Ch 2 1 Continued Isotopes Chemical Bonds Notes

Ch 2 -1 (Continued) Isotopes & Chemical Bonds Notes #5

What are isotopes? l Same element with different atomic mass. l # of protons stays the same, # of neutrons varies. l Identified by the Atomic Mass

Example of an Isotope Carbon-12 vs. Carbon-13 Atomic Mass: 12 13 # of Protons : 6 6 # of Neutrons: 6 7

Chemical Bonds l Hold atoms together l Involves e- that are found on the outer orbital

What is a Compound? ANS: Is a substance formed by a combination of 2 or more elements Ex. H 2 O = Water 2 Hydrogen combined to 1 Oxygen
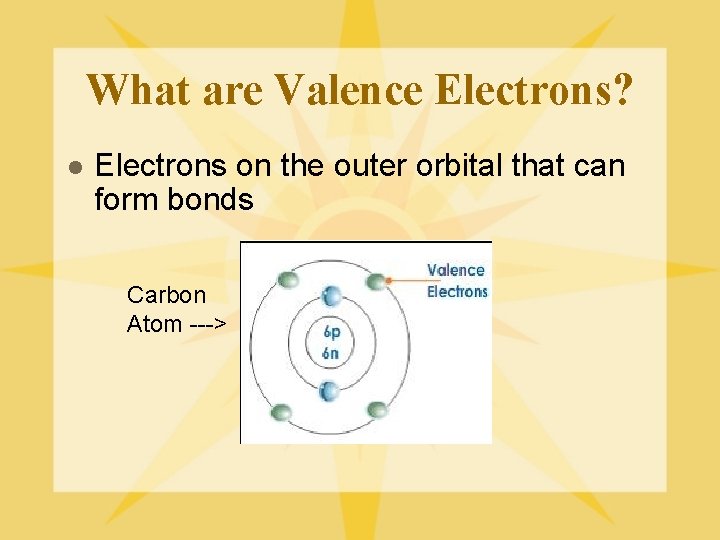
What are Valence Electrons? l Electrons on the outer orbital that can form bonds Carbon Atom --->
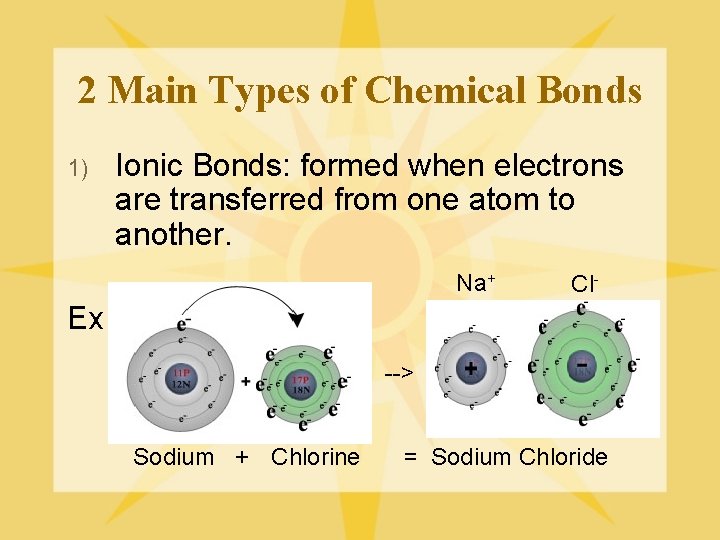
2 Main Types of Chemical Bonds 1) Ionic Bonds: formed when electrons are transferred from one atom to another. Na+ Cl- Ex. --> Sodium + Chlorine = Sodium Chloride

Result of Ionic bonds Table Salt!

What are Ions? ANS: Atoms that have gained or lost an electron 2 types a) Anion: Gained electrons; becomes more negatively charged. b) Cation: Lost electrons; becomes more positively charged. How do ions form?

Covalent Bonds l Bonds created by sharing electrons between atoms What are Molecules? -Smallest unit of compounds -Basic structure joining 2 atoms together
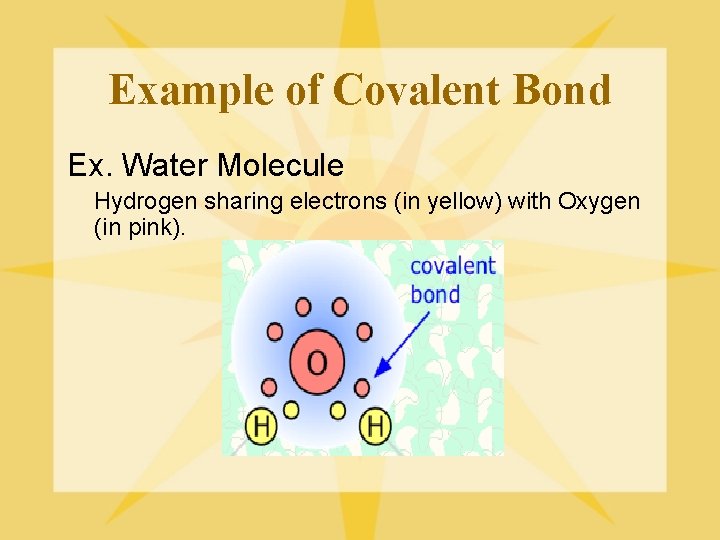
Example of Covalent Bond Ex. Water Molecule Hydrogen sharing electrons (in yellow) with Oxygen (in pink).

Ionic vs Covalent Bonding
- Slides: 12