Ch 11 Chemical Reactions 11 1 Describing Chemical

Ch. 11 Chemical Reactions 11. 1 Describing Chemical Reactions

I. Equation Basics • A. Fe(s) + O 2(g) Fe 2 O 3(s) Reactants Products • B. Skeleton equation: only indicates chemicals involved, not relative amounts • C. Symbols for states of matter: • solid (s) gas (g) liquid (l) aqueous (aq) Substance dissolved in water • D. Catalyst: substance that speeds up rate of reaction

II. Balancing Equations • A. Atoms are never lost or gained in a reaction, they are just rearranged. • B. Guidelines: – 1. Only use coefficients (#’s in front of substances) – 2. Must have same # of each element on each side – 3. Equation must be reduced
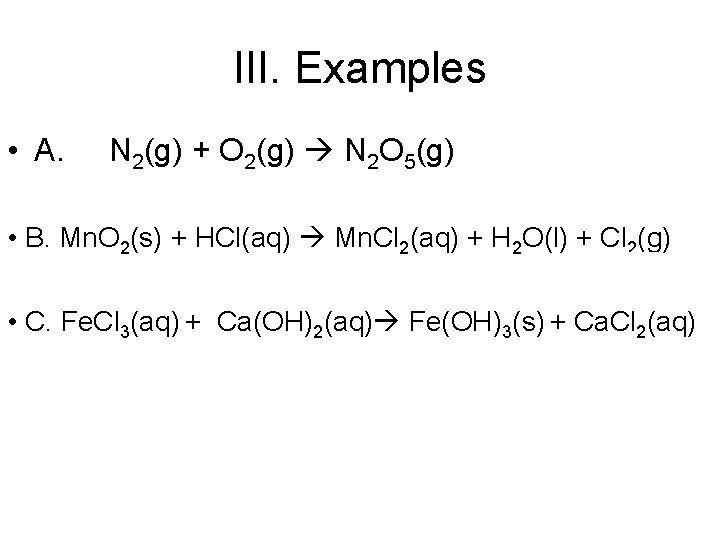
III. Examples • A. N 2(g) + O 2(g) N 2 O 5(g) • B. Mn. O 2(s) + HCl(aq) Mn. Cl 2(aq) + H 2 O(l) + Cl 2(g) • C. Fe. Cl 3(aq) + Ca(OH)2(aq) Fe(OH)3(s) + Ca. Cl 2(aq)

11. 2 Types of Chemical Reactions

I. Combination/Synthesis Rxns • A. Two or more substances combine to one • B. Na(s) + Cl 2(g) --> Na. Cl(s) + ( ) • C. If metal combines with non-metal, ionic compound produced • D. If non-metals combined, covalent compound produced

II. Decomposition Rxns • A. Single compound broken into two or more products Heat • B. Hg. O(s) Hg(l) + O 2(g) Means Heat Added

• C. Most decomp. rxns require energy (heat) as a catalyst • H 2 O 2(l) H 2 O(l) + O 2(g) • ***Demo: NI 3(s) N 2(g) + I 2(s) ***

III. Single-Replacement • A. Atoms of one element replace atoms of a second element in a compound • B. Mg + Zn(NO 3)2 Mg(NO 3)2 + Zn + + • C. Reactivity of metals determines whether one atom will replace another • D. Mg more reactive than Zn, removes nitrate ***Demo Thermite Rxn***

11. 3 Reactions in Aqueous Solution

I. Double Replacement Rxns • A. Two atoms switch places (often forming solid) • B. K 2 CO 3 (aq) + Ba. Cl 2 (aq) 2 KCl (aq) + Ba. CO 3 (s) • *** Demo: Silver nitrate and sodium chloride***

II. Net Ionic Equations • A. Equation showing only particles that take part in reaction • B. Process: 1. Start with full equation Ag. NO 3(aq) + Na 2 S(aq) Ag 2 S(s) + Na. NO 3(aq) • C. 2. Separate ions in aqueous form Ag+(aq) + NO 3 -(aq) + Na+(aq) + S 2 -(aq) Ag 2 S(s) + Na+(aq) + NO 3 -(aq)

• D. 3. Cross off ions appearing on both sides of reaction as aqueous Ag+(aq) + NO 3 -(aq) + Na+(aq) + S 2 -(aq) Ag 2 S (s) + Na+(aq) + NO 3 - (aq) • E. 4. Balance everything left to get… Net Ionic equation: 2 Ag+(aq) + S 2 -(aq) Ag 2 S(s)

Example • Write the balanced net ionic equation for the following: Step 1 Zn(s) + HCl(aq) Zn. Cl 2(aq) + H 2(g) Step 2 Zn(s) + H+(aq) + Cl-(aq) Zn 2+(aq) + Cl-(aq) + H 2(g) Step 3 Zn(s) + 2 H+(aq) Step 4 Zn 2+(aq) + H 2(g)

III. Precipitates • A. Solids formed when two aqueous mixtures form insoluble (“non-dissolvable”) compound Solubility Rules: • Always Soluble: Alkali metals (1 st column), NH 4+, NO 3 -, Cl. O 4 -, C 2 H 3 O 2 • Mostly Soluble: Cl-, Br-, I- (except Ag+, Pb 2+, Hg 22+) F- (except Ca 2+, Ba 2+, Sr 2+, Pb 2+, Mg 2+) SO 42 - (except Ca 2+, Ba 2+, Sr 2+, Pb 2+) • Mostly Insoluble: O 2 -, OH- (except w/ alkali metals, NH 4+, Ca 2+, Sr 2+, Ba 2+ somewhat soluble) CO 32 -, PO 43 -, S 2 -, SO 32 -, C 2 O 42 -, Cr. O 42 - (except w/ alkali metals, NH 4+)
- Slides: 15