Ch 13 Gases Gas Stoichiometry Stoichiometry Steps Review










- Slides: 13

Ch. 13 – Gases Gas Stoichiometry

* Stoichiometry Steps Review * 1. Write a balanced equation. 2. Identify known & unknown. 3. Line up conversion factors. Mole ratio – moles • • Mole moles • Molar mass – moles grams • Molar volume – moles liters gas • Molarity – moles liters soln Core step in all stoichiometry problems!! 4. Check answer.

* Molar Volume at STP 1 mol of a gas=22. 4 L at STP Standard Temperature & 0°C and 1 atm Pressure

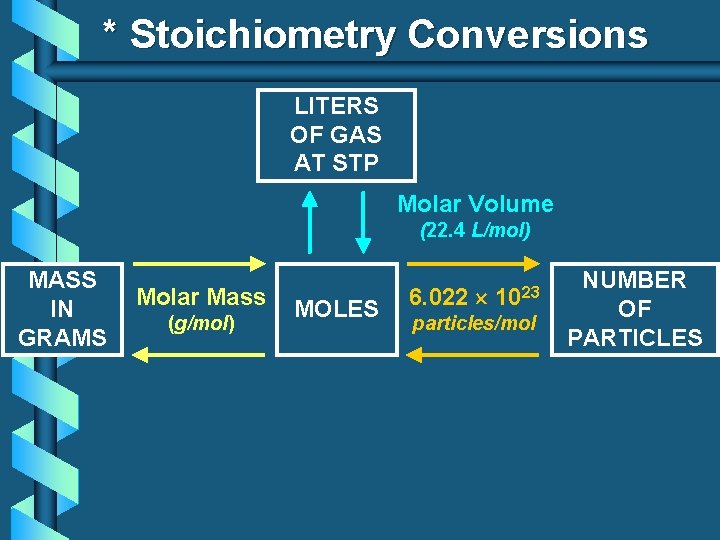
* Stoichiometry Conversions LITERS OF GAS AT STP Molar Volume (22. 4 L/mol) MASS IN GRAMS Molar Mass (g/mol) MOLES 6. 022 1023 particles/mol NUMBER OF PARTICLES

A. Gas Stoichiometry – Volume of known gas Liters of an unknown gas: • Avogadro’s Principle Ø Equal volumes of gases contain equal numbers of moles • Coefficients give mole ratios and volume ratios A. Liters

A. Gas Stoichiometry – Volume A. What volume of oxygen is needed for the complete combustion of 4. 00 L of propane (C 3 H 8)? C 3 H 8 + 5 O 2 3 CO 2+ 4 H 2 O 4. 00 L ? L 4. 00 L 5 L C 3 H 8 O 2 1 L C 3 H 8 = 20. 0 L O 2

B. Gas Stoichiometry – STP B. Moles or grams of one substance Liters of a Gas: • Pressure and temperature will change the volume of a gas, so these need to be known to calculate amounts of reactants needed and products produced • STP – use 22. 4 L/mol

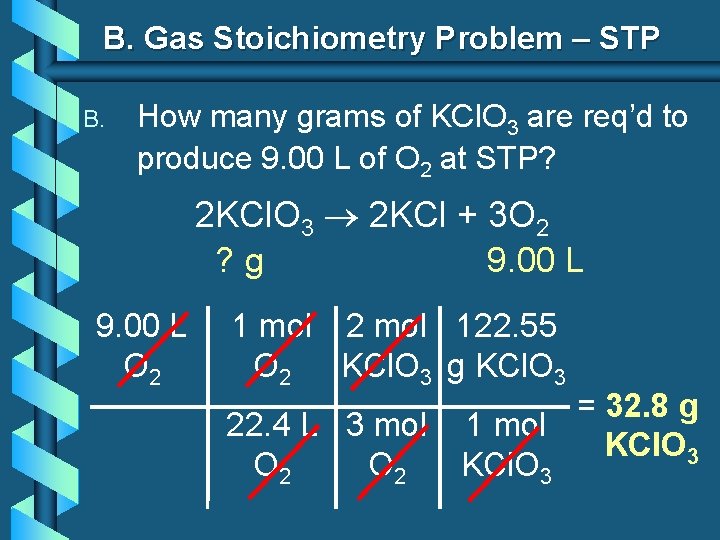
B. Gas Stoichiometry Problem – STP B. How many grams of KCl. O 3 are req’d to produce 9. 00 L of O 2 at STP? 2 KCl. O 3 2 KCl + 3 O 2 ? g 9. 00 L O 2 1 mol 2 mol 122. 55 O 2 KCl. O 3 g KCl. O 3 22. 4 L 3 mol O 2 = 32. 8 g 1 mol KCl. O 3

C. Gas Stoichiometry – Non-STP C. Non-STP 1. Given liters of gas? Ø start with ideal gas law 2. Looking for liters of gas? Ø start with stoichiometry conversion

C. Gas Stoichiometry Problem – Non-STP C. 1 How many grams of Al 2 O 3 are formed from 15. 0 L of O 2 at 97. 3 k. Pa & 21°C? 4 Al(s) + GIVEN: P = 97. 3 k. Pa V = 15. 0 L n=? T = 21°C = 294 K R = 8. 315 L k. Pa/mol K 3 O 2(g) 15. 0 L non. STP WORK: 2 Al 2 O 3(s) ? g Given liters: Start with Ideal Gas Law and calculate moles of O 2. PV = n. RT (97. 3 k. Pa) (15. 0 L) = n (8. 315 L k. Pa/mol K) NEXT (294 K)

C. Gas Stoichiometry Problem – Non-STP C. 1 How many grams of Al 2 O 3 are formed from 15. 0 L of O 2 at 97. 3 k. Pa & 21°C? + 3 O 2(g) Use stoich to convert moles 15. 0 L of O to grams Al O non 0. 597 2 mol STP 101. 96 mol O 2 Al 2 O 3 g Al 2 O 3 4 Al(s) 2 2 2 Al 2 O 3(s) ? g 3 3 mol O 2 = 40. 6 g 1 mol Al 2 O 3

C. Gas Stoichiometry Problem – Non-STP C. 2 What volume of CO 2 forms from 5. 25 g of Ca. CO 3 at 103 k. Pa & 25ºC? Ca. CO 3(s) 5. 25 g Ca. O(s) Looking for liters: Start with stoich and calculate moles of CO 2. 5. 25 g 1 mol Ca. CO 3 CO 2 + CO 2 (g) ? L non. P = 103 k. Pa STP NEXT V=? n=? = 0. 0525 mol R = 8. 315 3 k. Pa/mol. K 100. 09 1 mol Plug this dmthe into CO 2 Ideal Gas Law for n to find liters g Ca. C T = 298 K

C. Gas Stoichiometry Problem – Non-STP C. 2 GIVEN: What volume of CO 2 forms from 5. 25 g of Ca. CO 3 at 103 k. Pa & 25ºC? WORK: P = 103 k. Pa PV = n. RT V=? (103 k. Pa)V =(0. 0525 mol)(8. 315 n = 0. 0525 mol T = 25°C = 298 K L k. Pa/mol K) (298 K) R = 8. 315 L k. Pa/mol K V = 1. 26 L CO 2