Ch 13 Group 13 Element properties 2 Elemental


![11 B NMR for nido [B 11 H 14]- 14 11 B NMR for nido [B 11 H 14]- 14](https://slidetodoc.com/presentation_image_h2/b2fbacb05c6f3928f85288978dcc352d/image-14.jpg)



- Slides: 20

Ch 13. Group 13

Element properties 2

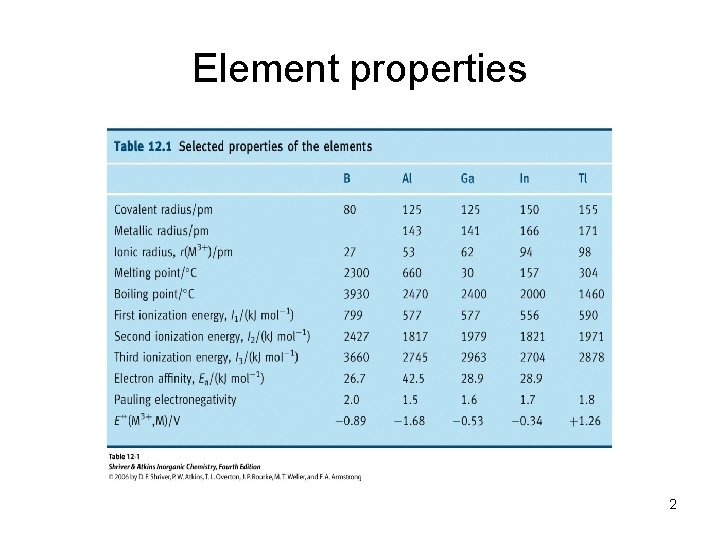
Elemental forms Boron has 3 polytypes, all contain B 12 icosahedra. Two major sources of B are in the Mohave desert, Turkey. Al to Tl All are cp metal structures (except Ga - mp 30°C, bp 2403°C) 3

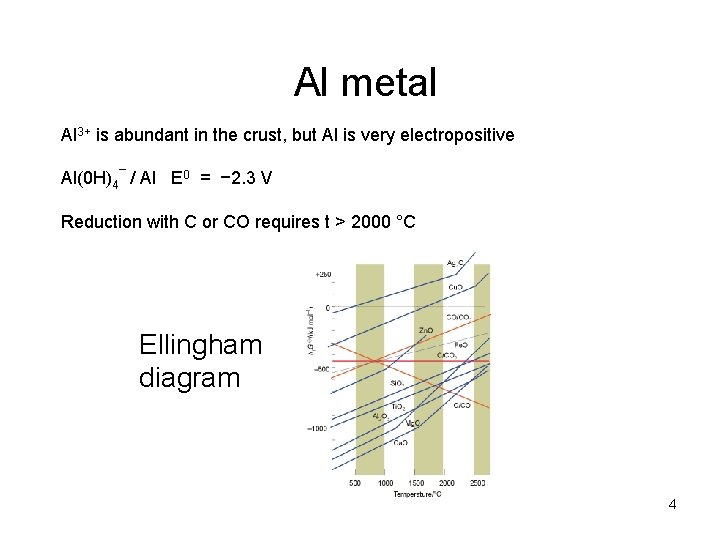
Al metal Al 3+ is abundant in the crust, but Al is very electropositive Al(0 H)4¯ / Al E 0 = − 2. 3 V Reduction with C or CO requires t > 2000 °C Ellingham diagram 4

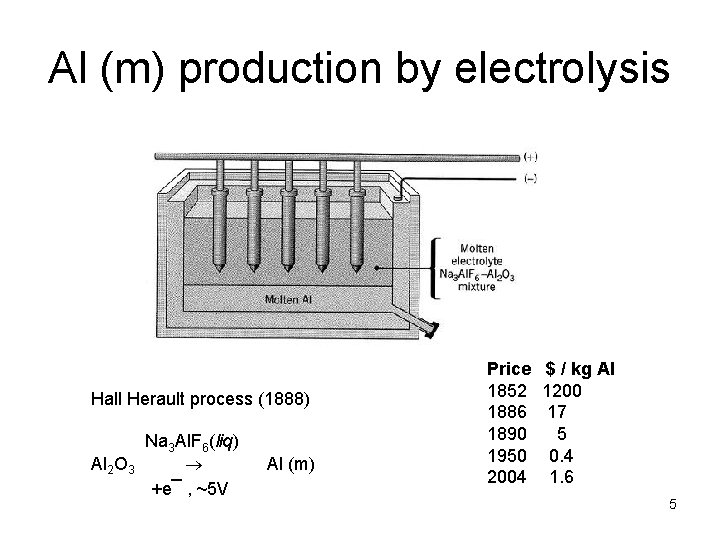
Al (m) production by electrolysis Hall Herault process (1888) Na 3 Al. F 6(liq) Al 2 O 3 +e¯ , ~5 V Al (m) Price 1852 1886 1890 1950 2004 $ / kg Al 1200 17 5 0. 4 1. 6 5

Halides BX 3 : Lewis acidity BBr 3 > BCl 3 > BF 3 This is the opposite of expected inductive effect B(s) + 3/2 F 2(g) 3 F(g) B(g) + 3 F(g) So Davg (B-F) = 583 BF 3(g) 3/2 F 2(g) B(s) BF 3(g) − 3/2 D(F-F) = − 1109 − 233 − 406 − 1748 note for comparison that D(C-F) ~ 490 k. J/mol But ΔH (BF 3(g) + F− (g) BF 4− (g)) = − 380 The lower LA is attributed to loss of π-bonding upon D 3 h Td geometry change Only BF 3 is stable in aqueous solution: BCl 3 + 3 H 2 O B(OH)3 + 3 HCl Similarly, BF 4− is stable in aqu acid, but not BCl 4−, BBr 4− Al. F 4− Td Al. F 63 − Oh (only period 3 and higher can be hypervalent 6

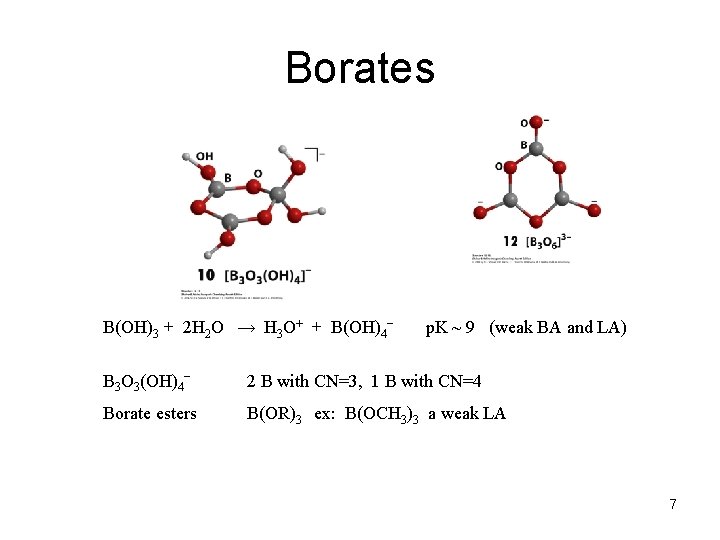
Borates B(OH)3 + 2 H 2 O → H 3 O+ + B(OH)4− p. K ~ 9 (weak BA and LA) B 3 O 3(OH)4− 2 B with CN=3, 1 B with CN=4 Borate esters B(OR)3 ex: B(OCH 3)3 a weak LA 7

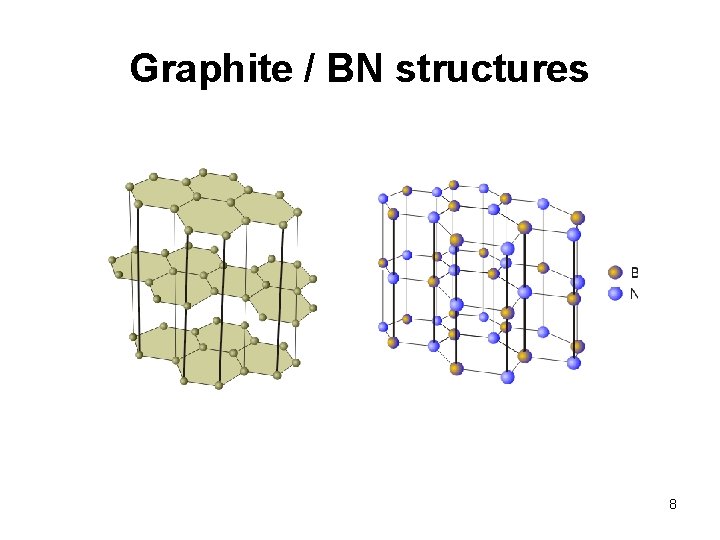
Graphite / BN structures 8

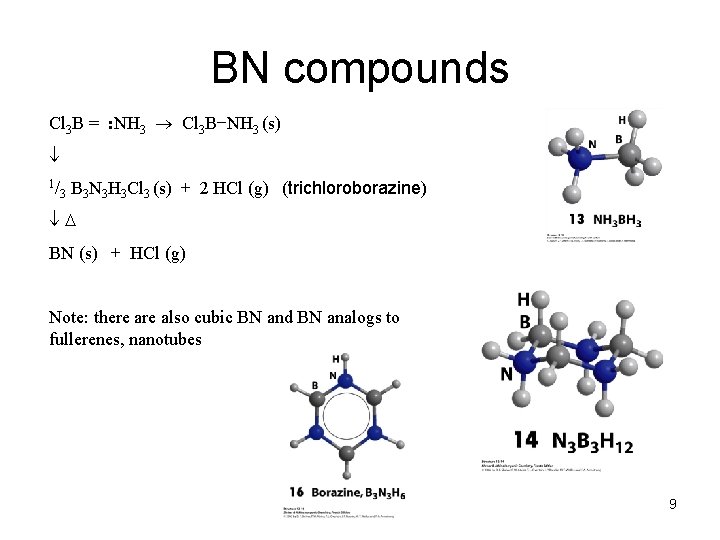
BN compounds Cl 3 B = : NH 3 Cl 3 B−NH 3 (s) 1/ 3 B 3 N 3 H 3 Cl 3 (s) + 2 HCl (g) (trichloroborazine) BN (s) + HCl (g) Note: there also cubic BN and BN analogs to fullerenes, nanotubes 9

Heavier congeners 10

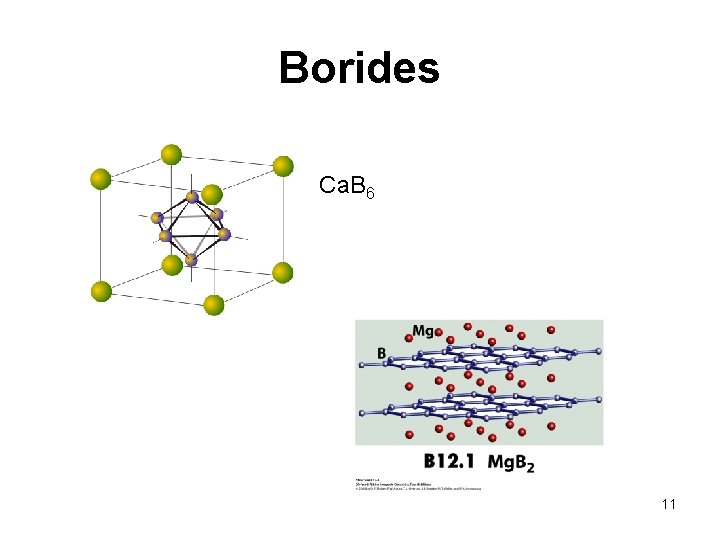
Borides Ca. B 6 Al. B 2 11

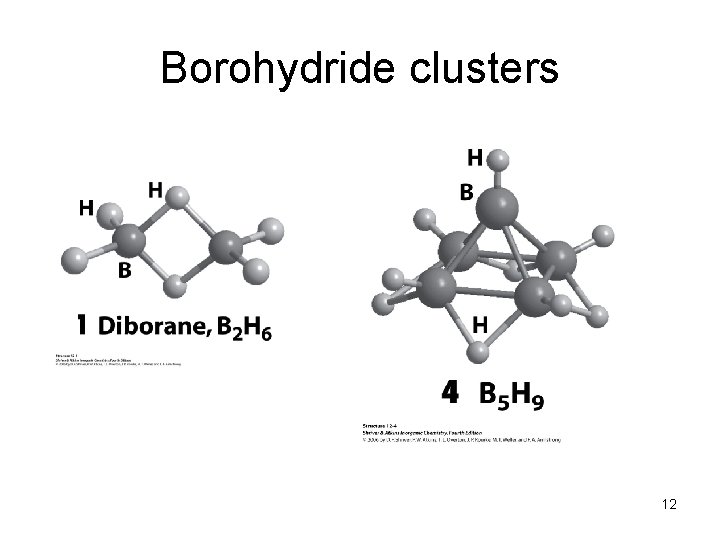
Borohydride clusters 12

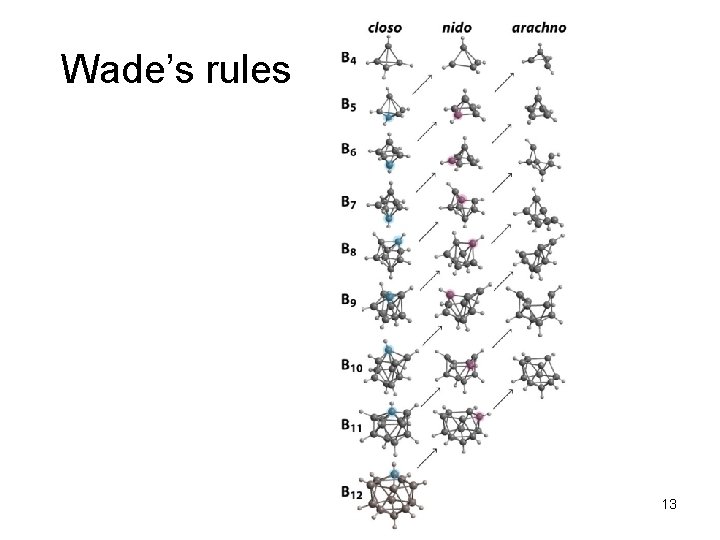
Wade’s rules 13
![11 B NMR for nido B 11 H 14 14 11 B NMR for nido [B 11 H 14]- 14](https://slidetodoc.com/presentation_image_h2/b2fbacb05c6f3928f85288978dcc352d/image-14.jpg)
11 B NMR for nido [B 11 H 14]- 14

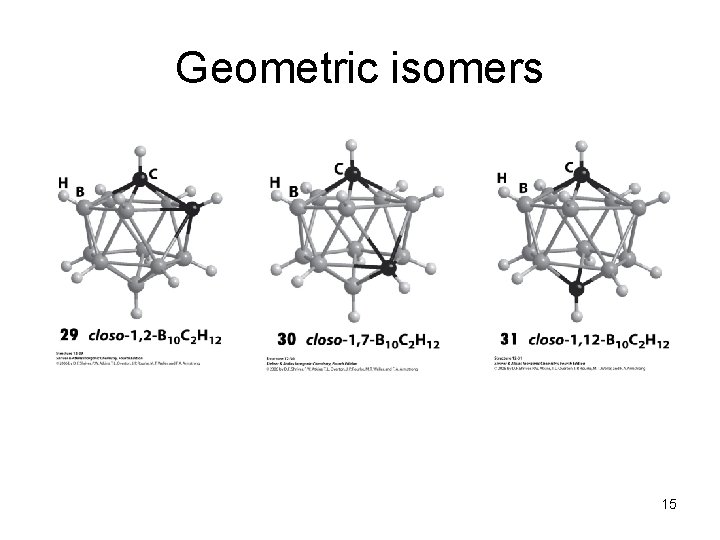
Geometric isomers 15

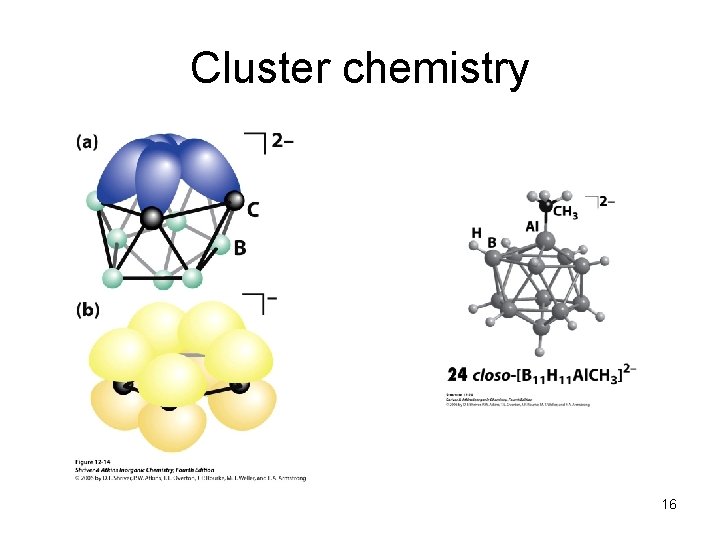
Cluster chemistry 16

Stack compounds 17

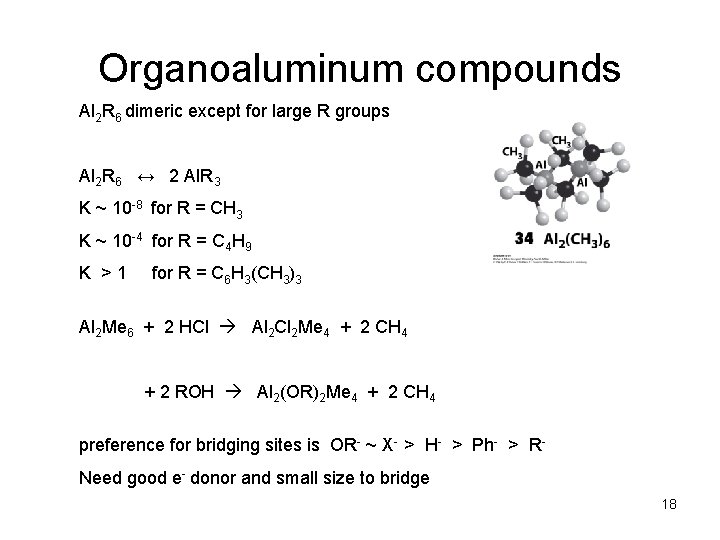
Organoaluminum compounds Al 2 R 6 dimeric except for large R groups Al 2 R 6 ↔ 2 Al. R 3 K ~ 10 -8 for R = CH 3 K ~ 10 -4 for R = C 4 H 9 K >1 for R = C 6 H 3(CH 3)3 Al 2 Me 6 + 2 HCl Al 2 Cl 2 Me 4 + 2 CH 4 + 2 ROH Al 2(OR)2 Me 4 + 2 CH 4 preference for bridging sites is OR- ~ X- > H- > Ph- > RNeed good e- donor and small size to bridge 18

Organometallic compounds Δ Al(iso-C 4 H 9)3 Al 2 H 2(iso-C 4 H 9)4 + 2 H 2 C=C(CH 3)2 alkene elimination 3 Al 2 Cl 2 Me 4 + 6 Na 2 Al 2 Me 6 + 2 Al + 6 Na. Cl Tl(CH 3)3 Tl(CH 3)2+ is linear, stable in aqu base, isoelectronic w/ Hg(CH 3)2 (η 5 -C 5 H 5)Tl is air stable monomeric in gas phase, chain structure in solid state 19

Ziegler-Natta catalysis ½ Al 2 R 6 + C 2 H 4 R 2 Al(CH 2 CH 2)R + C 2 H 4 R 2 Al(CH 2)2 R etc. . 3 n C 2 H 4 Al 2 R 6 H 2 O, Na. OH Al[(CH 2)n(CH 2 CH 3]3 n=7 -10 detergents, soaps Al 2 Et 6, Ti. Cl 4 n C 2 H 4 heptane (C 2 H 4)n (HDPE) n CH 2=C(H)CH 3 (CH 2 C(H)CH 3)n Na. O(CH 2)n. CH 3 PP (isotactic) HDPE = 50 x 106 tons/yr ≈10 lbs / person ≈500 x 106 barrels oil / yr (US consumes 720 x 106 barrels / day) 20