Ch 19 dBlock Metals Hvap in k Jmol
Ch 19. d-Block Metals
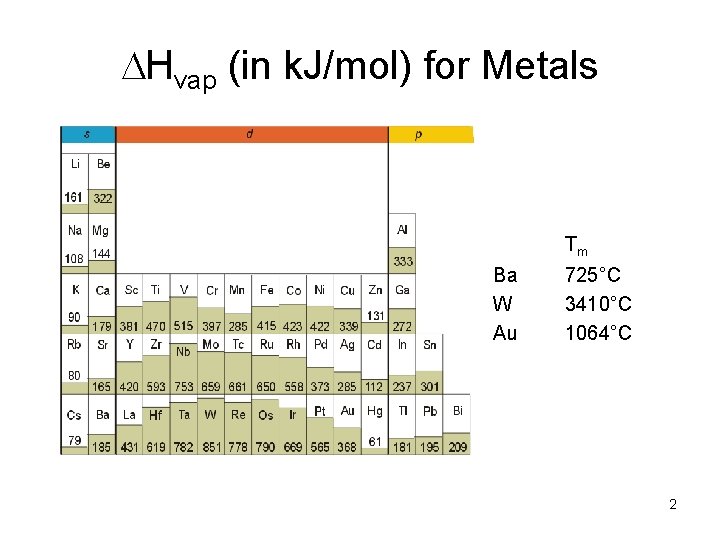
Hvap (in k. J/mol) for Metals Tm Ba W Au 725°C 3410°C 1064°C 2
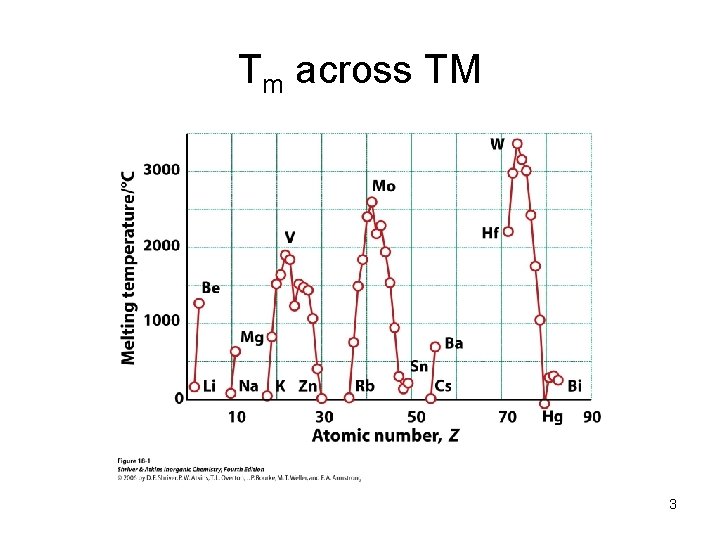
Tm across TM 3

Quick Review of Redox Rxns To balance a half-reaction: 1. 2. 3. 4. Identify and balance redox atoms Add e as needed Add H+ or OH- to balance charge Add H 2 O as needed Ex: Balance HMn. O 4 Mn 2+ in acidic soln 5 e + HMn. O 4 + 7 H+ Mn 2+ + 4 H 2 O Balance VO 43 V 2 O 3 in basic solution 4 e + 2 VO 43 + 5 H 2 O V 2 O 3 + 10 OH E = 1. 37 V at p. H = 14 4

Quick Review of Redox Rxns Nernst relation E = E - (0. 059 V / n) log Q What is E (VO 43 / V 2 O 3) at p. H = 12 ? E = E - (0. 059 V / 4) log [OH ]10 = E + (10) (0. 059 V / 4) ( p. OH) = +1. 37 V + (0. 148) (2) = +1. 66 V produced) (E increases with decr p. H because OH is 5

Quick Review of Redox Rxns Latimer diagrams 1. Reverse direction, reverse sign 2. n E are additive, not E 1. 5 -1. 18 Mn 3+ Mn 2+ Mn E = (1) (1. 5 V) + 2( 1. 18 V) / 3 = 0. 28 V 3. E is independent of stoichiometry 6

Quick Review of Redox Rxns e- + Fe 3+ → Fe 2+ E = 0. 77 V e- + Fe(OH)3 + 3 H+ → Fe 2+ + 3 H 2 O E = E 0 - 3(0. 059) p. H e- + Fe(OH)3 → Fe(OH)2 + OHE = E 0 - 0. 059 p. H 7
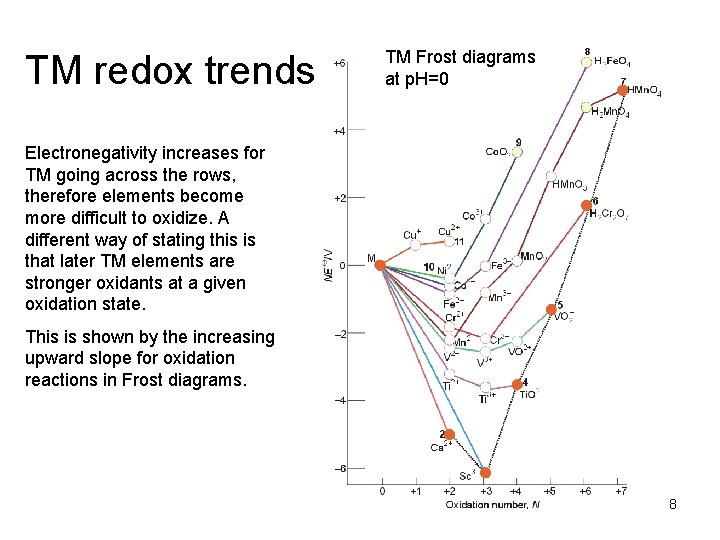
TM redox trends TM Frost diagrams at p. H=0 Electronegativity increases for TM going across the rows, therefore elements become more difficult to oxidize. A different way of stating this is that later TM elements are stronger oxidants at a given oxidation state. This is shown by the increasing upward slope for oxidation reactions in Frost diagrams. 8

TM Pourbaix diagrams show increasing E for M/M 2+ and M 2+/M 3+ equilibria 9

Early vs late TMs 2 e + Co. O 2 Co 2+ E = 1. 66 V 2 e + Ti. O 2+ Ti 2+ E = - 0. 14 V Note that Co. O 2 is unstable in H 2 O because: 2 e + 4 H+ + Co. O 2 2 H 2 O 2 Co. O 2 + 4 H+ Co 2+ + 2 H 2 O O 2 + 4 e + 4 H + 2 Co 2+ + O 2 + 2 H 2 O E = 1. 66 E = -1. 23 E = +0. 43 10

TM redox trends More valence e- going across the rows means higher oxidation states are possible, but later TM are too electronegative to be oxidized to their group number. 3 4 5 6 7 8 9 10 11 12 Sc Ti V Cr Mn Fe Co Ni Cu Zn +3 +4 +5 +6 +7 +6 +4 +2, 3 +2 Highest oxidation states accessible in aqueous solution 11
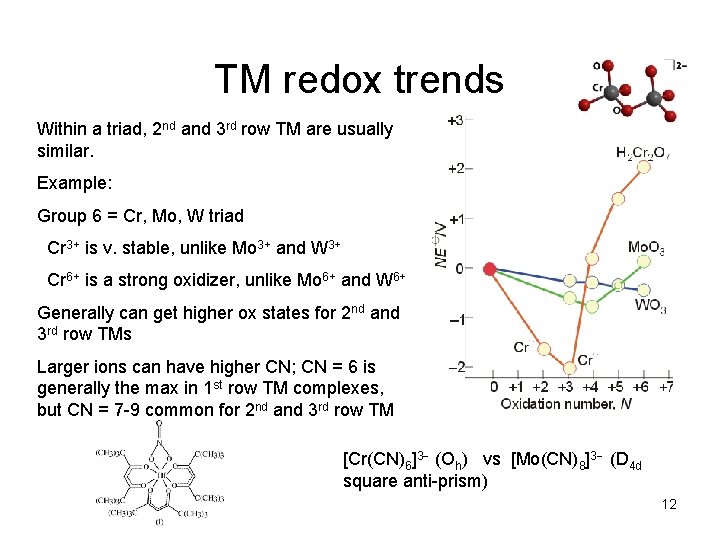
TM redox trends Within a triad, 2 nd and 3 rd row TM are usually similar. Example: Group 6 = Cr, Mo, W triad Cr 3+ is v. stable, unlike Mo 3+ and W 3+ Cr 6+ is a strong oxidizer, unlike Mo 6+ and W 6+ Generally can get higher ox states for 2 nd and 3 rd row TMs Larger ions can have higher CN; CN = 6 is generally the max in 1 st row TM complexes, but CN = 7 -9 common for 2 nd and 3 rd row TM [Cr(CN)6]3 (Oh) vs [Mo(CN)8]3 (D 4 d square anti-prism) 12
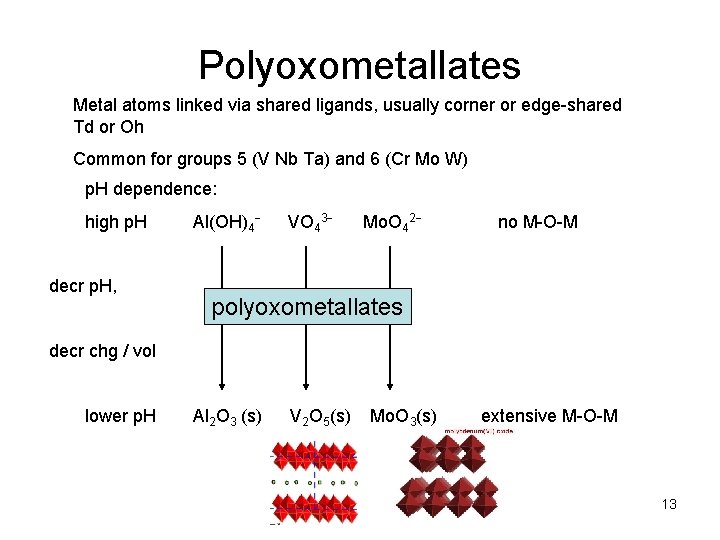
Polyoxometallates Metal atoms linked via shared ligands, usually corner or edge-shared Td or Oh Common for groups 5 (V Nb Ta) and 6 (Cr Mo W) p. H dependence: high p. H decr p. H, Al(OH)4 VO 43 Mo. O 42 no M-O-M polyoxometallates decr chg / vol lower p. H Al 2 O 3 (s) V 2 O 5(s) Mo. O 3(s) extensive M-O-M 13
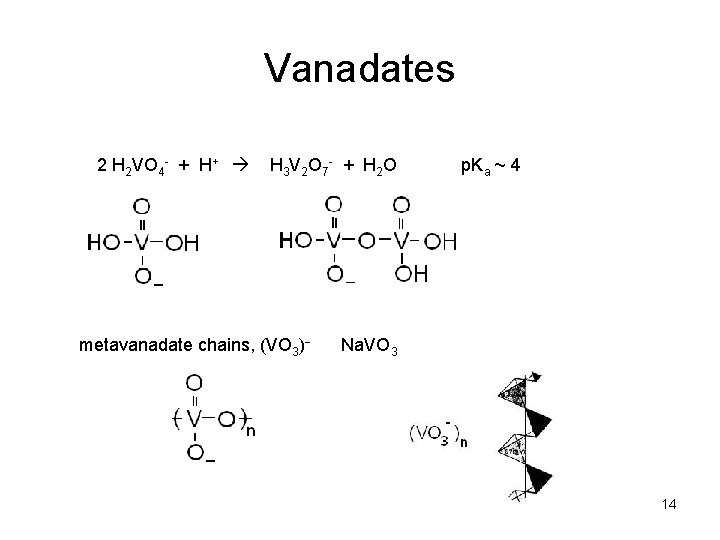
Vanadates 2 H 2 VO 4 - + H+ H 3 V 2 O 7 - + H 2 O metavanadate chains, (VO 3) p. Ka ~ 4 Na. VO 3 14
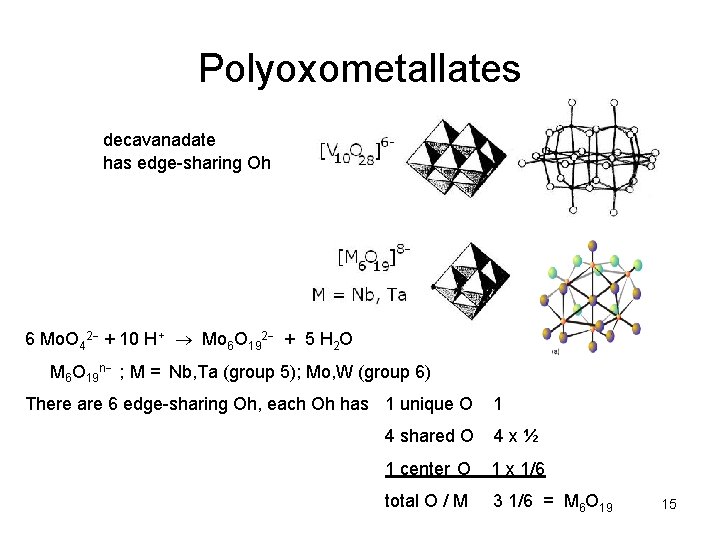
Polyoxometallates decavanadate has edge-sharing Oh 6 Mo. O 42 + 10 H+ Mo 6 O 192 + 5 H 2 O M 6 O 19 n ; M = Nb, Ta (group 5); Mo, W (group 6) There are 6 edge-sharing Oh, each Oh has 1 unique O 1 4 shared O 4 x½ 1 center O 1 x 1/6 total O / M 3 1/6 = M 6 O 19 15
![Keggin structure [PMo 12 O 40]3 Keggin structures Td site at cluster center, can Keggin structure [PMo 12 O 40]3 Keggin structures Td site at cluster center, can](http://slidetodoc.com/presentation_image_h/28017e9c07aee3db49ec2c0abadc2969/image-16.jpg)
Keggin structure [PMo 12 O 40]3 Keggin structures Td site at cluster center, can also be As, Si, B, Te, Ti PO 43 - + 12 WO 42 - + 27 H+ H 3 PW 12 O 40 + 12 H 2 O http: //en. wikipedia. org/wiki/Keggin_structure (ref Fig below) X 2 M 18 O 62 n− Dawson structure 16

Ferrodoxins 17
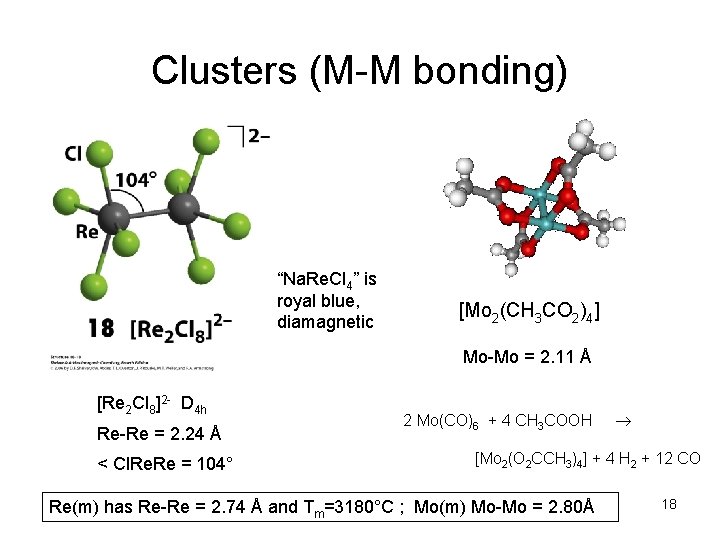
Clusters (M-M bonding) “Na. Re. Cl 4” is royal blue, diamagnetic [Mo 2(CH 3 CO 2)4] Mo-Mo = 2. 11 Å [Re 2 Cl 8]2 - D 4 h Re-Re = 2. 24 Å < Cl. Re = 104° 2 Mo(CO)6 + 4 CH 3 COOH [Mo 2(O 2 CCH 3)4] + 4 H 2 + 12 CO Re(m) has Re-Re = 2. 74 Å and Tm=3180°C ; Mo(m) Mo-Mo = 2. 80Å 18
![M-M bonding interactions [M 2 X 8]n common in groups 6 -9 (Mo, W, M-M bonding interactions [M 2 X 8]n common in groups 6 -9 (Mo, W,](http://slidetodoc.com/presentation_image_h/28017e9c07aee3db49ec2c0abadc2969/image-19.jpg)
M-M bonding interactions [M 2 X 8]n common in groups 6 -9 (Mo, W, Re, Ru, Rh) 19
![Electronic configurations Cluster ions config b. o. b. l. [Mo 2(SO 4)4]4 Mo(II) d Electronic configurations Cluster ions config b. o. b. l. [Mo 2(SO 4)4]4 Mo(II) d](http://slidetodoc.com/presentation_image_h/28017e9c07aee3db49ec2c0abadc2969/image-20.jpg)
Electronic configurations Cluster ions config b. o. b. l. [Mo 2(SO 4)4]4 Mo(II) d 4 σ 2 4 2. 11 Å [Mo 2(SO 4)4]3 Mo(II) d 4 σ 2 4 1 3. 5 2. 17 Å Mo(III) d 3 20
![Electronic configurations Cluster ions config b. o. b. l. [Mo 2(HPO 4)4]2 Mo(III) d Electronic configurations Cluster ions config b. o. b. l. [Mo 2(HPO 4)4]2 Mo(III) d](http://slidetodoc.com/presentation_image_h/28017e9c07aee3db49ec2c0abadc2969/image-21.jpg)
Electronic configurations Cluster ions config b. o. b. l. [Mo 2(HPO 4)4]2 Mo(III) d 3 σ2 4 3 2. 22 Å [Ru 2 Cl 2(O 2 CCl)4] Ru(II) d 6 Ru(III) d 5 σ2 4 2 * *2 2. 5 2. 27 Å 21

Electronic Configurations 22
![Larger Metal Clusters [Re 3 Cl 12]3 - 3 Zr(s) + Zr. Cl 4(g) Larger Metal Clusters [Re 3 Cl 12]3 - 3 Zr(s) + Zr. Cl 4(g)](http://slidetodoc.com/presentation_image_h/28017e9c07aee3db49ec2c0abadc2969/image-23.jpg)
Larger Metal Clusters [Re 3 Cl 12]3 - 3 Zr(s) + Zr. Cl 4(g) 4 Zr. Cl (s) Zr. Cl Zr-Zr bondlengths intrasheet 3. 03 Å Intersheet 3. 42 Å In Zr (m) 3. 19 Å 23
![Mo. Cl 2 and [Mo 6 Cl 14]2[Mo 6 Cl 14 ]2 - HCl Mo. Cl 2 and [Mo 6 Cl 14]2[Mo 6 Cl 14 ]2 - HCl](http://slidetodoc.com/presentation_image_h/28017e9c07aee3db49ec2c0abadc2969/image-24.jpg)
Mo. Cl 2 and [Mo 6 Cl 14]2[Mo 6 Cl 14 ]2 - HCl (aqu) Mo. Cl 2 4 of the 6 Cl bridge to other Mo 6 clusters For each Mo 6: 8 Cl capping faces 4 (½ Cl) bridging 2 Cl unique 12 Cl / Mo 6 cluster Similar for M = Mo, W, Nb, Ta 24
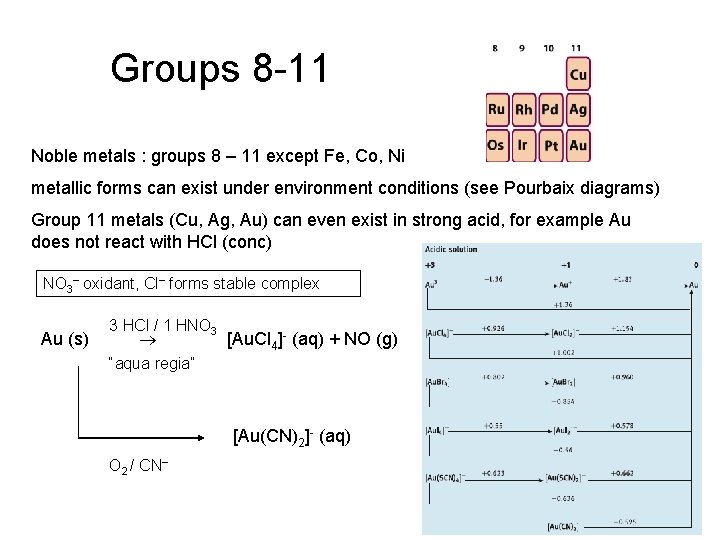
Groups 8 -11 Noble metals : groups 8 – 11 except Fe, Co, Ni metallic forms can exist under environment conditions (see Pourbaix diagrams) Group 11 metals (Cu, Ag, Au) can even exist in strong acid, for example Au does not react with HCl (conc) NO 3 oxidant, Cl forms stable complex Au (s) 3 HCl / 1 HNO 3 [Au. Cl 4]- (aq) + NO (g) “aqua regia” [Au(CN)2]- (aq) O 2 / CN 25
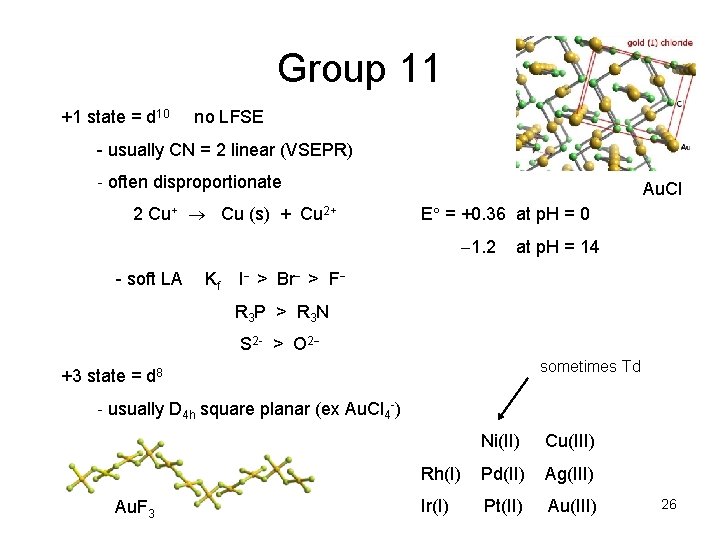
Group 11 +1 state = d 10 no LFSE - usually CN = 2 linear (VSEPR) - often disproportionate 2 Cu+ Cu (s) + Cu 2+ Au. Cl E = +0. 36 at p. H = 0 1. 2 - soft LA Kf at p. H = 14 I > Br > F R 3 P > R 3 N S 2 - > O 2 sometimes Td +3 state = d 8 - usually D 4 h square planar (ex Au. Cl 4 -) Au. F 3 Ni(II) Cu(III) Rh(I) Pd(II) Ag(III) Ir(I) Pt(II) Au(III) 26

Group 12 (Zn, Cd, Hg) Not noble metals; Zn, Cd are readily oxidized p. H = 0 Fe/Fe 2+ E 0 = + 0. 44 V Cu/Cu 2+ E 0 = 0. 34 V Zn/Zn 2+ E 0 = + 0. 76 V Zn(m) is used for anodic protection (sacrificial anode) www. boatzincs. com/shaft. html Why the aperiodic change from group 11 to 12 ? B–H approach: Cu Zn M (s) M (g) + 338 +131 k. J/mol M (g) M 2+ (g) + 2 e +2704 +2639 M (s) M 2+ (g) + 2 e +3012 +2770 27

Group 12 has d 10 s 2 filled orbitals, much weaker M–M bonding, and lower IE MP Cu 1080°C Zn 420 C Cd 320 Hg - 39 Zn 2+ common CN = 4 (6) Cd 2+ common CN = 6 (4) Hg 2+ common CN = 2 (linear) Hg 2+ is stable in aqu solution Hg. Cl – mercurous chloride (calomel) is [Hg 2]2+ 2 Cl bondlengths Raman band at 171 cm 1 Hg–Hg stretch Hg (m) 300 pm Diamagnetic (Hg+ would be d 10 s 1) Hg 22+ 250 -270 pm XRD 28
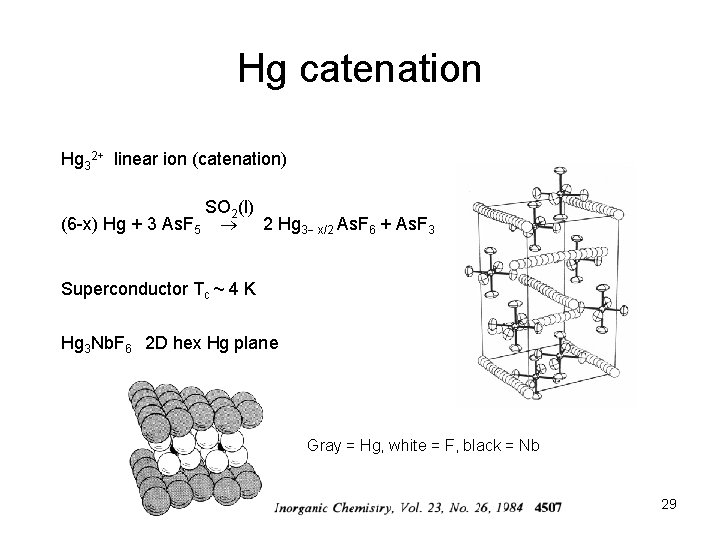
Hg catenation Hg 32+ linear ion (catenation) SO 2(l) (6 -x) Hg + 3 As. F 5 2 Hg 3 x/2 As. F 6 + As. F 3 Superconductor Tc ~ 4 K Hg 3 Nb. F 6 2 D hex Hg plane Gray = Hg, white = F, black = Nb 29

f-block elements Relatively constant electroneg across block (shielding keeps Z* = Z-σ nearly constant), so chemistry is very consistent across f-block Ions – have only f valence e Ce = [Xe]4 f 2 6 s 2 Ce 3+ = [Xe]4 f 1 Ce 4+ = [Xe] All Ln have 3+ as their most stable oxidation state Ce 4+ is relatively stable (f ) E 0 (Ce 4+/ Ce 3+) = +1. 76 V strong oxidant Eu 2+ E 0 (Eu 2+/ Eu 3+) = + 0. 35 V mild reductant “ “ “ (f 7) 30
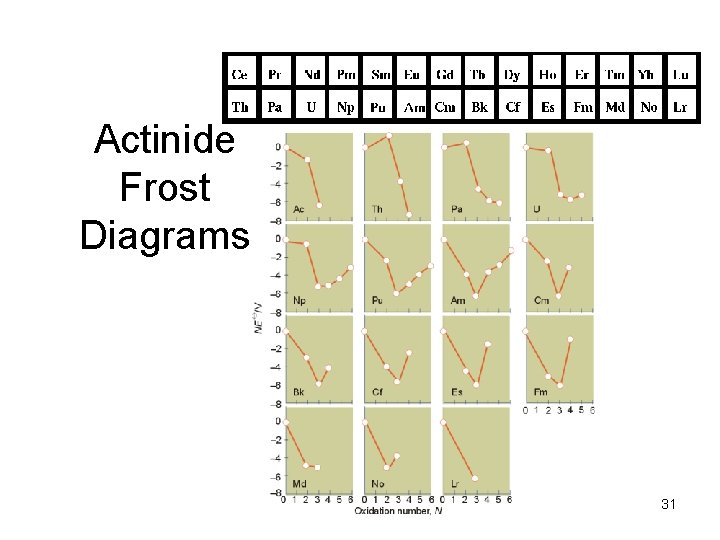
Actinide Frost Diagrams 31
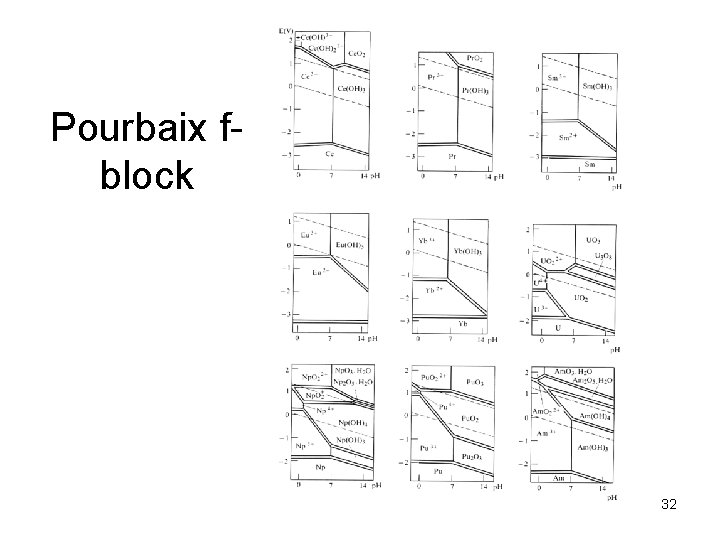
Pourbaix fblock 32

Ligand interactions f-block metal – ligand interactions: Ligands have less influence on f orbitals f–f electronic transitions are sharp, relatively independent of ligand type, and long-lived (slow non-radiative energy transfer) luminescence d–d transition forbidden (Laporte selection rules) Eu(III) 1 % gives bright orange-red luminescence Gd 2 O 2 S: Pr Gd(III) = f 7 colorless (spin forbidden transitions) Pr(III) = f 2 green 33
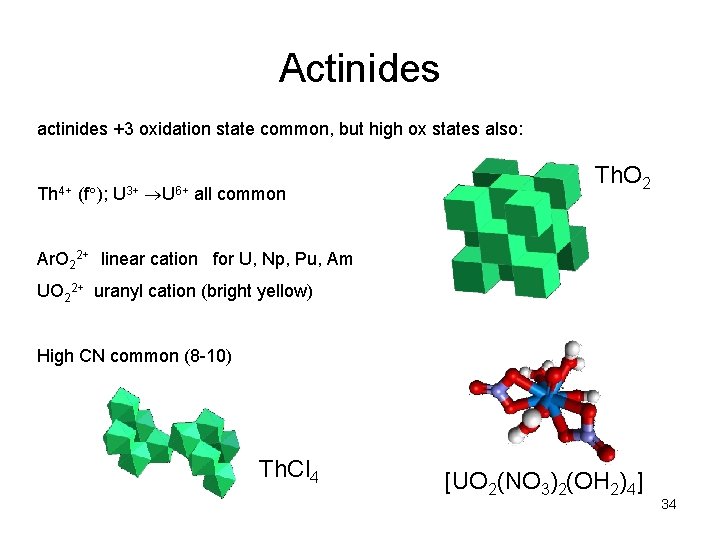
Actinides actinides +3 oxidation state common, but high ox states also: Th 4+ (f ); U 3+ U 6+ all common Th. O 2 Ar. O 22+ linear cation for U, Np, Pu, Am UO 22+ uranyl cation (bright yellow) High CN common (8 -10) Th. Cl 4 [UO 2(NO 3)2(OH 2)4] 34
- Slides: 34