Ch 1314 Solutions Mixtures Electrolytes Solutions soluble l

Ch. 13/14: Solutions Mixtures, Electrolytes

Solutions Ø soluble l l capable of being dissolved solid gradually disappears as particles leave the crystal and mix with water Ø solution l l l homogeneous mixture of two or more substances particles cannot be seen 0. 01 to 1 nm in diameter
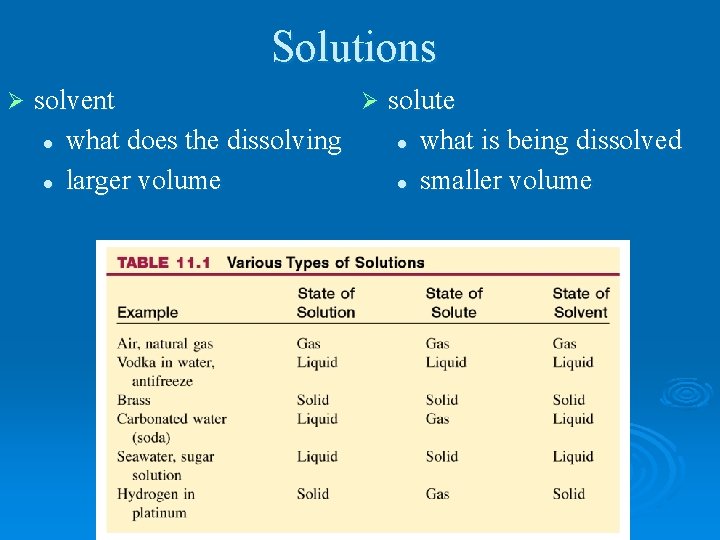
Solutions Ø solvent Ø solute l what does the dissolving l what is being dissolved l larger volume l smaller volume

Suspensions Ø a mixture with very large particles Ø so large that they will settle out if given the time Ø >1000 nm in diameter Ø can be separated using filtration Ø considered a heterogeneous mixture Ø ex. flour and water

Colloids Ø medium sized particles (1 -1000 nm) Ø in between a solution and suspension Ø particles do not settle- but stay suspended in the solution Ø can appear cloudy or clear

Colloids solution colloid Ø Tyndall Effect l l l used to differentiate between solution and colloid since they can both appear to be clear beam of light is visible in a colloid light is reflected off of the particles because of their larger size

Solutions and Colloids

Electrolytes Ø Strong Electrolytes l l completely create ions in water conduct electricity Ø Weak Electrolytes l l only partially create ions in water partially conduct electricity Ø Nonelectrolytes l l create no ions in water do not conduct electricity
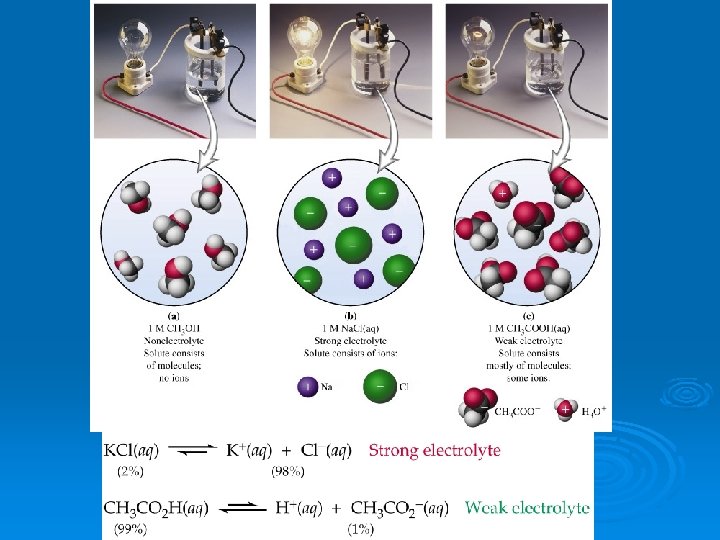

Electrolytes Ø Strong Electrolytes l l soluble ionic compounds strong acids Ø Weak Electrolytes l l water weak acids and weak bases Ø Nonelectrolytes l l molecular compounds ex: sugar

How ions are made Ø Dissociation l l when an ionic compound breaks apart into its individual ions in water Ca(NO 3)2 Ca 2+ + 2 NO 31 - Ø Ionization l l l when a molecular compound creates ions in water happens only when the bond is weak HCl H+ + Cl-

Practice Writing Equations Ø sodium phosphate l Na 3 PO 4 (s) 3 Na+ (aq) + PO 43 - (aq) Ø calcium nitrite l Ca(NO 2)2 (s) Ca 2+ (aq) + 2 NO 21 - (aq) Ø ammonium sulfate l (NH 4)2 SO 4 (s) 2 NH 4+ (aq) + SO 42 - (aq)
- Slides: 12