CH 103 ACIDBASE TITRATIONS An analyte is the

CH 103: ACID-BASE TITRATIONS • An analyte is the substance that is being analyzed. For example, the concentration of glucose in blood is commonly analyzed by diabetics. Glucose is the analyte. • A titrant is a solution of reagent that reacts with the analyte. The concentration of this reagent is accurately and precisely known. • In a titration, incremental volumes of titrant are added to the analyte until the reaction is complete. • A buret is often used to measure the volume of titrant added to the analyte.

REQUIREMENTS OF A TITRATION 1. The reaction must be stoichiometric. For example, the net ionic equation for the reaction of potassium hydrogen phthalate (KHP, HOOC-C 6 H 4 -COOK) and sodium hydroxide (Na. OH) is quantitative. Exactly 1 mole of KHP reacts with exactly 1 mole of Na. OH. 1. HOOC-C 6 H 4 -COO- + OH- → -OOC-C 6 H 4 -COO- + H 2 O 2. The reaction should be rapid. 3. The reaction should be specific; that is, there should be no competing reactions. Systematic error caused by interferences must be eliminated or reduced. For example, HOOC-C 6 H 4 -COO- should be the only acid in the above reaction. 4. There should be a marked change when the reaction is complete. For example, there is a marked increase in p. H when the above reaction is complete. This increase in p. H can be observed with an indicator that changes color immediately after the reaction between the titrant and analyte is completed.

EQUIVALENCE POINT, END POINT, AND INDICATORS • The equivalence point occurs when the volume of titrant added to the analyte is the exact stoichiometric amount that is needed to bring the reaction to completion. • The end point occurs when the indicator changes color. • We want to measure the equivalence point. We actually measure the end point. We need to select an indicator that has the same end point as the equivalence point. • The indicator is added to the analyte. It is NOT added to the titrant.

EQUIVALENCE POINT, END POINT, AND INDICATORS Titration using Phenolphthalein as an Indicator • When do you stop adding titrant to the analyte? • At the end point.

EQUIVALENCE POINT, END POINT, AND INDICATORS • The titration of 0. 100 M acetic acid (CH 3 COOH) with 0. 100 M Na. OH has an equivalence point at p. H 8. 73. What indicator should you use for this reaction? • Phenolphthalein.

STANDARDIZATION • Today we will standardize Na. OH against potassium hydrogen phthalate (KHP, HOOC-C 6 H 4 -COOK). • KHP is a primary standard. • For example, 25. 71 m. L of a Na. OH solution are used to titrate 1. 095 g of KHP to a phenolphthalein end point. The molar mass of KHP is 204. 2 g/mol. One mole of KHP reacts with 1 mole of Na. OH. What is the molarity (M) of this Na. OH solution? • Next week you will use your Na. OH solution to determine the molecular weight of an unknown acid. • This Na. OH solution is a secondary standard.

REQUIREMENTS OF A PRIMARY STANDARD 1. A primary standard should be 100. 00% pure; although a 0. 01% to 0. 02% impurity is tolerable if it is accurately known. 2. A primary standard should be stable at drying temperatures, and it should be stable indefinitely at room temperature. (A primary standard is always dried before weighing, unless it is a hydrate. ) 3. It should be readily available. 4. It should have a relatively large formula weight. Therefore, a relatively large mass of it will be weighed for titration. This will reduce error. • Explain this last point.

TITRATION OF A STRONG ACID WITH A STRONG BASE • • • Where is the equivalence point? At p. H 7. Where is the end point? Between p. H 8. 0 and 9. 5. Is there a significant difference between the volume of titrant used to reach the equivalence and end points? • No. Therefore, phenolphthalein is an appropriate indicator for this titration.
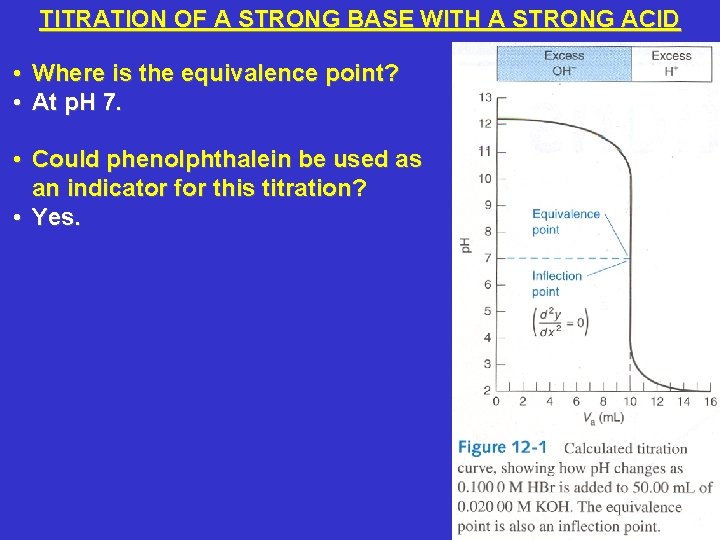
TITRATION OF A STRONG BASE WITH A STRONG ACID • Where is the equivalence point? • At p. H 7. • Could phenolphthalein be used as an indicator for this titration? • Yes.
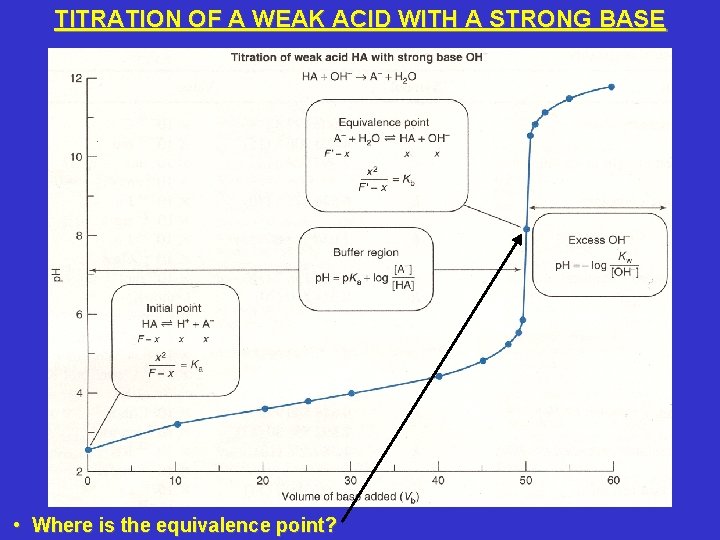
TITRATION OF A WEAK ACID WITH A STRONG BASE • Where is the equivalence point?
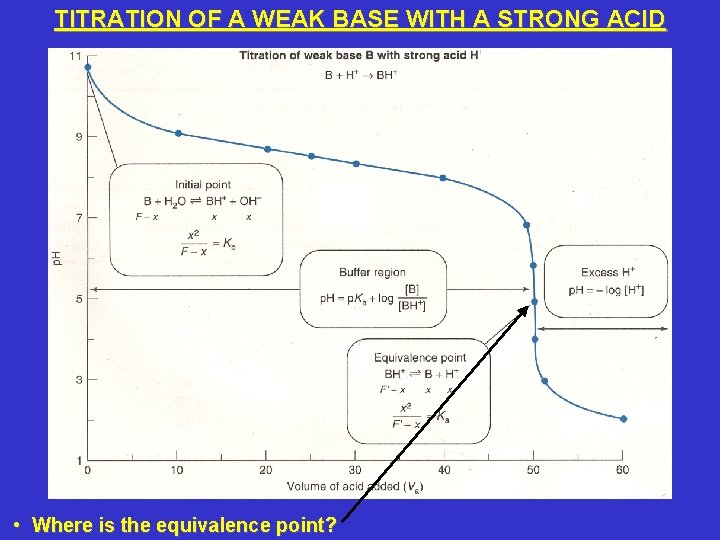
TITRATION OF A WEAK BASE WITH A STRONG ACID • Where is the equivalence point?

STANDARD DEVIATION • Standard deviation is a measure of precision. It is used to quantify the agreement between repeated measurements of the sample.

SAFETY • Give at least 1 safety concern for the following procedure. • Using HOOC-C 6 H 4 -COOK, Na. OH, phenolphthalein, and an unknown acid. • These are irritants. Wear your goggles at all times. Immediately clean all spills. If you do get either of these in your eye, immediately flush with water. • Your laboratory manual has an extensive list of safety procedures. Read and understand this section. • Ask your instructor if you ever have any questions about safety.

SOURCES • Christian, G. D. 1986. Analytical Chemistry, 3 rd ed. New York, NY: John Wiley & Sons, Inc. • Harris, D. C. 1999. Quantitative Chemical Analysis, 5 th ed. New York, NY: W. H. Freeman Company. • Traverso M. 2006. Titration using Phenolphthalein as an Indicator. Available: www. chemistry. wustl. edu/. . . /Acid. Base/phph. htm [accessed 14 September 2006].
- Slides: 14