Ch 25 Discovery of ElectronNucleus and Radiation Thomsons










- Slides: 14

Ch. 25 Discovery of Electron/Nucleus and Radiation

Thomson’s cathode ray experiment led to the discovery of electrons. Beam of Electrons Positive Magnet

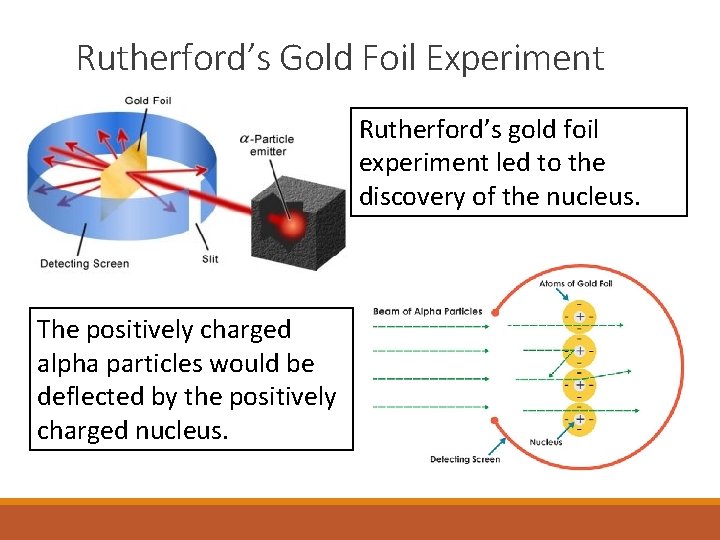
Rutherford’s Gold Foil Experiment Rutherford’s gold foil experiment led to the discovery of the nucleus. The positively charged alpha particles would be deflected by the positively charged nucleus.

Why can particles pass through atoms? -Atoms are mostly empty space! -If a hydrogen atom were a football field, a marble at its center would be the nucleus.

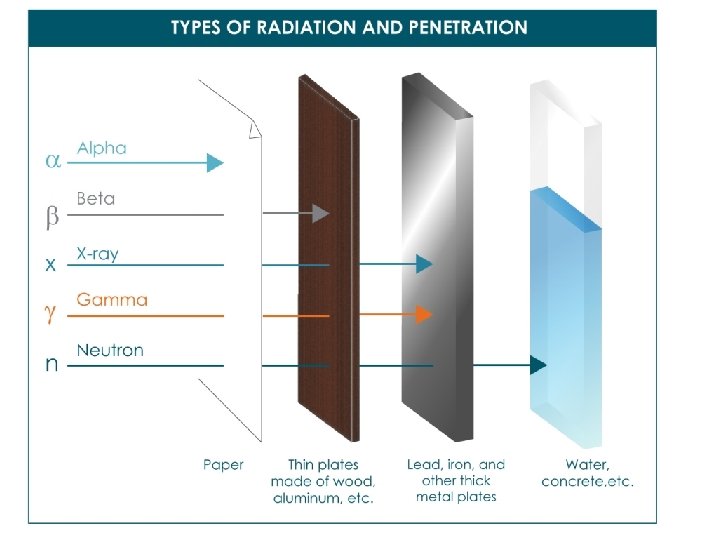
What happens during nuclear radiation? -Unstable nuclei want to become stable. -They emit energy and particles to achieve stability. -Three types of radiation: alpha a, beta b, and gamma g.

Alpha Radiation A helium atom without the electrons, has a +2 charge. An alpha particle is the nucleus of a helium atom.

Learning Check Write the correct products after the following elements undergo alpha decay.

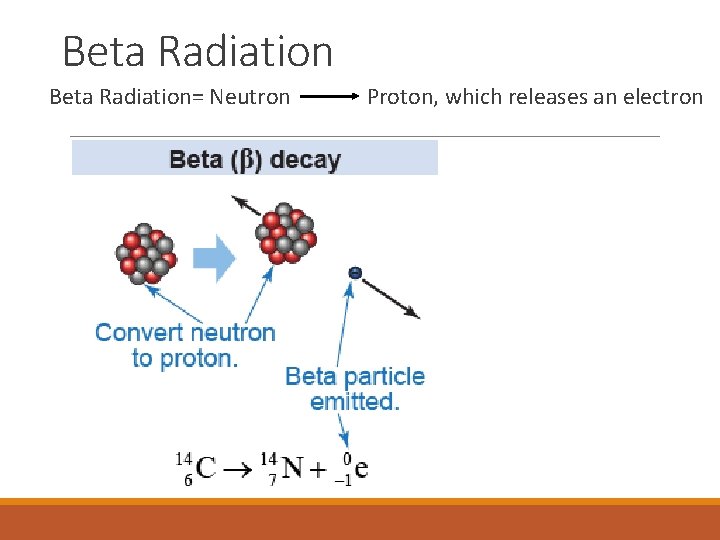
Beta Radiation= Neutron Proton, which releases an electron

Learning Check Write the correct products after the following elements undergo beta decay.

Gamma Radiation Often released alongside a and b radiation. - Pure energy that is released during alpha and beta radiation. -Represented by the symbol g


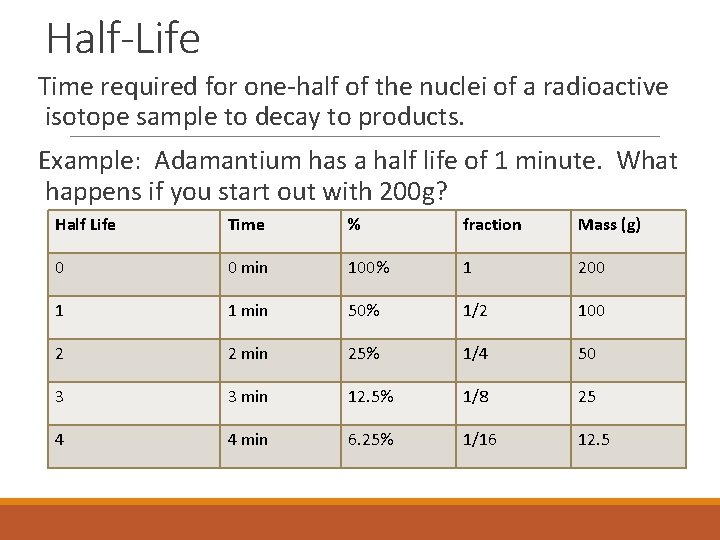
Half-Life Time required for one-half of the nuclei of a radioactive isotope sample to decay to products. Example: Adamantium has a half life of 1 minute. What happens if you start out with 200 g? Half Life Time % fraction Mass (g) 0 0 min 100% 1 200 1 1 min 50% 1/2 100 2 2 min 25% 1/4 50 3 3 min 12. 5% 1/8 25 4 4 min 6. 25% 1/16 12. 5

Learning Check 1. The half life of Carbon-14 is 5, 730 years. A sample of carbon was found at an archaeological site and found to be 17, 190 years old. If the original sample of Carbon-14 had a mass of 1, 000 g what is the current mass of the sample? Key Information Steps C-14 Half life = 5, 730 years 1. Find # of half lives Current age = 17, 190 years 2. Use # of half lives to find Original mass = 1, 000 g current mass. Find current mass Current mass = 125 g

Learning Check 2. Uranium has a half life of 4. 5 billion years. If 50 g of uranium spill into the ocean, how many years will it take for the 50 g to reduce to 0. 78 g? Key Information Steps Uranium’s half life = 4. 5 billion years 1. Find # of half lives to Amt. of Uranium spilled = 50 g reach 0. 78 g. 6 half lives Find # of years to reduce to 0. 78 g 2. Multiply that # by 4. 5 billion years x 6 = 27 billion years Uranium’s half life.