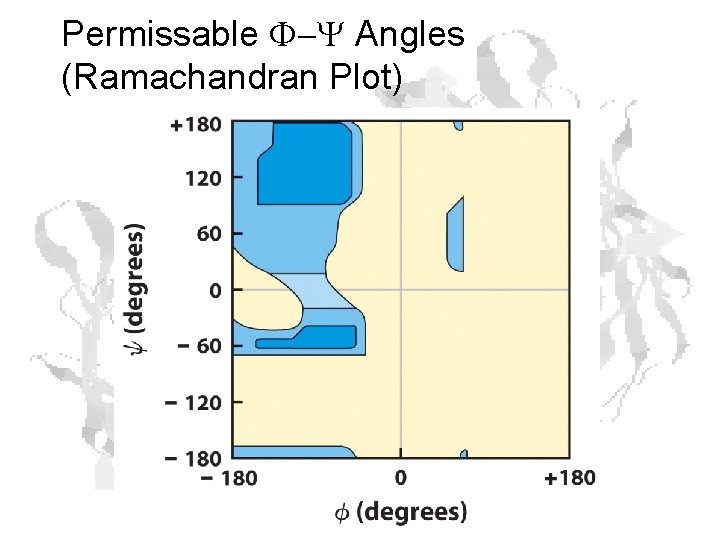
Permissable F-Y Angles (Ramachandran Plot)

Secondary Structures • Represent interactions among backbone atoms • Examples § § a-helices Other helices b-sheets b- and g-turns § These structures have characteristic f and y angles

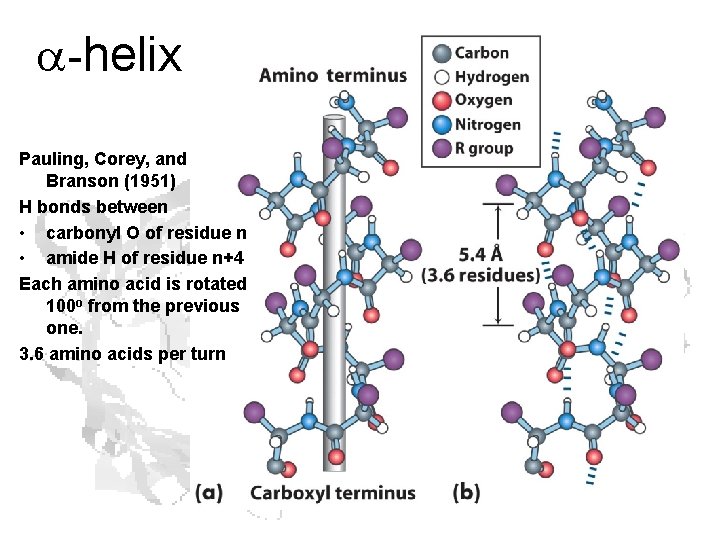
a-helix Pauling, Corey, and Branson (1951) H bonds between • carbonyl O of residue n • amide H of residue n+4 Each amino acid is rotated 100 o from the previous one. 3. 6 amino acids per turn

R/V Alpha Helix Woods Hole Oceanographic Institute 1966 -2011

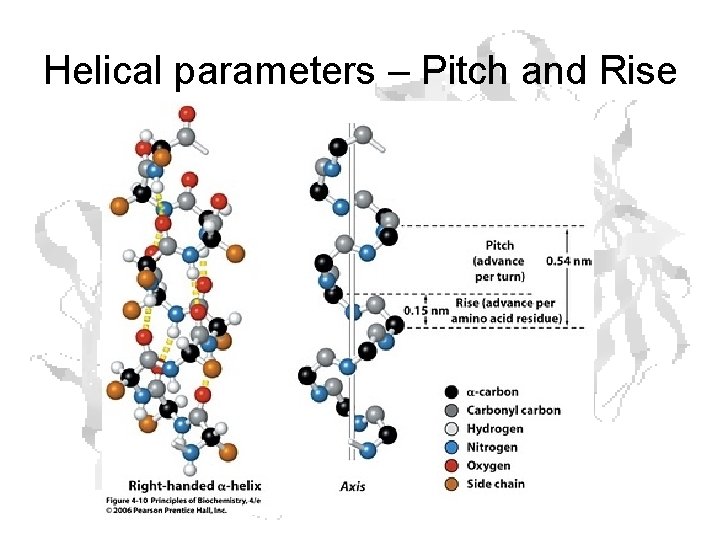
Helical parameters – Pitch and Rise

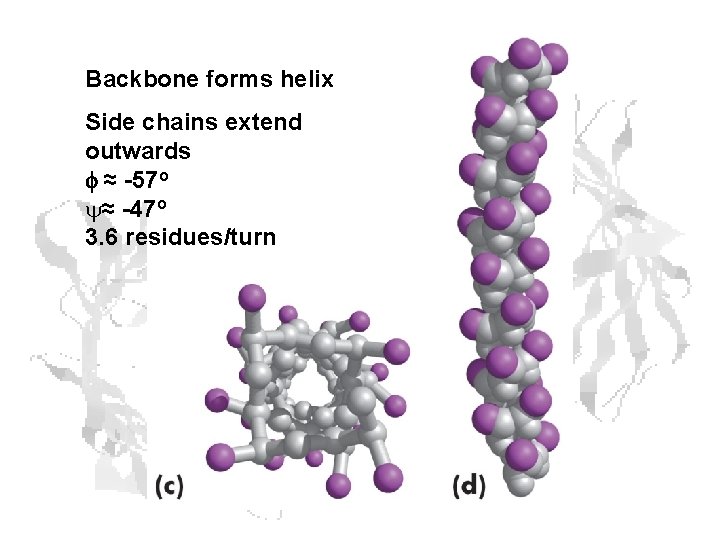
Backbone forms helix Side chains extend outwards f ≈ -57 o y≈ -47 o 3. 6 residues/turn

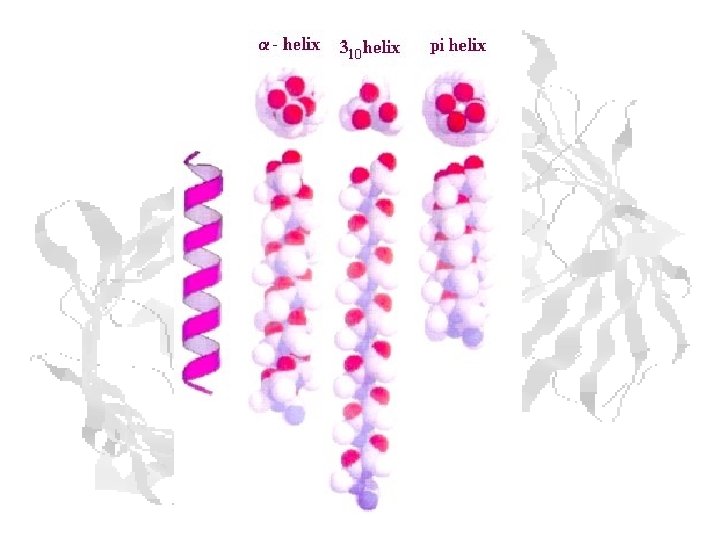
Helix Types a-helix: C=O H-bonded to NH of residue n+4 (aka 3. 613 helix) 310 helix: C=O H-bonded to NH of residue n+3 – (f ≈ -49 o y ≈ -26 o) p-helix: C=O H-bonded to NH of residue n+5 (aka 4. 116 helix) (f ≈ -57 o y ≈ -80 o)

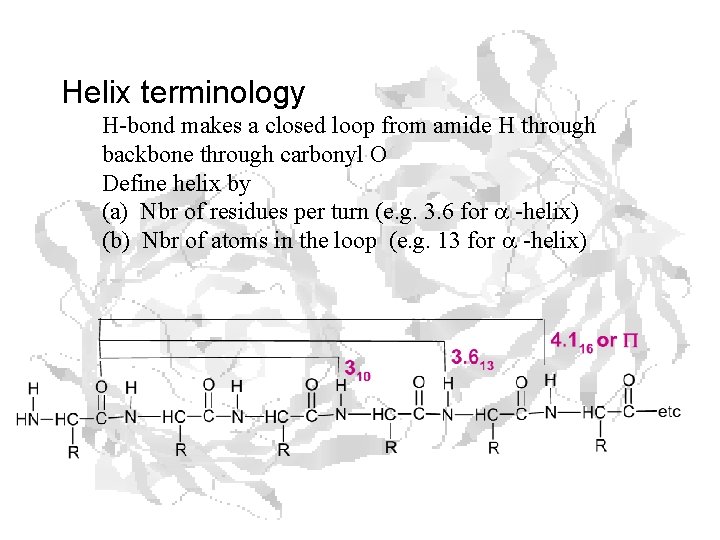
Helix terminology H-bond makes a closed loop from amide H through backbone through carbonyl O Define helix by (a) Nbr of residues per turn (e. g. 3. 6 for a -helix) (b) Nbr of atoms in the loop (e. g. 13 for a -helix)


Idealized Helices

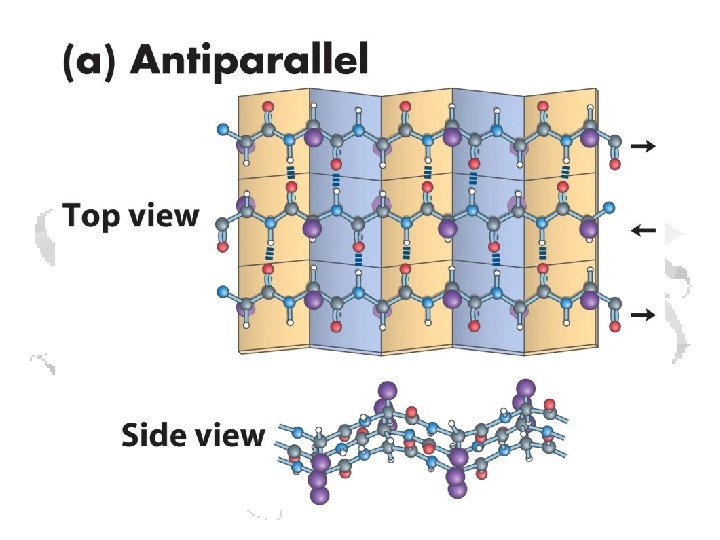
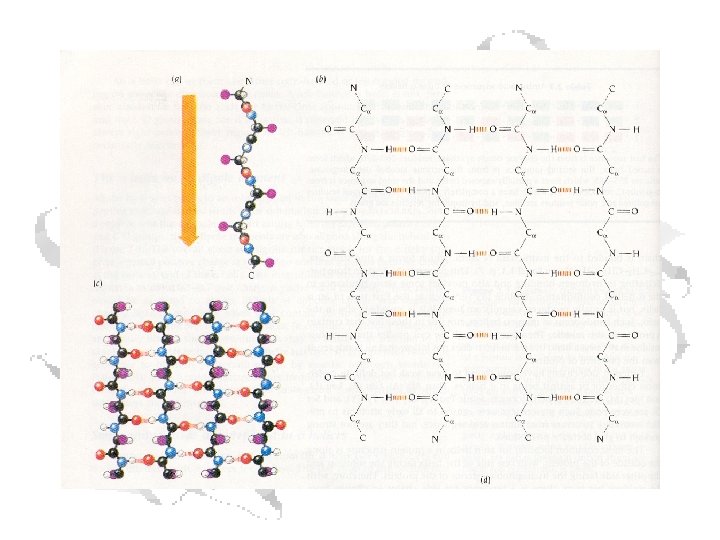
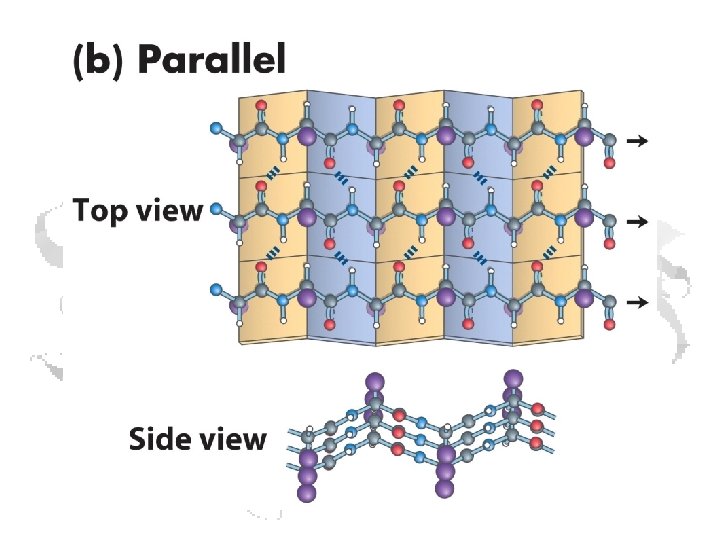
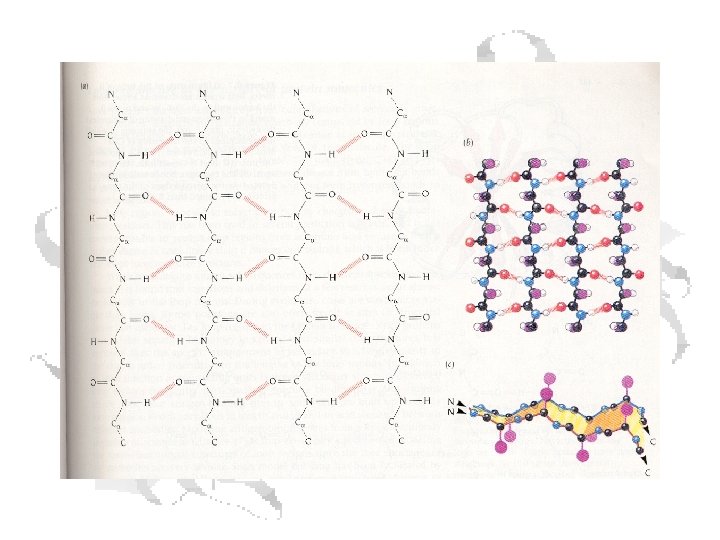
b-Sheets • Can be thought of as helix with two residues per helix • Backbone atoms run in a plane • Side chains extend up and down from plane § f ≈ -110 o to -140 o § y ≈ +110 o to +135 o





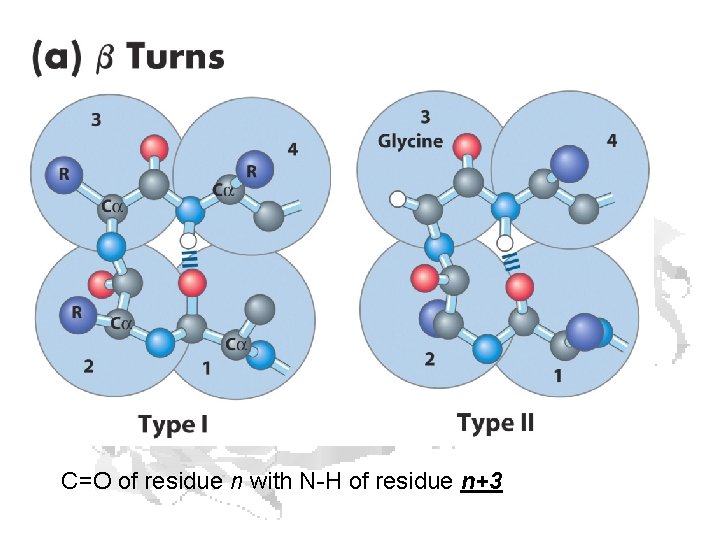
C=O of residue n with N-H of residue n+3

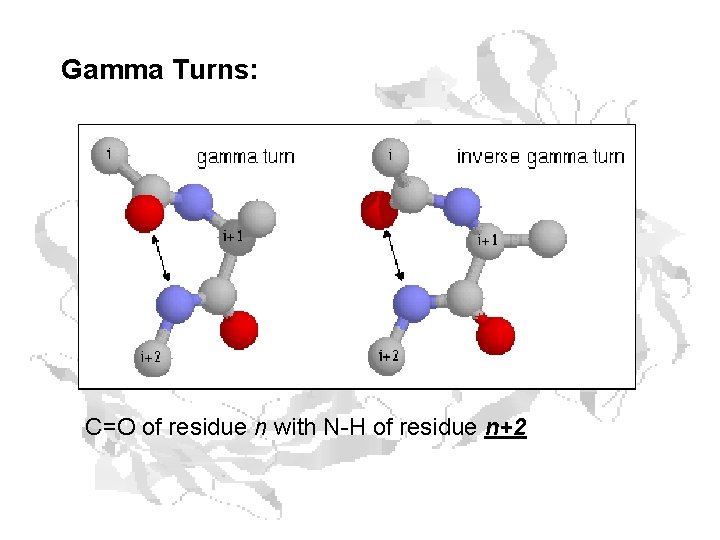
Gamma Turns: C=O of residue n with N-H of residue n+2

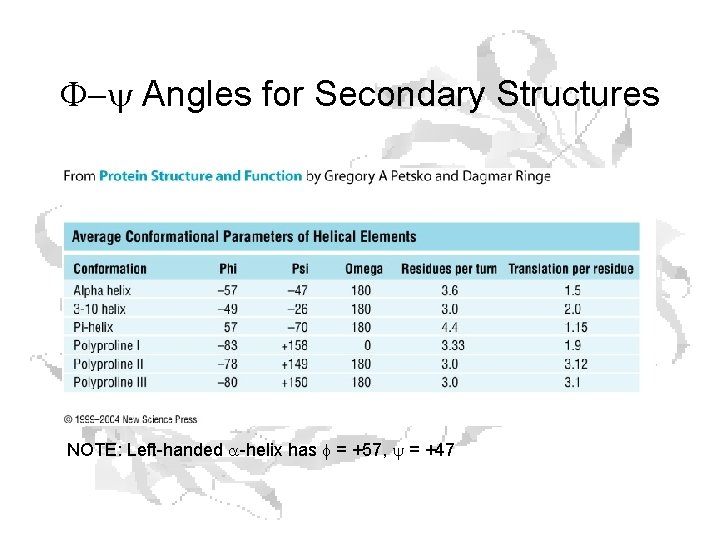
F-y Angles for Secondary Structures NOTE: Left-handed a-helix has f = +57, y = +47

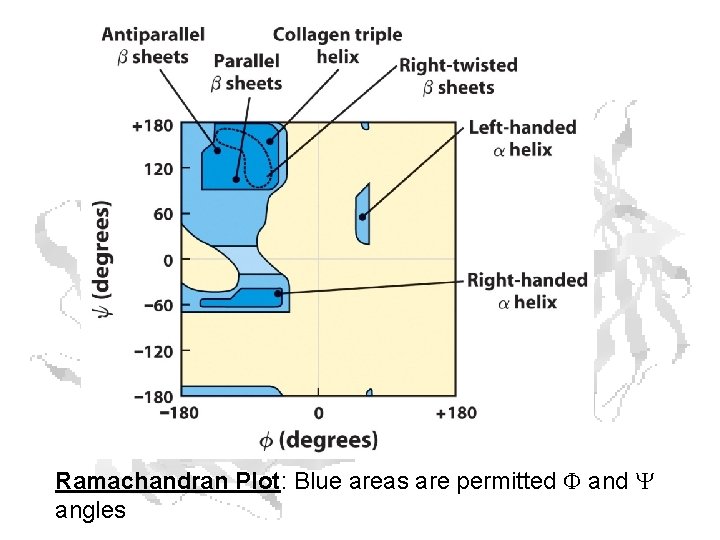
Ramachandran Plot: Blue areas are permitted F and Y angles

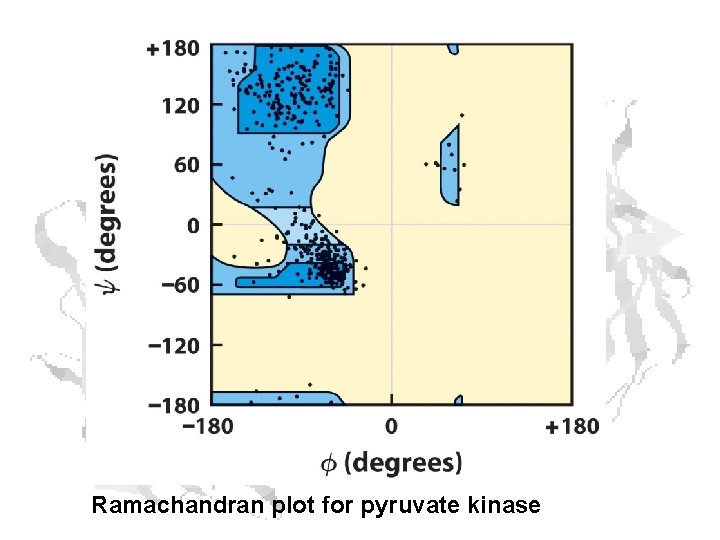
Ramachandran plot for pyruvate kinase

Tertiary Structures • Three dimensional folding • Determined by side chain interactions – Salt links – H-Bonds – Disulfides – Hydrophobic interactions • Fibrous Proteins • Globular Proteins

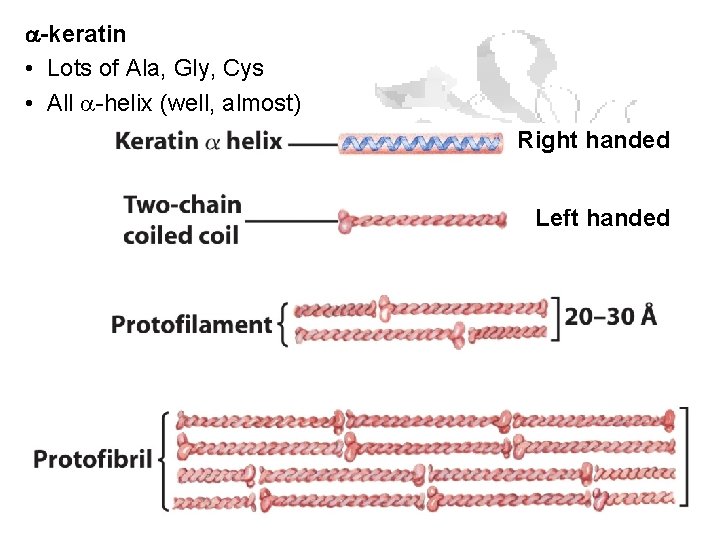
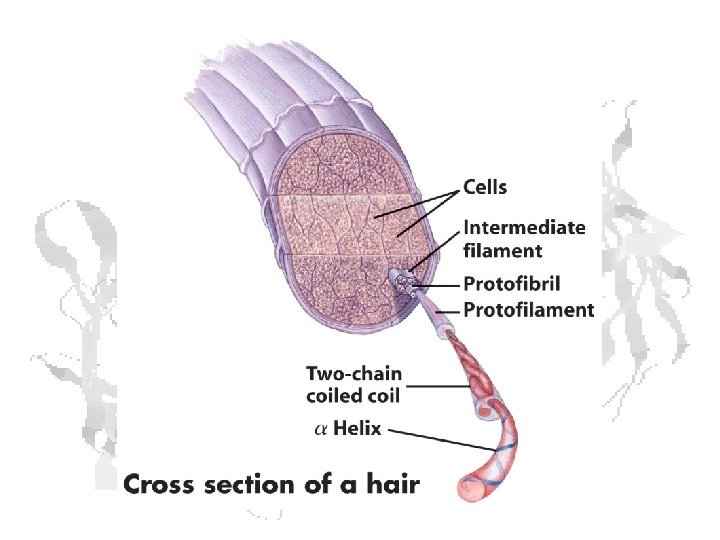
Fibrous Proteins Keratin a-keratin: hair, horns, and hoofs of mammals b-keratin: scales, claws and shells of reptiles, beaks and claws of birds, porcupine quills

a-keratin • Lots of Ala, Gly, Cys • All a-helix (well, almost) Right handed Left handed


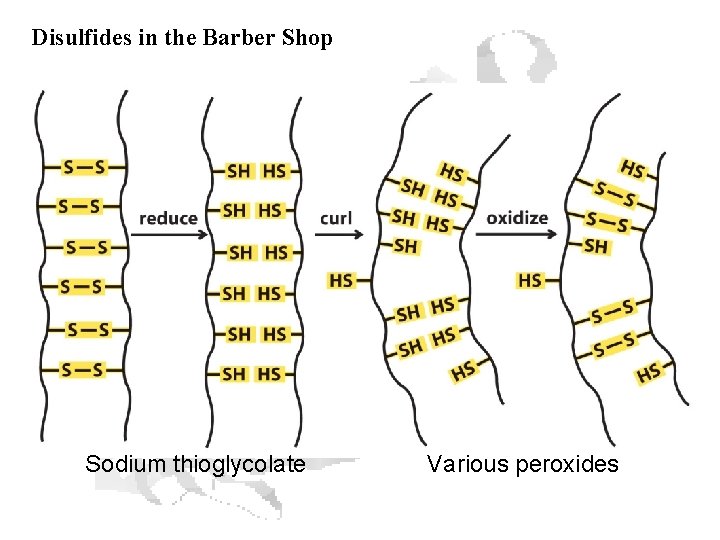
Disulfides in the Barber Shop Sodium thioglycolate Various peroxides

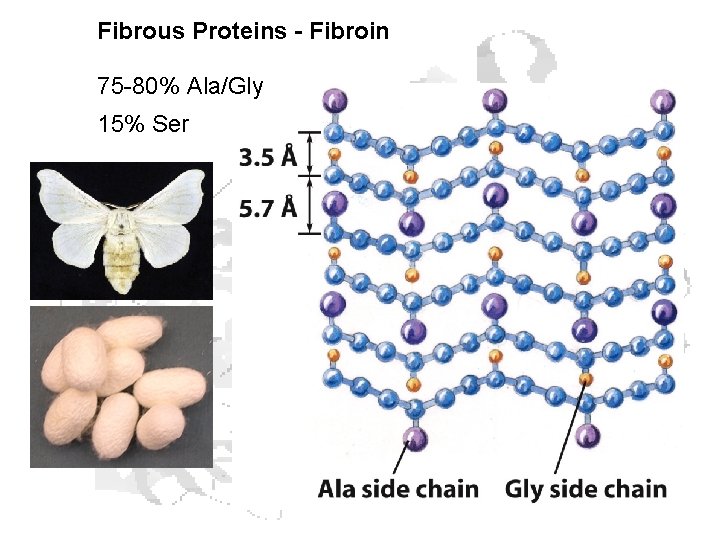
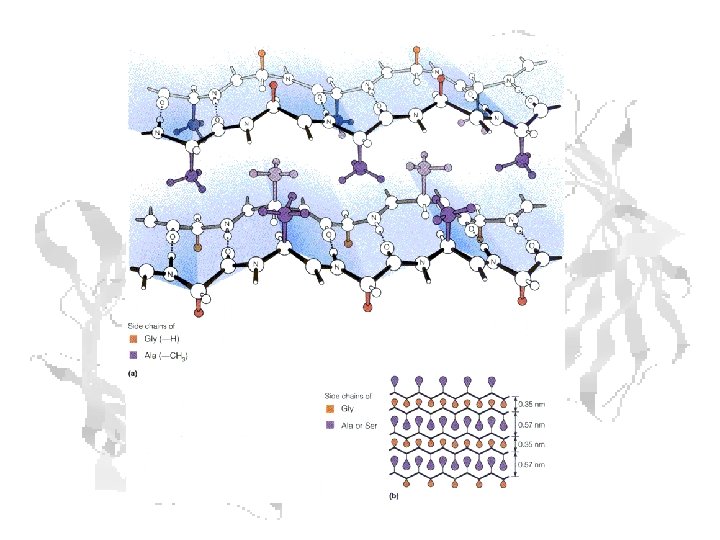
Fibrous Proteins - Fibroin 75 -80% Ala/Gly 15% Ser



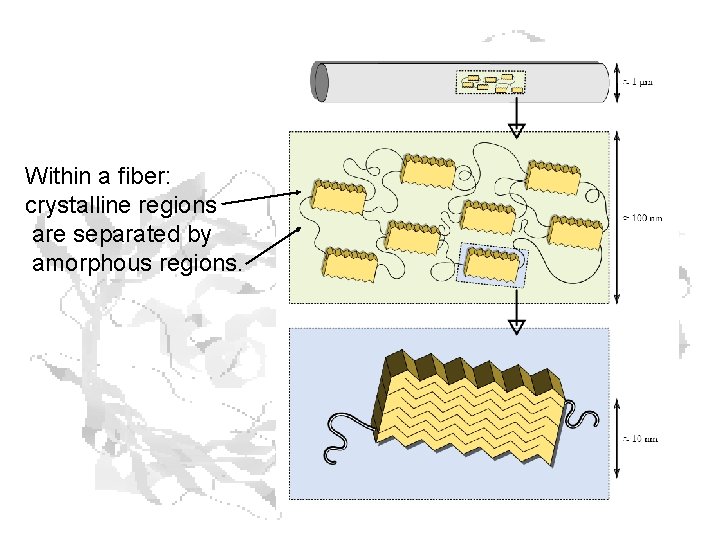
Within a fiber: crystalline regions are separated by amorphous regions.

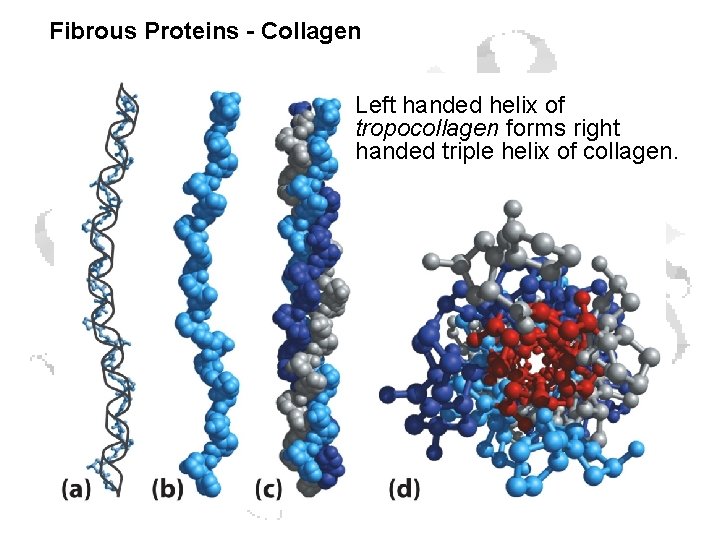
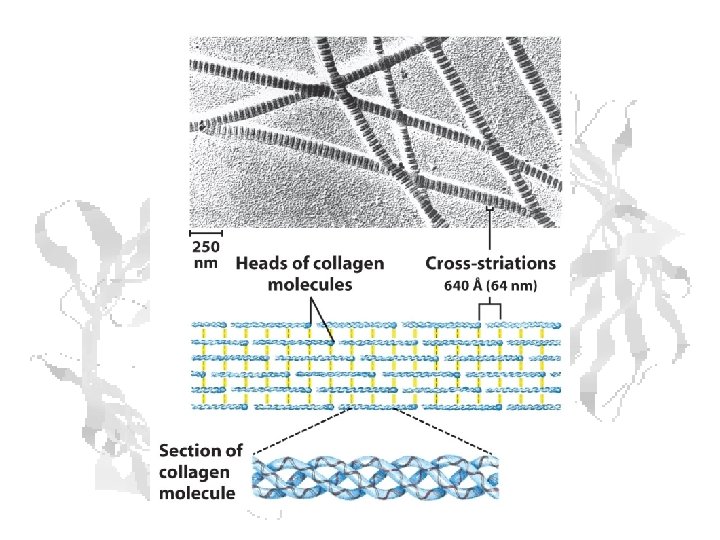
Fibrous Proteins - Collagen Left handed helix of tropocollagen forms right handed triple helix of collagen.


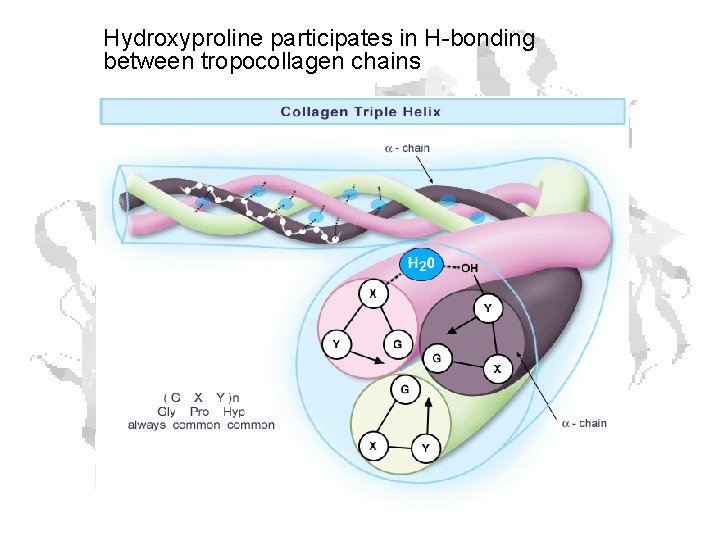
Hydroxyproline participates in H-bonding between tropocollagen chains

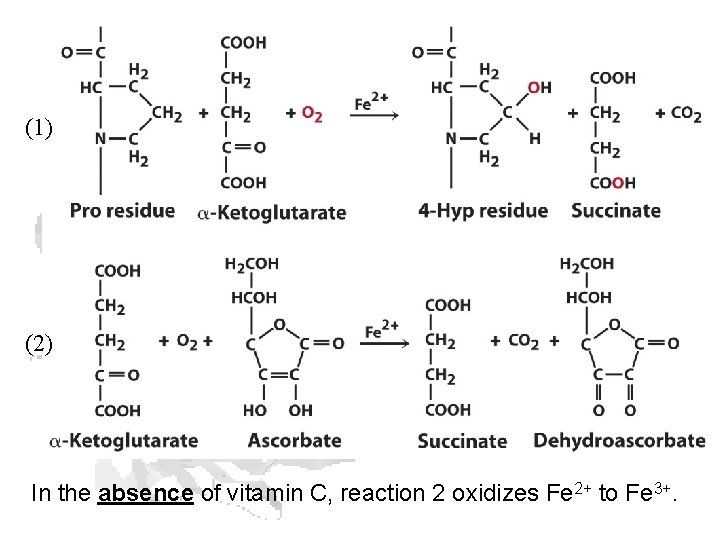
(1) (2) In the absence of vitamin C, reaction 2 oxidizes Fe 2+ to Fe 3+.

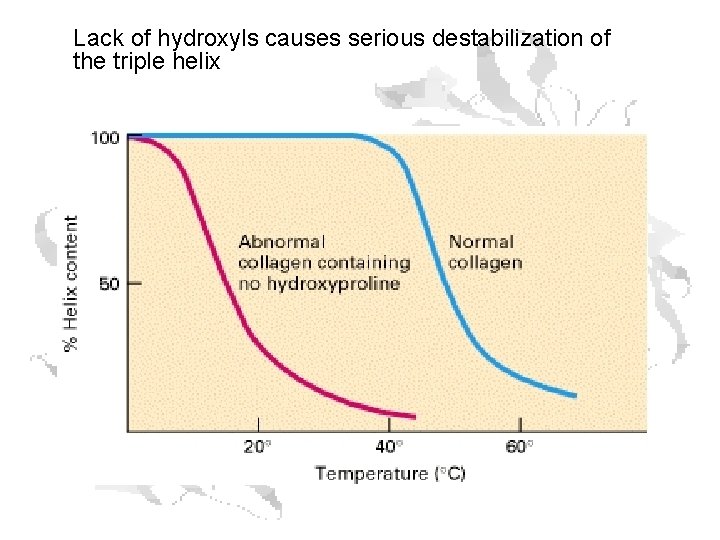
Lack of hydroxyls causes serious destabilization of the triple helix

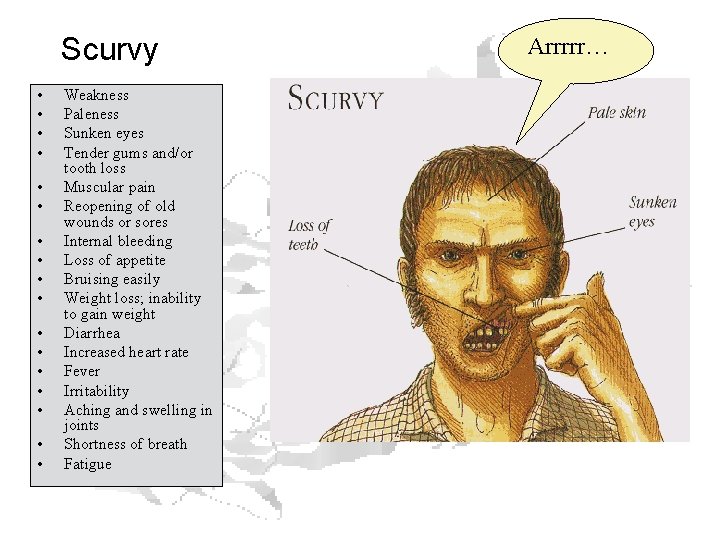
Scurvy • • • • • Weakness Paleness Sunken eyes Tender gums and/or tooth loss Muscular pain Reopening of old wounds or sores Internal bleeding Loss of appetite Bruising easily Weight loss; inability to gain weight Diarrhea Increased heart rate Fever Irritability Aching and swelling in joints Shortness of breath Fatigue Arrrrr…

The Brits Found the Link Between Fruits and Veggies and Healthy Sailors Walk wide o' the Widow at Windsor, For 'alf o' Creation she owns: We 'ave bought 'er the same with the sword an' the flame, An' we've salted it down with our bones. (Poor beggars! -- it's blue with our bones!) The Widow at Windsor – Kipling We broke a King and we built a road -A court-house stands where the reg'ment goed. And the river's clean where the raw blood flowed When the Widow give the party. (Bugle: Ta--rara--ra-ra-rara!) The Widow’s Party - Kipling

British Empire at its Peak • A healthy navy is a victorious navy (of course, my ancestors were less than thrilled…)

Protein structure cartoons a-helix Antiparallel b-sheet

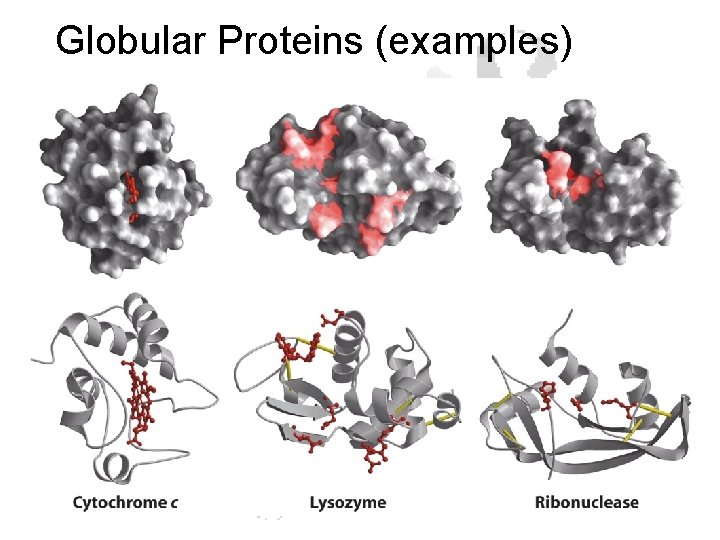
Globular Proteins (examples)


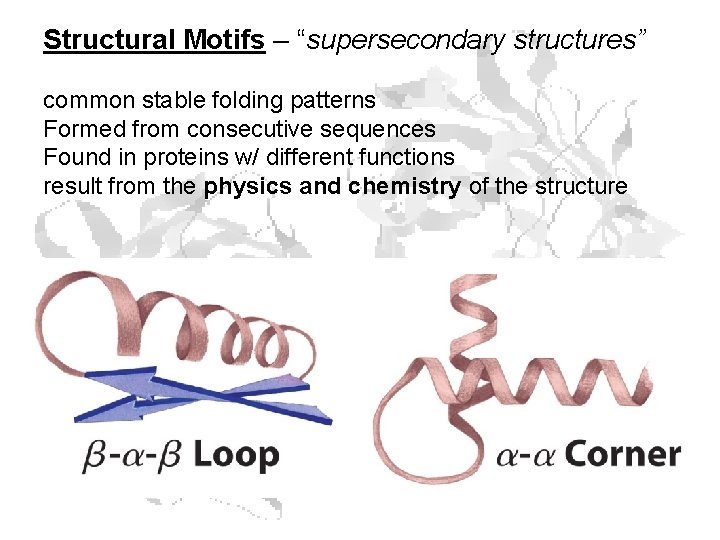
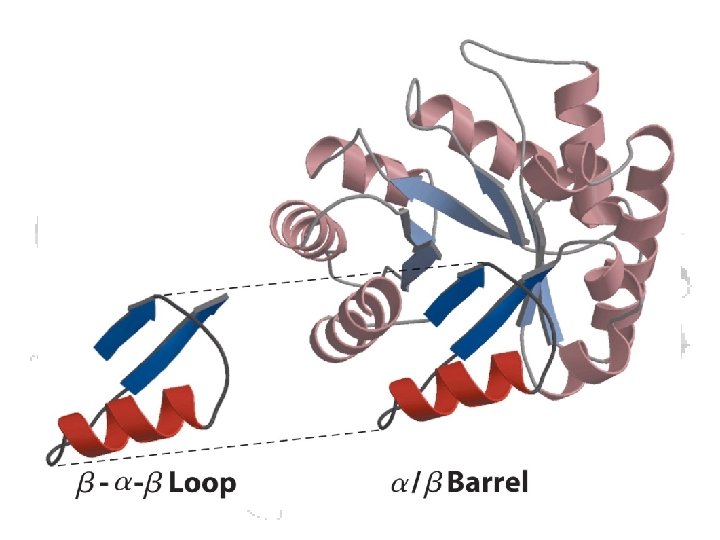
Structural Motifs – “supersecondary structures” common stable folding patterns Formed from consecutive sequences Found in proteins w/ different functions result from the physics and chemistry of the structure

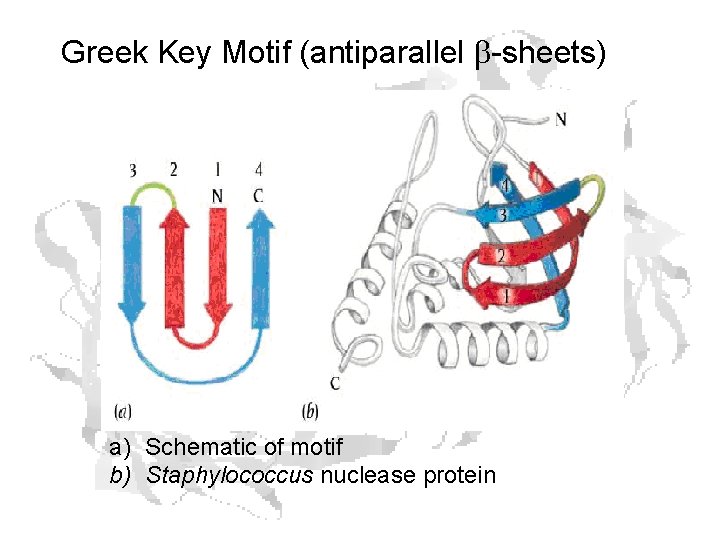
Greek Key Motif (antiparallel b-sheets) a) Schematic of motif b) Staphylococcus nuclease protein

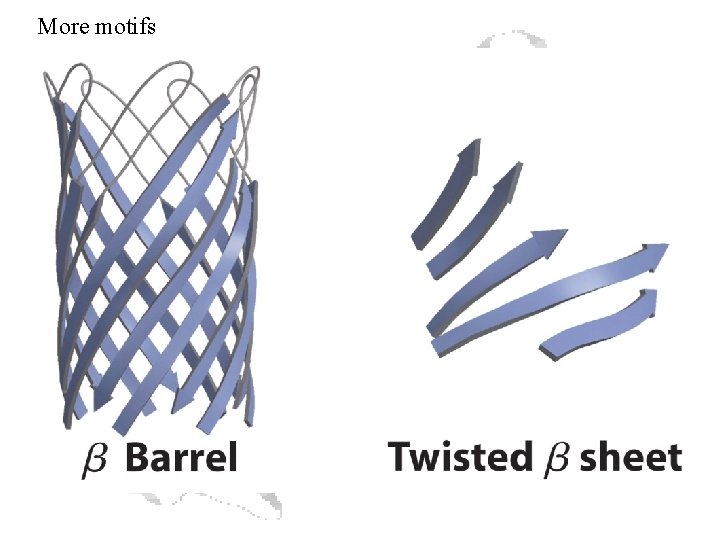
More motifs

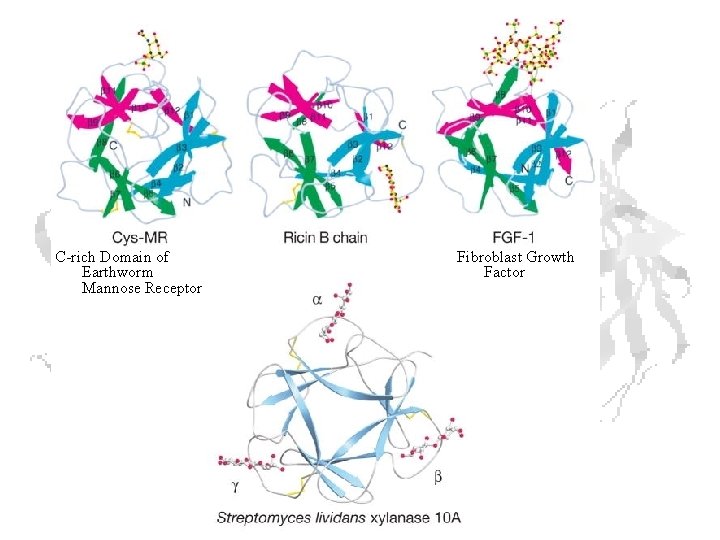
Domains – • Stable, independently folded, globular units • Common patterns found in different proteins • Typically have similar function • Caused by evolution (gene recombination / duplication) • Frequently (not always!) correspond to exons in genes Ricin B chain • Two domains • Each domain is a trefoil • 3 repeats of a sheet-loop structure • i. e. 6 repeats of a primitive fold

C-rich Domain of Earthworm Mannose Receptor Fibroblast Growth Factor

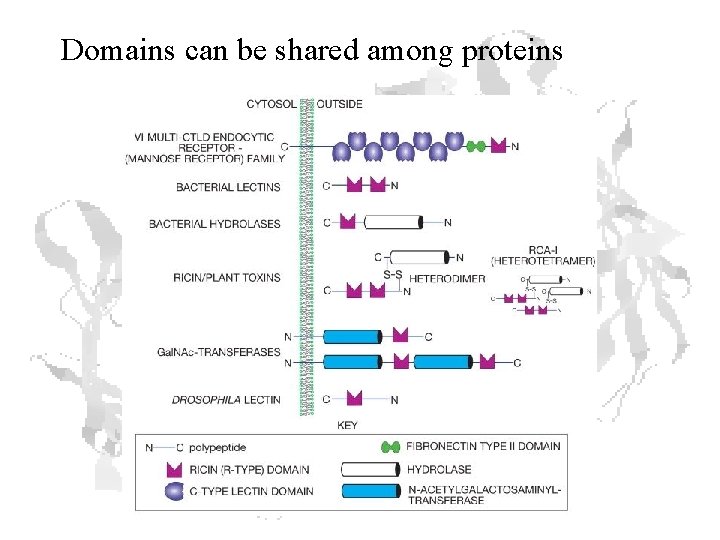
Domains can be shared among proteins


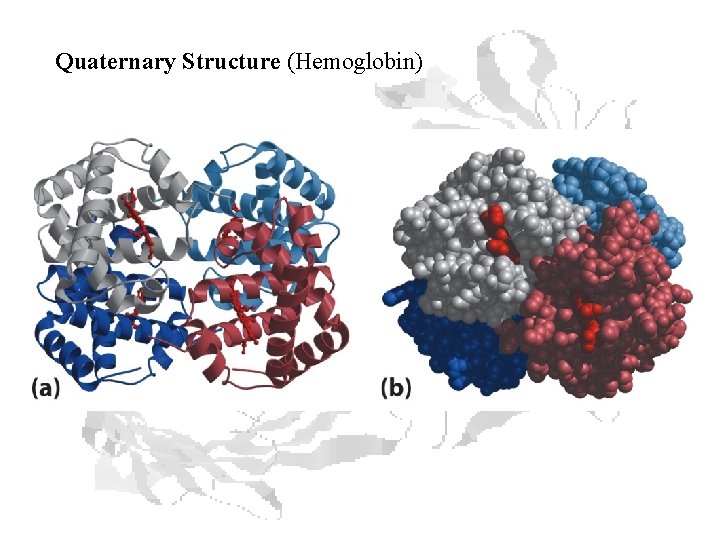
Quaternary Structure (Hemoglobin)

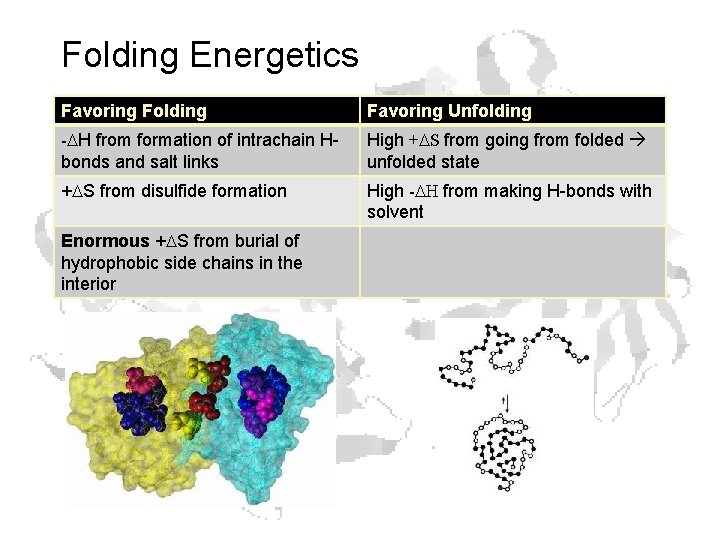
Folding Energetics Favoring Folding Favoring Unfolding -DH from formation of intrachain Hbonds and salt links High +DS from going from folded unfolded state +DS from disulfide formation High -DH from making H-bonds with solvent Enormous +DS from burial of hydrophobic side chains in the interior

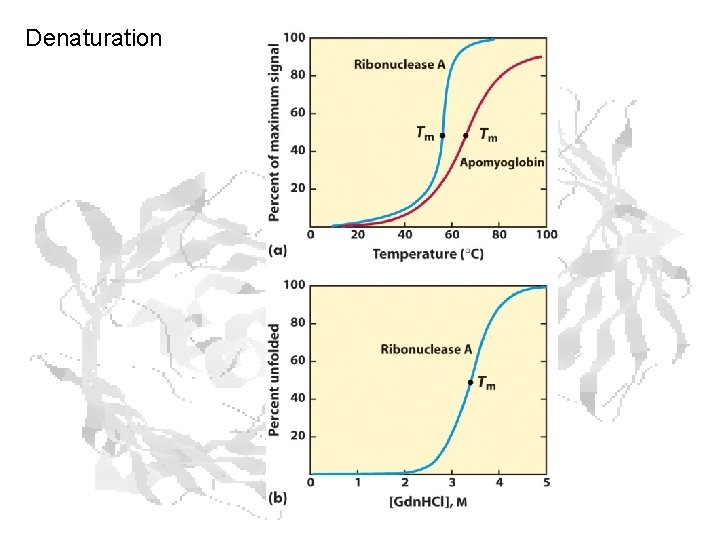
Denaturation

Denaturants • • • Heat (increases negative TDS contribution) Cold (H 2 O becomes less disordered) Pressure High and low p. H (electrostatic effects) Low-polarity and non-polar solvents (e. g. Et. OH) • Chaotropes (urea, guanidinium chloride)

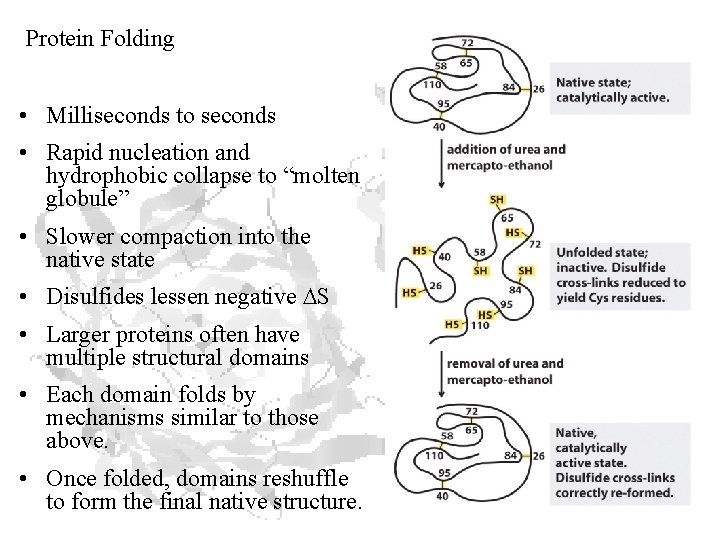
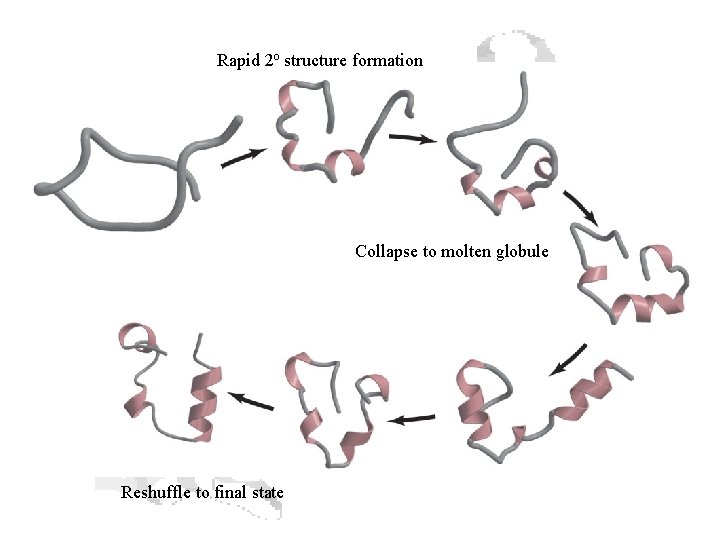
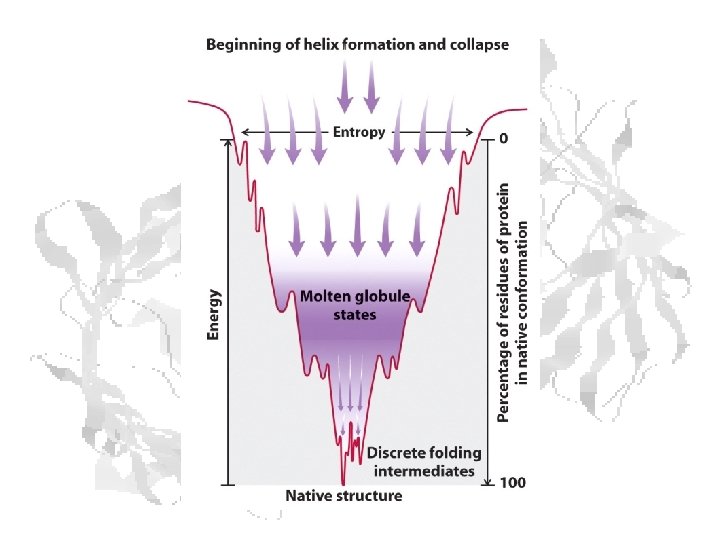
Protein Folding • Milliseconds to seconds • Rapid nucleation and hydrophobic collapse to “molten globule” • Slower compaction into the native state • Disulfides lessen negative DS • Larger proteins often have multiple structural domains • Each domain folds by mechanisms similar to those above. • Once folded, domains reshuffle to form the final native structure.

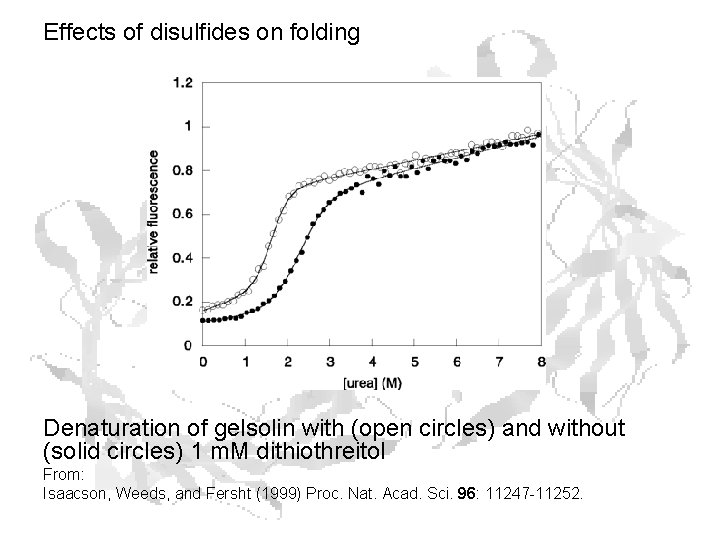
Effects of disulfides on folding Denaturation of gelsolin with (open circles) and without (solid circles) 1 m. M dithiothreitol From: Isaacson, Weeds, and Fersht (1999) Proc. Nat. Acad. Sci. 96: 11247 -11252.

Rapid 2 o structure formation Collapse to molten globule Reshuffle to final state


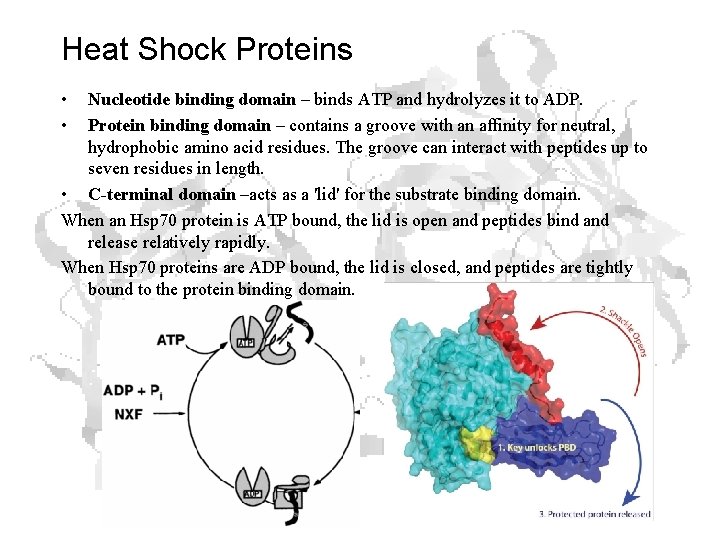
Heat Shock Proteins • • Nucleotide binding domain – binds ATP and hydrolyzes it to ADP. Protein binding domain – contains a groove with an affinity for neutral, hydrophobic amino acid residues. The groove can interact with peptides up to seven residues in length. • C-terminal domain –acts as a 'lid' for the substrate binding domain. When an Hsp 70 protein is ATP bound, the lid is open and peptides bind and release relatively rapidly. When Hsp 70 proteins are ADP bound, the lid is closed, and peptides are tightly bound to the protein binding domain.

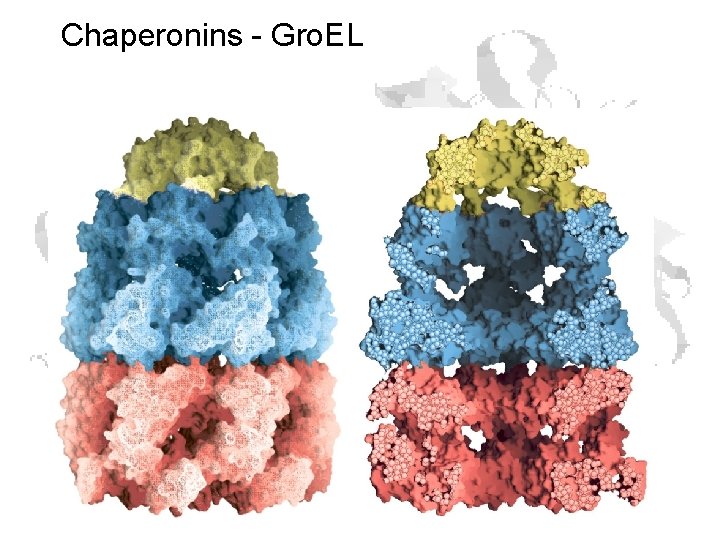
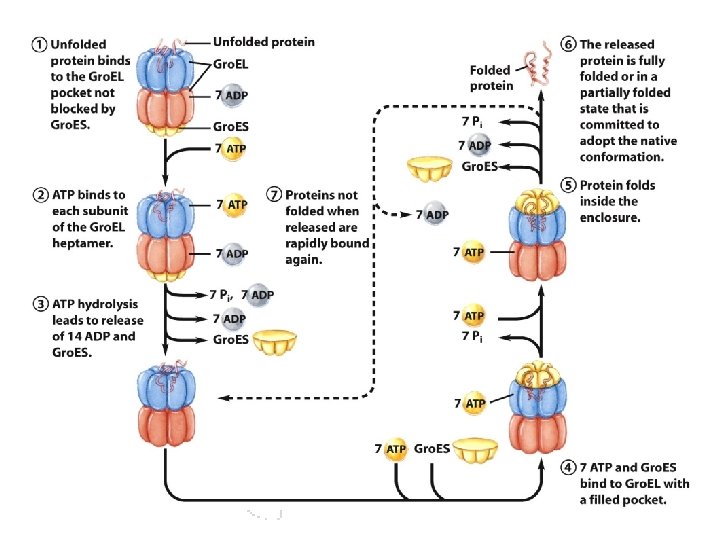
Chaperonins - Gro. EL


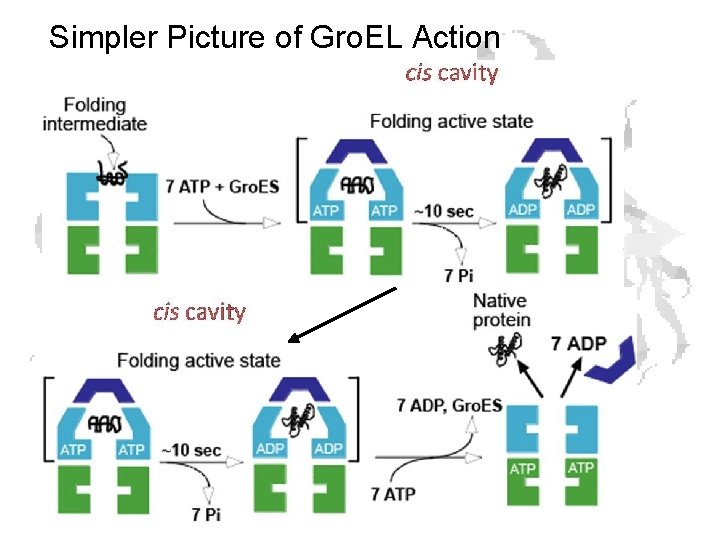
Simpler Picture of Gro. EL Action

A Problem in Folding Creutzfeldt-Jakob Disease, Mad Cows, and the Laughing Disease of the New Guinea Cannibals Initially, persons may have difficulty sleeping, experience depression, problems with muscular coordination, impaired vision, and personality and behavioral changes such as impaired memory, judgment, and thinking. As the disease progresses, mental impairment becomes severe and involuntary muscle jerks (myoclonus) often occur along with blindness. Eventually, the ability to move or speak is lost and the person enters a coma until death occurs. (100% fatal)

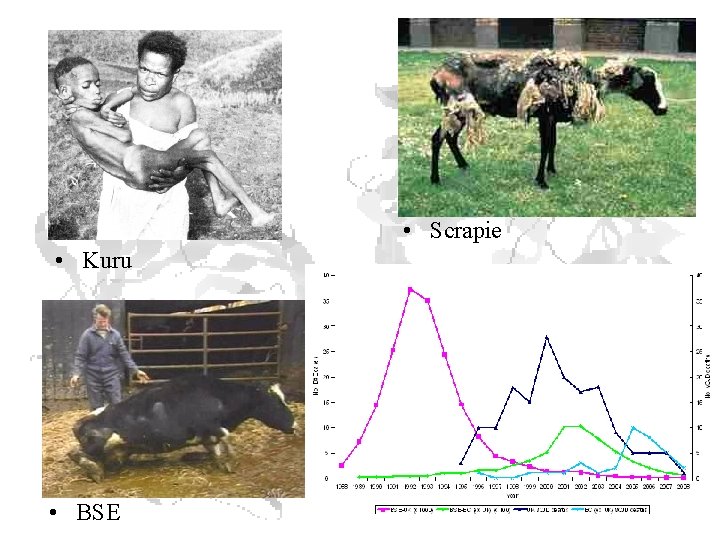
Prion Diseases • Human Prion Diseases • • • Creutzfeldt-Jakob Disease (CJD) Variant Creutzfeldt-Jakob Disease (v. CJD) Gerstmann-Straussler-Scheinker Syndrome Fatal Familial Insomnia Kuru • Animal Prion Diseases • • • Bovine Spongiform Encephalopathy (BSE) Chronic Wasting Disease (CWD) Scrapie Transmissible mink encephalopathy Feline spongiform encephalopathy Ungulate spongiform encephalopathy

• Scrapie • Kuru • BSE

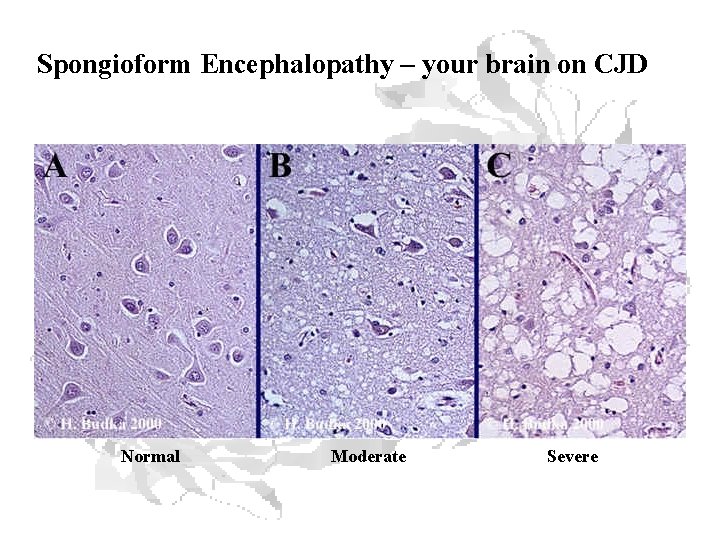
Spongioform Encephalopathy – your brain on CJD Normal Moderate Severe

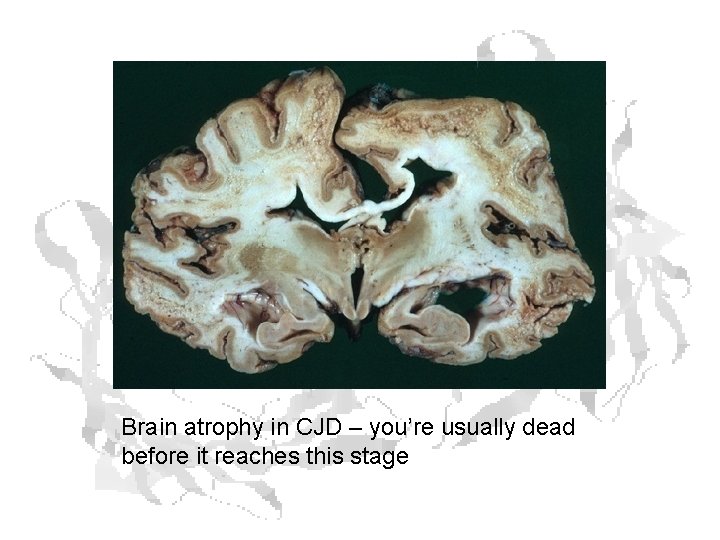
Brain atrophy in CJD – you’re usually dead before it reaches this stage

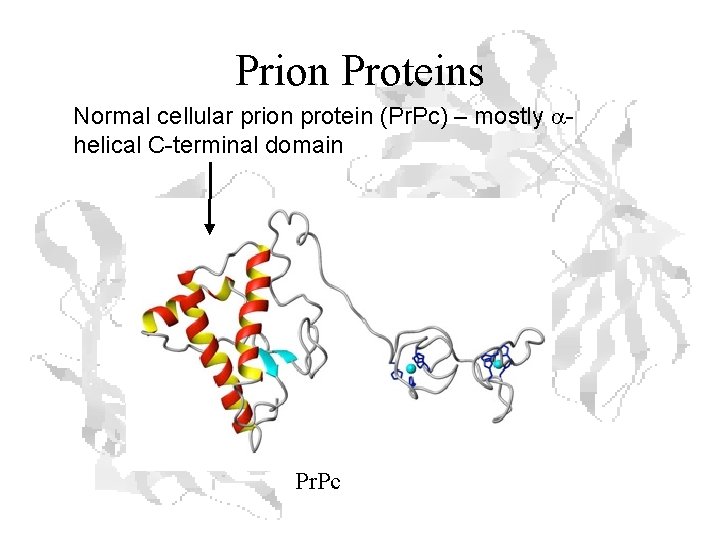
Prion Proteins Normal cellular prion protein (Pr. Pc) – mostly ahelical C-terminal domain Pr. Pc

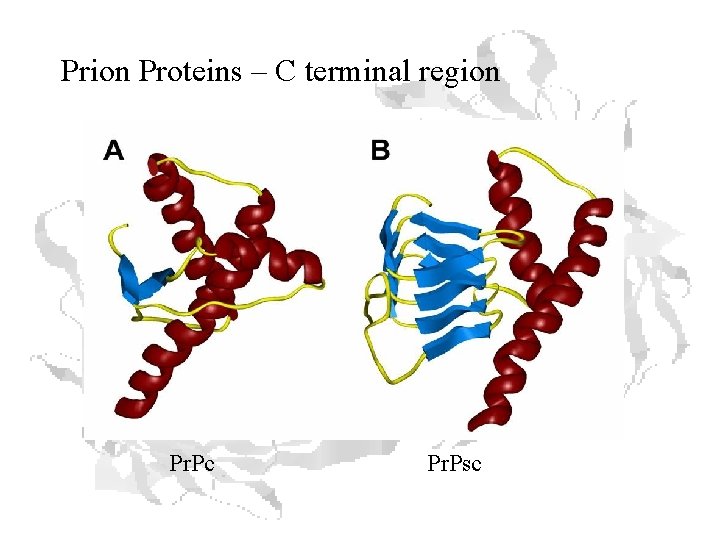
Prion Proteins – C terminal region Pr. Pc Pr. Psc

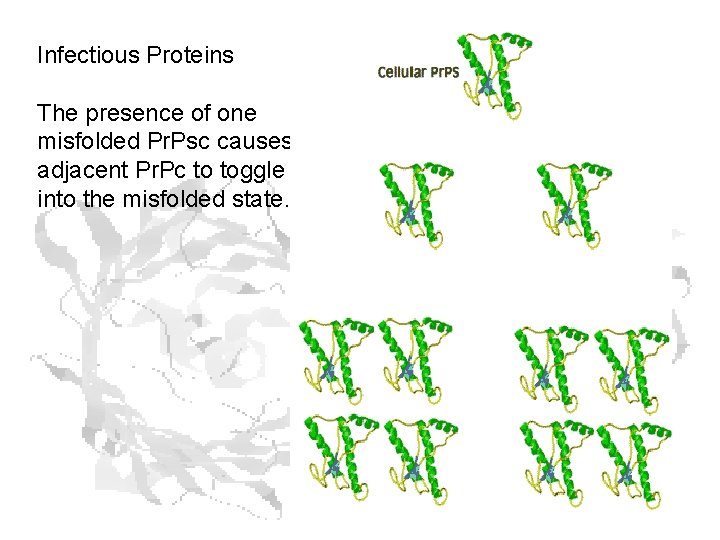
Infectious Proteins The presence of one misfolded Pr. Psc causes adjacent Pr. Pc to toggle into the misfolded state.

Various Mutations in CJD Prion Proteins Codon Amino acid change Reference 178 aspartate to asparagine Goldfarb 1991 b 180 valine to isoleucine Kitamoto 1993 a 188 threonine to alanine Collins 2000 196 glutamate to lysine Peoc’h 2000 200** glutamate to lysine Goldgaber 1989 203 valine to isoleucine Peoc’h 2000 208 arginine to histidine Mastrianni 1996 210 valine to isoleucine Pocchiari 1993 211 glutamate to glutamine Peoc’h 2000 232 methionine to arginine Kitamoto 1993 a
