Ch 15 Acids Bases l ACIDS Have a

Ch. 15 – Acids & Bases + l ACIDS – Have a H ion (Hydronium ion) l Corrosive l p. H lower than 7 l Electrolyte l Sour taste l Mix acid to water to dilute; never mix water to acid.
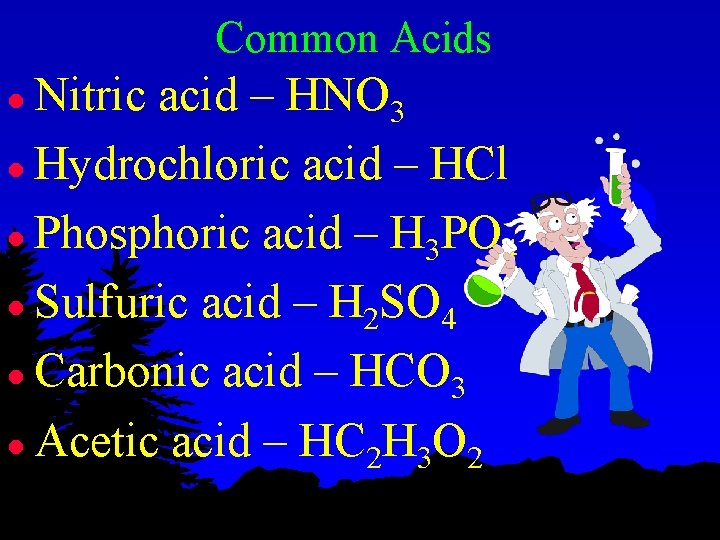
Common Acids Nitric acid – HNO 3 l Hydrochloric acid – HCl l Phosphoric acid – H 3 PO 4 l Sulfuric acid – H 2 SO 4 l Carbonic acid – HCO 3 l Acetic acid – HC 2 H 3 O 2 l

Phosphoric acid l Flavor agent in beverages l Acetic acid l 5% of vinegar l Fermentation product in juices, wines, & beer l Hydrochloric acid l Stomach acid l Used to pickle metals l

Nitric acid l Making explosives l Turns skin yellow – protein indicator l Sulfuric acid l Strongest acid l Used in car batteries l Dehydrating agent (anhydrous) l

Bases Base – has an OH- ion (hydroxide ion) l Corrosive l p. H higher than 7 l Electrolyte l Bitter taste l Pink with phenophalthene as an indicator + l H & OH ions tend to neutralize each other out Neutralization l

Common Bases Lithium hydroxide – Li. OH strong l Calcium hydroxide – Ca(OH)2 weak l Sodium hydroxide – Na. OH strong l Lye & drain cleaner l Potassium hydroxide – KOH strong l Magnesium hydroxide – Mg(OH)2 weak l Ant-acid l

A substance with OH- form a soluble base is called an alkaline. l An acid with a compound of H, O, & a third element is called an Oxyacid. l Ex. HNO 3, H 2 SO 4 l NH 4 is the only base without OH in it. l

l Basic anhydride is an oxide that reacts with water to form an alkaline solution. l A base without water l Takes water out of a substance when it comes in contact with it. l Very strong bases (lye) l The stronger the base the more anhydric it is.

l Acid anhydride is an oxide that reacts with water to form an acid. l An acid without water l Takes water out of substances when it comes in contact with them. l Very strong acids (sulfuric acid) l The stronger the acid the more anhydric it is.

ACIDS & BASES COMBINE TO FORM A SALT & WATER. l Na. OH + HCl H 2 O + Na. Cl l HCl + Li. OH H 2 O + Li. Cl l Amphoteric is any solution that can react as either an acid or a base. l
- Slides: 10