CH 104 DETERMINATION OF A SOLUBILITY PRODUCT CONSTANT

CH 104: DETERMINATION OF A SOLUBILITY PRODUCT CONSTANT • A solution is saturated if it has a solid at equilibrium with its solute. The concentration of solute in a saturated solution is the largest that is normally possible. • For a saturated solution of ionic solid in water: Cx. Ay(s) = x. Cn+(aq) + y. Am–(aq) • Solubility Product Constant = Ksp = [Cn+]x[Am–]y • Where • Cx. Ay(s) is a slightly soluble ionic solid. • [Cn+] and [Am–] are the equilibrium concentrations of ions in moles per liter. • x and y are the stoichiometric coefficients from the balance reaction. • The solubility product constant (Ksp) is the equilibrium constant for the dissolution of a slightly soluble ionic compound at a specified temperature. • By convention the “concentration” of the solid, Cx. Ay, is NOT used to calculate Ksp. (That is, the activity of a pure solid is 1. )

SOLUBILITY PRODUCT CONSTANT • What is the reaction for a saturated solution of Bi 2 S 3(s) in water? Bi 2 S 3(s) = 2 Bi 3+(aq) + 3 S 2–(aq) • What is the Ksp for this reaction? Ksp = [Bi 3+]2[S 2–]3 • Notice the “concentration” of Bi 2 S 3(s) is NOT used to calculate Ksp. • What is the reaction for a saturated solution of Ag 2 Cr. O 4(s) in water? Ag 2 Cr. O 4(s) = 2 Ag+(aq) + Cr. O 42–(aq) • What is the Ksp for this reaction? Ksp = [Ag+]2[Cr. O 42–]

SOLUBILITY PRODUCT CONSTANT • Calcium fluoride (Ca. F 2) is slightly soluble in water. In a saturated solution the Ca. F 2(s) is dissolving at the same rate that Ca 2+(aq) and F–(aq) crystallize. That is, the solid and solute are at equilibrium. Ca. F 2(s) = Ca 2+(aq) + 2 F–(aq)

CALCULATING SOLUBILITY FROM Ksp • A saturated solution is made by adding excess Ca. F 2(s) to distilled water. What is the solubility of this Ca. F 2(s) at 25° C? • Step #1: Write the balanced reaction and Ksp equation. Ca. F 2(s) = Ca 2+(aq) + 2 F–(aq) Ksp = [Ca 2+][F–]2 = 2. 7 x 10– 11 at 25° C • Step #2: The initial concentrations of Ca 2+(aq) and F–(aq) are 0. The equilibrium concentrations of Ca 2+(aq) and F–(aq) are given algebraic variables based on the stoichiometric coefficients from the balance reaction. Write these equilibrium concentrations of Ca 2+(aq) and F–(aq). [Ca 2+] = x [F–] = 2 x

CALCULATING SOLUBILITY FROM Ksp • Step #3: Use the Ksp equation to solve for [Ca 2+] and [F–]. Ksp = 2. 7 x 10– 11 = [Ca 2+][F–]2 = (x)(2 x)2 = 4 x 3 = 2. 7 x 10– 11 / 4 = 6. 75 x 10– 12 [F–] = 2 x = 3. 8 x 10– 4 M • Step #4: Solve for the solubility of Ca. F 2(s). One mole of Ca 2+(aq) is produced for every mole of Ca. F 2(s) that dissolves; therefore, the solubility of Ca. F 2(s) = [Ca 2+] = 1. 9 x 10– 4 M.

THE COMMON ION EFFECT • In the previous example the pure solid (Ca. F 2(s)) was the only source of its dissolved ions (Ca 2+(aq) and F–(aq)). • However, if the common ion F–(aq) is added it will react with Ca 2+(aq) to decrease the solubility of Ca. F 2(s). The new concentration of Ca 2+(aq) is less than in the original equilibrium. And the new concentration of F–(aq) is greater than in the original equilibrium. This is called Le Châtelier’s principle. Ca. F 2(s) = Ca 2+(aq) + 2 F–(aq) Ksp = [Ca 2+][F–]2 = 2. 7 x 10– 11 at 25° C • Similarly, if the common ion Ca 2+(aq) is added it will react with F–(aq) to decrease the solubility of Ca. F 2(s). The new concentration of F–(aq) is less than in the original equilibrium. And the new concentration of Ca 2+(aq) is greater than in the original equilibrium.

THE COMMON ION EFFECT • The common ion F–(aq) is added to a saturated solution of Ca. F 2(s) in distilled water. What is the concentration of Ca 2+(aq) in equilibrium with 1. 0 M F–(aq) and Ca. F 2(s) at 25° C? Ksp = 2. 7 x 10– 11 = [Ca 2+][F–]2 = [Ca 2+](12) [Ca 2+] = 2. 7 x 10– 11 M • Compared to the previous example, did the concentration of Ca 2+(aq) increase or decrease? • It decreased from 1. 9 x 10– 4 M to 2. 7 x 10– 11 M. • Did the concentration of F–(aq) increase or decrease? • It increased from 3. 8 x 10– 4 M to 1. 0 M. • Does this agree with the common ion effect? • Yes. The concentration of Ca 2+(aq) decreased. The concentration of F–(aq) increased. And the solubility of Ca. F 2(s) decreased.

THE SALT EFFECT • Common ions decrease the solubility of ionic solids. • In contrast, the presence of “uncommon” ions tends to increase solubility of ionic solids. This is called the “salt effect”, the “uncommon ion effect”, or the “diverse ion effect”. • Soluble uncommon ions increase the interionic attractions of a solution. As a result, these uncommon ions decrease the effective concentrations (or activities) of other solutes and increase the solubility of ionic solids.
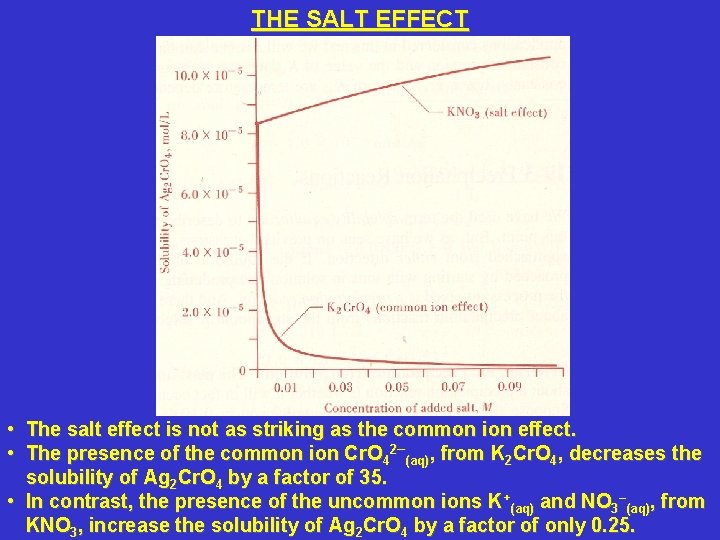
THE SALT EFFECT • • The salt effect is not as striking as the common ion effect. The presence of the common ion Cr. O 42–(aq), from K 2 Cr. O 4, decreases the solubility of Ag 2 Cr. O 4 by a factor of 35. • In contrast, the presence of the uncommon ions K+(aq) and NO 3–(aq), from KNO 3, increase the solubility of Ag 2 Cr. O 4 by a factor of only 0. 25.

CALCULATING Ksp FROM SOLUBILITY • In today’s experiment you will measure the solubility of potassium hydrogen tartrate (KOOC(CHOH)2 COOH). KOOC(CHOH)2 COOH(s) = K+(aq) + –OOC(CHOH)2 COOH(aq) • What is the Ksp for this reaction? Ksp = [K+][–OOC(CHOH)2 COOH] • You will make a saturated solution of KOOC(CHOH)2 COOH in 0. 10 M Na. Cl and in 0. 10 M KNO 3. • • Is the Na. Cl a source of a common ion or uncommon ions? Na+ and Cl– are uncommon ions. This Na. Cl should increase or decrease the solubility of KOOC(CHOH)2 COOH? Increase. Is the KNO 3 a source of a common ion or uncommon ions? K+ is a common ion. NO 3– is an uncommon ion. This KNO 3 should increase or decrease the solubility of KOOC(CHOH)2 COOH? Decrease. The common ion effect is usually greater than the uncommon ion effect.
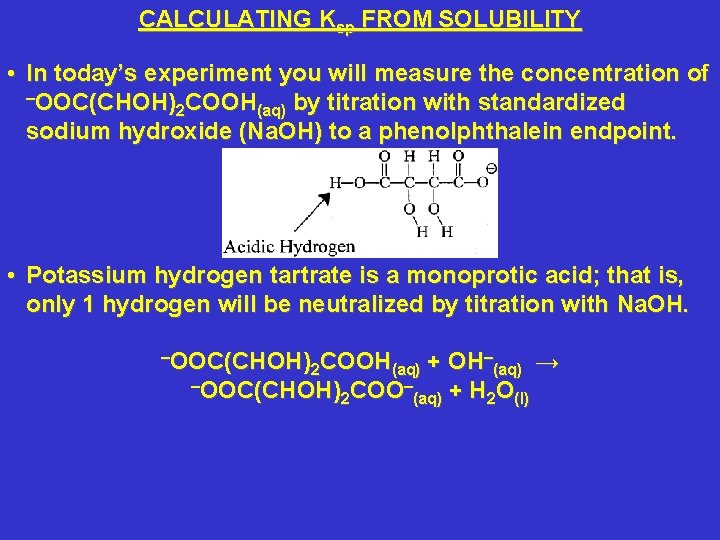
CALCULATING Ksp FROM SOLUBILITY • In today’s experiment you will measure the concentration of –OOC(CHOH) COOH 2 (aq) by titration with standardized sodium hydroxide (Na. OH) to a phenolphthalein endpoint. • Potassium hydrogen tartrate is a monoprotic acid; that is, only 1 hydrogen will be neutralized by titration with Na. OH. –OOC(CHOH) COOH 2 (aq) + –OOC(CHOH) COO– 2 (aq) OH–(aq) → + H 2 O(l)

TITRATION USING PHENOLPHTHALEIN AS AN INDICATOR • Stop adding base when the indicator just begins to turn a faint but stable pink. This is the endpoint.

SAFETY • Give at least 1 safety concern for the following procedure. • Using KOOC(CHOH)2 COOH, Na. Cl, KNO 3, Na. OH, and phenolphthalein. • These are irritants. Wear your goggles at all times. Immediately clean all spills. If you do get either of these in your eye, immediately flush with water. • Your laboratory manual has an extensive list of safety procedures. Read and understand this section. • Ask your instructor if you ever have any questions about safety.

SOURCES • Mc. Murry, J. , R. C. Fay. 2004. Chemistry, 4 th ed. Upper Saddle River, NJ: Prentice Hall. • Petrucci, R. H. 1985. General Chemistry Principles and Modern Applications, 4 th ed. New York, NY: Macmillan Publishing Company. • Traverso M. 2006. Titration using Phenolphthalein as an Indicator. Available: www. chemistry. wustl. edu/. . . /Acid. Base/phph. htm [accessed 14 September 2006].
- Slides: 14