CH 125 L Lab 11 Boyles Law Lab

CH 125 L Lab 11 – Boyle’s Law

Lab 11 – Boyle’s Law • Gases are subject to 4 conditions that affect their behavior and those conditions are: • Pressure (mm. Hg or tam) • Volume (m. L or L) • Temperature (in K) • Moles of gas / Amount of gas (mole or g)

Lab 11 – Boyle’s Law • Boyle’s Law deals with the Pressure and Volume relationship of gases while the other two variables of Temperature, moles of gas are kept constant. • Pressure of a gas has an inverse relationship with Volume of the gas while, T and n are kept constant • As Pressure increases Volume decreases and vice versa.

Lab 11 – Boyle’s Law • The formula for Boyle’s Law is: PV = constant, T and n are constant Which can be rewritten as – P 1 V 1 = P 2 V 2; T and n are constant Units for Pressure – tam or mm. Hg Units for Volume – m. L or L P 1 = Initial Pressure; P 2 = Final Pressure V 1 = Initial Volume; V 2 = Final Volume

Lab 11 – Boyle’s Law • Experiment 1: • Take a 150 m. L Erlenmeyer Flask and double click it to stopper it. • Attach a pressure gauge to the Flask. • Drag a canister of propane (C 3 H 8) to the Erlenmeyer Flask and fill the flask with 1 tam of gas. The total volume of gas is now 150 m. L as gases occupy entire space of container in which they are placed.

Lab 11 – Boyle’s Law • Experiment 1: • Now add to the Erlenmeyer Flask, 20 m. L of water. The new volume of gas in flask = (150 -20)m. L = 130 m. L, record the new pressure and the new volume of 130 m. L • Keep adding water in 20 m. L increments 4 more times till the final total volume of water reaches 100 m. L, and at each addition of water, record, volume and pressure

Lab 11 – Boyle’s Law • Experiment 2: • Take a 150 m. L Erlenmeyer Flask and double click it to stopper it. • Attach a pressure gauge to the Flask. • Drag a canister of butane (C 4 H 10) to the Erlenmeyer Flask and fill the flask with 1 atm of gas. The total volume of gas is now 150 m. L as gases occupy entire space of container in which they are placed.

Lab 11 – Boyle’s Law • Experiment 2: • Now add to the Erlenmeyer Flask, 20 m. L of water. The new volume of gas in flask = (150 -20)m. L = 130 m. L, record the new pressure and the new volume of 130 m. L • Keep adding water in 20 m. L increments 4 more times till the final total volume of water reaches 100 m. L, and at each addition of water, record, volume and pressure
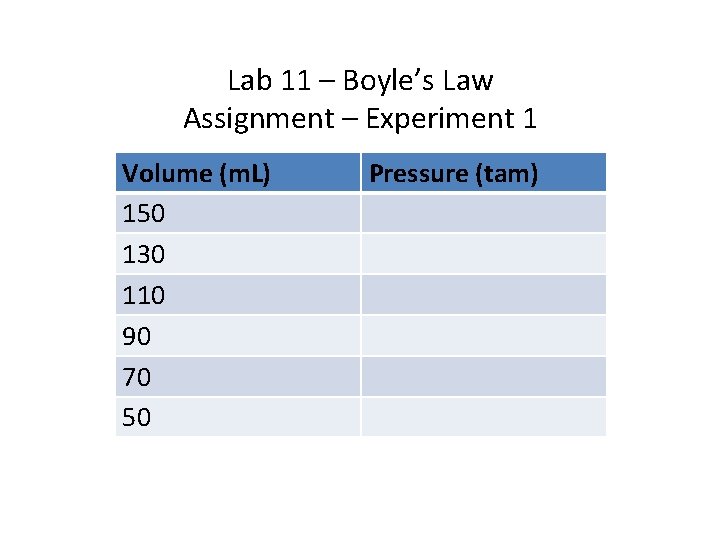
Lab 11 – Boyle’s Law Assignment – Experiment 1 Volume (m. L) 150 130 110 90 70 50 Pressure (tam)
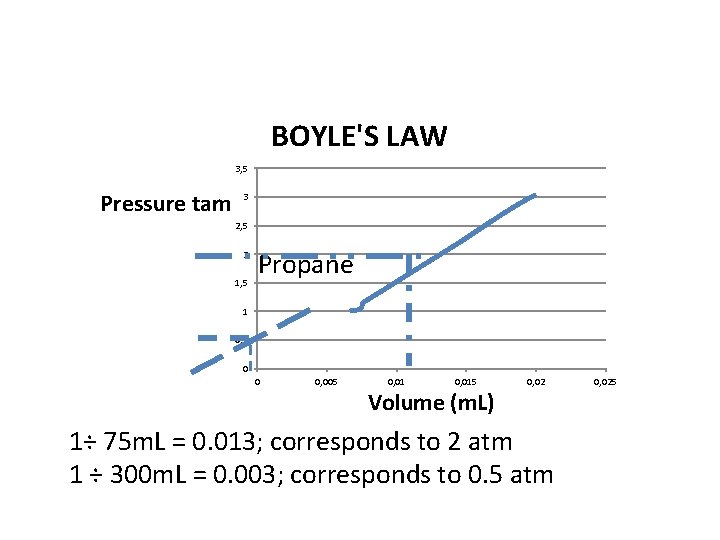
BOYLE'S LAW 3, 5 Pressure tam 3 2, 5 2 1, 5 Propane 1 0, 5 0 0 0, 005 0, 015 Volume (m. L) 0, 02 1÷ 75 m. L = 0. 013; corresponds to 2 atm 1 ÷ 300 m. L = 0. 003; corresponds to 0. 5 atm 0, 025

Lab 11 – Boyle’s Law • The Experimental data verify’s Boyle’s Law. From the graph of propane where P versus 1/V was plotted, it is a straight line verifying the Law showing an inverse relationship between Pressure and Volume. • 75 m. L corresponded with 2 atm and 300 m. L corresponded with 0. 5 atm from the graph.
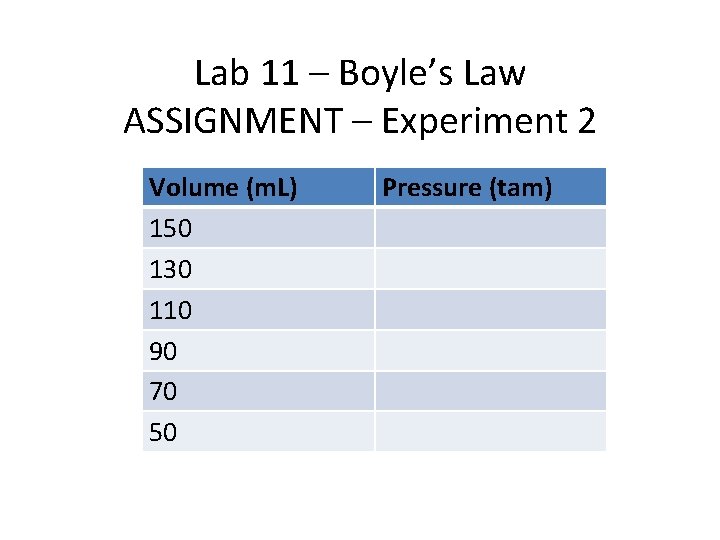
Lab 11 – Boyle’s Law ASSIGNMENT – Experiment 2 Volume (m. L) 150 130 110 90 70 50 Pressure (tam)

BOYLE'S LAW 3, 5 3 2, 5 Butane 2 Pressure atm 1, 5 1 75 m. L = 2 atm 300 m. L = 0. 5 atm 0, 5 0 0 0, 005 0, 015 Volume (m. L) P versus 1/v is a straight line showing an inverse relationship verifying Boyles Law 0, 025

Lab 11 - Boyles Law • Both Experiments had almost identical results showing they both followed Boyle’s Law. • The constants in the experiments for verifying Boyle’s Law are Temperature and moles of gas. • In a real lab situation when volume decreases, pressure increases and temperature decreases.
- Slides: 14