Ch 10 Gases The Kinetic Molecular Theory Gases





























- Slides: 54

Ch 10: Gases & The Kinetic Molecular Theory Gases

Characteristics of Gases Condensed phases • Unlike liquids and solids, gases. . Ø Are highly compressible. Ø Expand to fill their containers. Ø Have extremely low densities. Gases

Characteristics of Gases • Variables affecting the behavior of gases Ø Ø Amount = number of moles ( ) Pressure (P) Volume (V) Temperature (T in Kelvin) {Prop. Gases*} Gases

Pressure • Pressure is the amount of force applied to an area. F P= A = 105 Newtons meter 2 • Atmospheric pressure is the weight of air per unit of area. Force = mass x acceleration Newton = 1 kg. m/sec 2 105 Newtons = (104 kg)(10 m/sec 2) Approx. 12 miles = 101. 325 k. Pa Gases

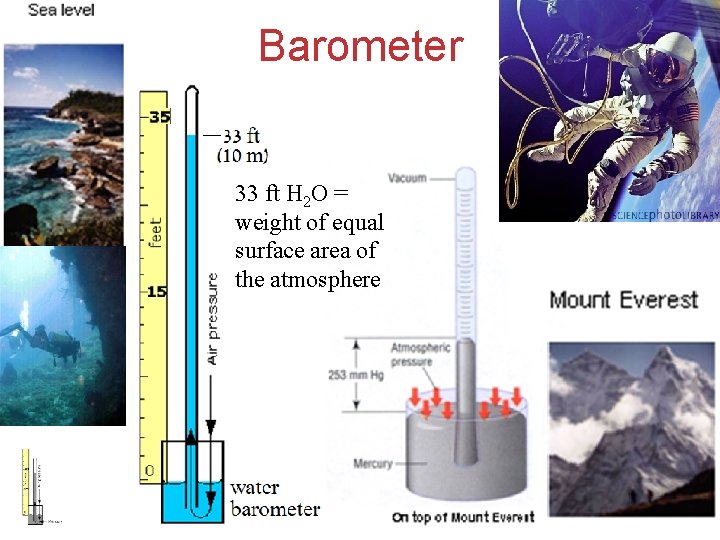
Units of Pressure Torricelli’s Atmosphere 1. 00 atm = 760 mm Hg (torr) = 101. 325 k. Pa 760 mm Hg = weight of equal surface area of (Normal atmospheric the atmosphere pressure at sea level). Gases

Barometer 33 ft H 2 O = weight of equal surface area of the atmosphere Gases

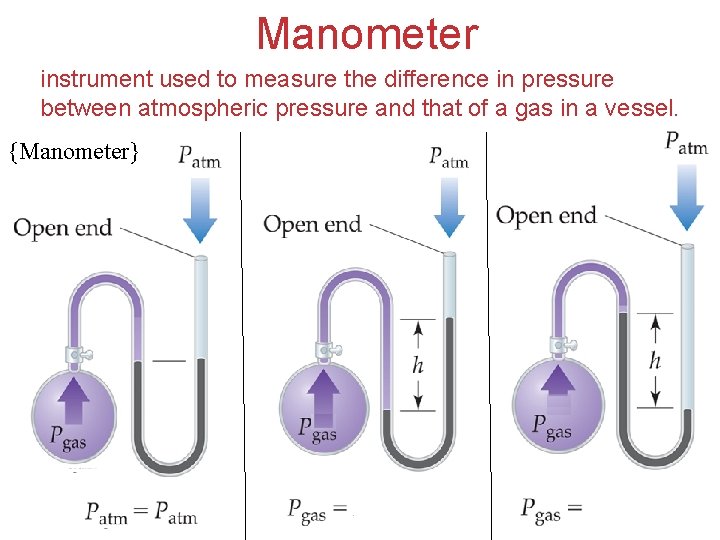
Manometer instrument used to measure the difference in pressure between atmospheric pressure and that of a gas in a vessel. {Manometer} Gases

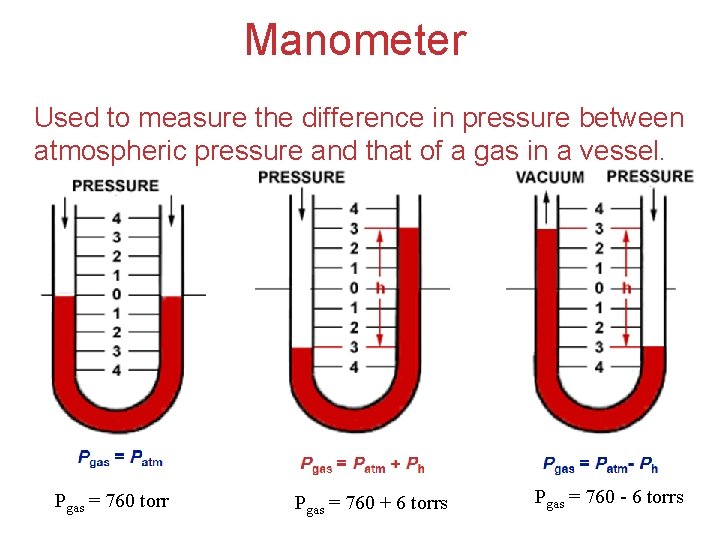
Manometer Used to measure the difference in pressure between atmospheric pressure and that of a gas in a vessel. Pgas = 760 torr Pgas = 760 + 6 torrs Gases Pgas = 760 - 6 torrs

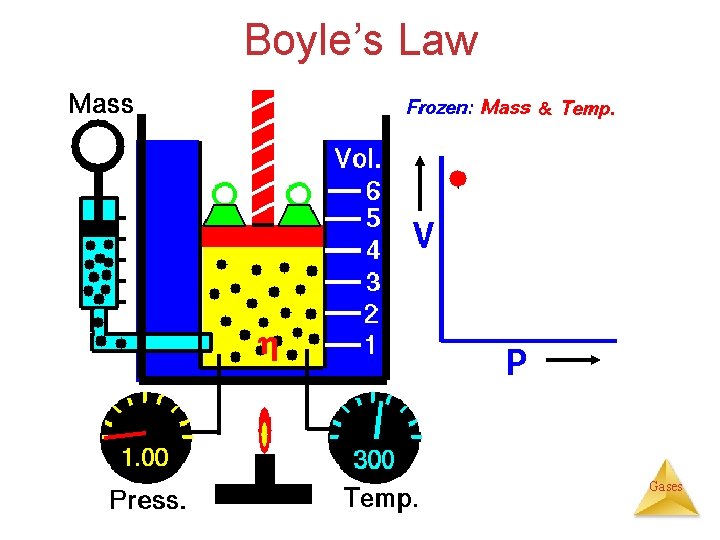
Gas Laws Variables affecting gases: moles (η), pressure (P), volume (V) and Temperature (T) 1. Boyle’s Law Ø Compared: P versus V ( & T are held constant). 2. Charles’s Law Ø Compared: V versus T ( & P are held constant). • Avogadro’s Law Ø Compared: V versus η (P & T are held constant). • Combined Gas Law Ø Compared: P vs V vs. T ( is held constant). 1. Ideal Gas Law Ø Compared: P vs V vs. η vs T (no variable held constant). 2. Dalton’s Law of Partial Pressure Ø Compared: individual pressures of gases in a mixture Gases

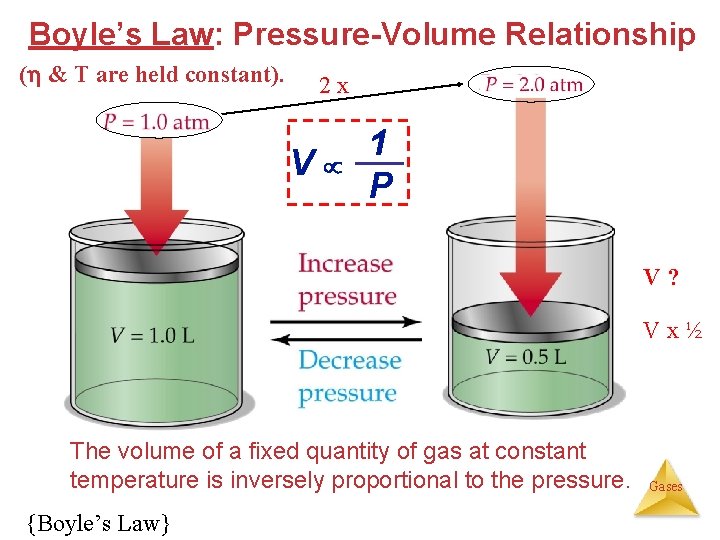
Boyle’s Law: Pressure-Volume Relationship ( & T are held constant). 2 x V 1 P V? Vx½ The volume of a fixed quantity of gas at constant temperature is inversely proportional to the pressure. {Boyle’s Law} Gases

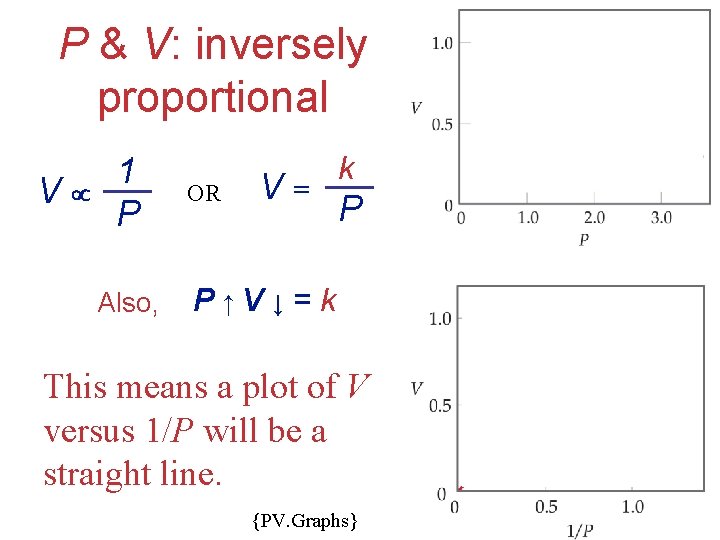
P & V: inversely proportional V 1 P Also, OR V= k P P ↑V ↓ = k This means a plot of V versus 1/P will be a straight line. {PV. Graphs} Gases

Boyle’s Law Gases

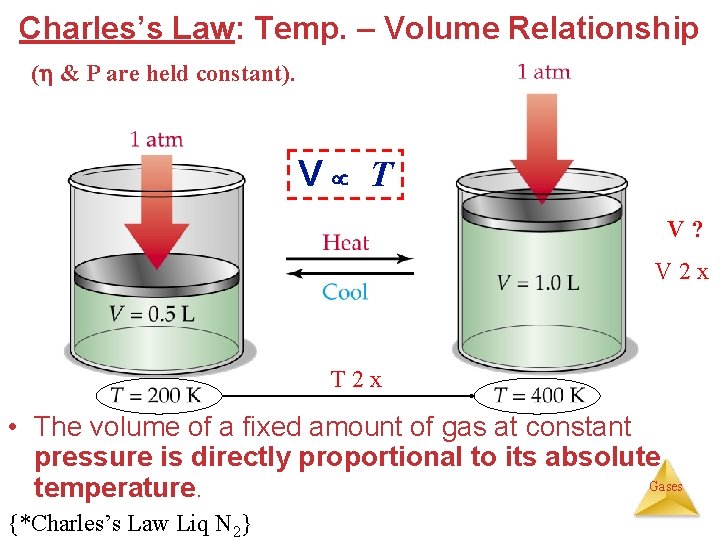
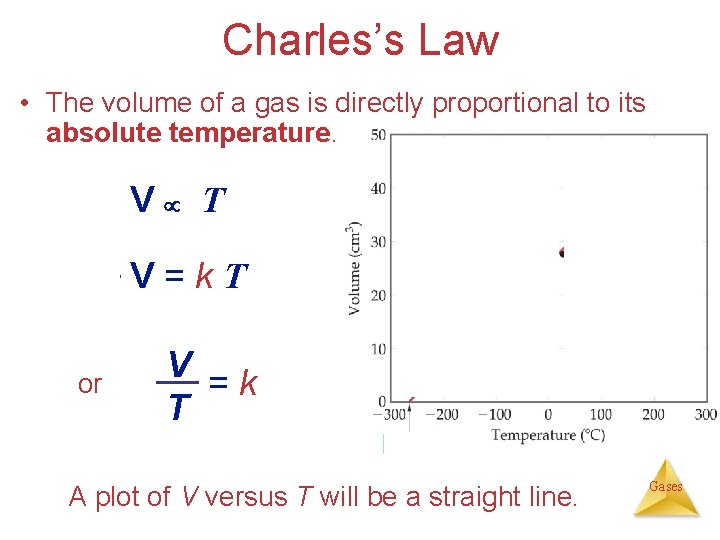
Charles’s Law: Temp. – Volume Relationship ( & P are held constant). V T V? V 2 x T 2 x • The volume of a fixed amount of gas at constant pressure is directly proportional to its absolute Gases temperature. {*Charles’s Law Liq N 2}

Charles’s Law • The volume of a gas is directly proportional to its absolute temperature. V T V=k. T or V =k T A plot of V versus T will be a straight line. Gases

Charles’s Law Gases

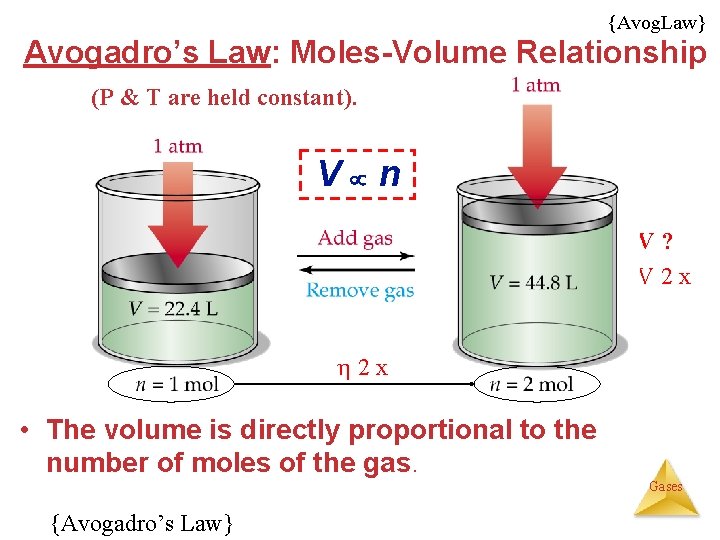
{Avog. Law} Avogadro’s Law: Moles-Volume Relationship (P & T are held constant). V n V? V 2 x • The volume is directly proportional to the number of moles of the gas. Gases {Avogadro’s Law}

Avogadro’s Law • Mathematically, this means or, V n V=kn Gases {*Avogadro’s Law in Reactions}

Standard Temperature & Pressure (STP) and Molar Volume • Standard Temperature: 00 C or 273 K • Standard Pressure: 760 torr (1 atm) At STP the Molar Volume of any gas is 22. 4 L (11. 1 in)3 or (28. 2 cm)3 1 mole = 6. 022 x 1023 part. = g. MM = 22. 4 L Gases

Standard Temperature & Pressure (STP) and Molar Volume At STP the Molar Volume of any gas is 22. 4 L 1 mole = 6. 023 x 1023 part. = g. MM = 22. 4 L H 2 = 2. 0 g O 2 = 32. 0 g CO 2 = 44. 0 g Problem: At STP, what volume in m. L would 75 g of CO 2 occupy? Gases

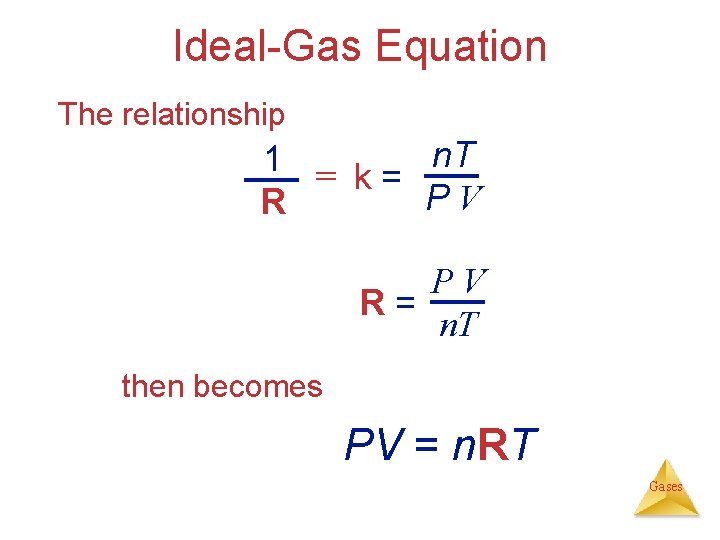
Ideal-Gas Equation The Gas Laws: V 1/P (Boyle’s law) V T (Charles’s law) V n (Avogadro’s law) Combining these, we get n. T V P or kn. T V= P or n. T k= PV Gases

Ideal-Gas Equation The relationship n. T 1 = k= PV R= n. T then becomes PV = n. RT Gases

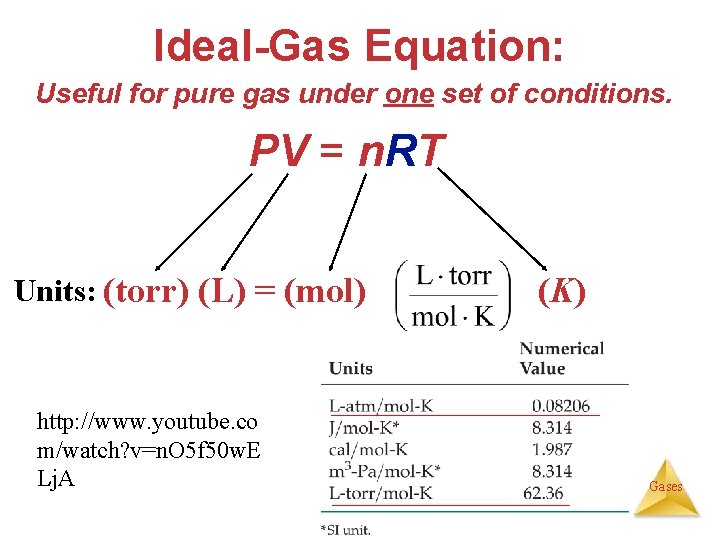
Ideal-Gas Equation: Useful for pure gas under one set of conditions. PV = n. RT Units: (torr) (L) = (mol) http: //www. youtube. co m/watch? v=n. O 5 f 50 w. E Lj. A R (K) Gases

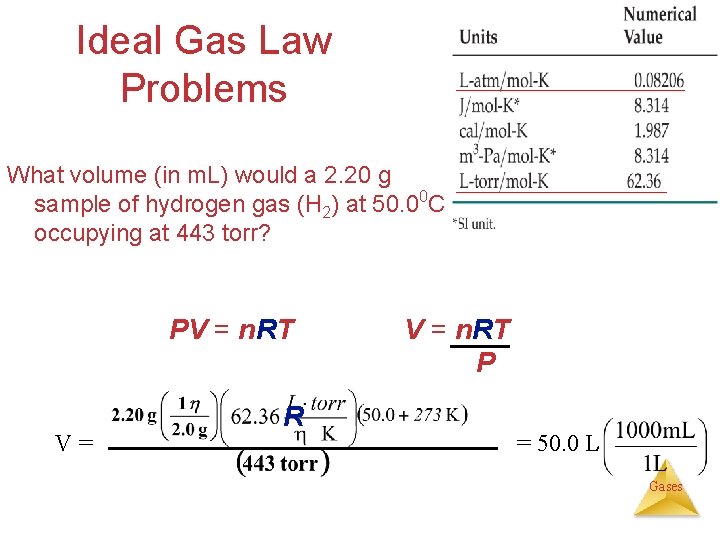
Ideal Gas Law Problems What volume (in m. L) would a 2. 20 g sample of hydrogen gas (H 2) at 50. 00 C occupying at 443 torr? PV = n. RT V= R V = n. RT P = 50. 0 L Gases

Ideal-Gas Equation: Densities of Gases For Ideal Gas Equation: PV = n. RT Since Then and Dividing both sides of the equation on the left by V we get ( ) Where d = Density of Gas If we solve the equation for density, we get……. . Gases

Ideal-Gas Equation: Densities and Molecular Weigh of Gases Problem: What is the density of the oxygen in a tank in an AC room (25°C) and whose pressure gauge reads 25. 0 atm Problem: A gas whose density is 0. 0131 g/m. L and is in a container at room temperature and whose pressure gauge reads 1. 9 x 104 mm. Hg. What is its MW? Gases

Ideal-Gas Equation: Densities & Molecular Weigh of Gases Problems What is the density (in g/m. L) of SO 2 at STP? PV = n. RT () = = 2. 62 g/L Gases

Ideal-Gas Equation: Densities & Molecular Weigh of Gases Problems What is the molecular weight of a gas whose density @ STP is 7. 78 g/L? PV = n. RT () = Gases

2006 A Gases

Gases

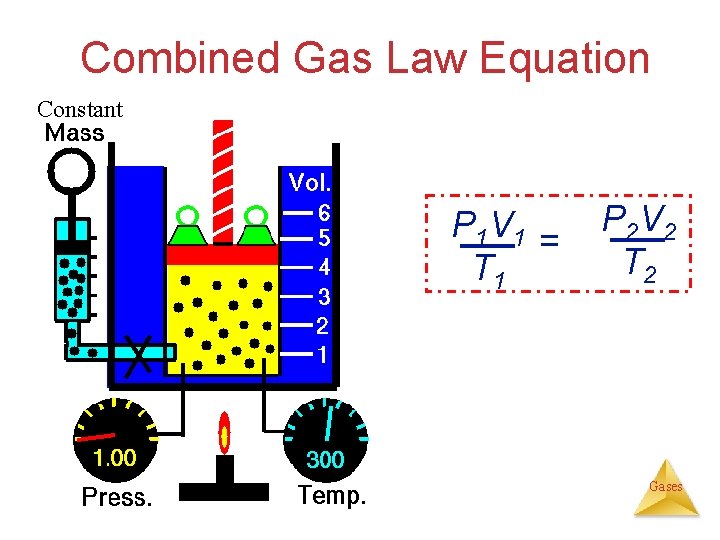
Combined Gas Law Equation The Gas Laws: V 1/P (Boyle’s law) V T (Charles’s law) Combining, we can get The Combined Gas Law P 1 V 1 P V 2 2 k T 1 = T 2 Useful for a constant amount of a pure gas under Gases two different conditions.

Combined Gas Law Equation Constant P 1 V 1 = T 1 P 2 V 2 T 2 Gases

Combined Gas Law Problem A scuba diver takes a gas filled 1. 0 L balloon from the surface where the temperature is 34 0 C down to a depth of 66 ft (33 ft H 2 O = 1 atm). What volume will the gas balloon have at that depth if the temperature is 15 0 C? P 1 V 1 T 1 = P 2 V 2 T 2 V 2 Gases

Dalton’s Law of Partial Pressures • The total pressure of a mixture of gases equals the sum of the pressures that each would exert if it were present alone. • In other words, Ptotal = P 1 + P 2 + P 3 + … Pair = P N 2 + PO 2 + PH 2 O + … Gases

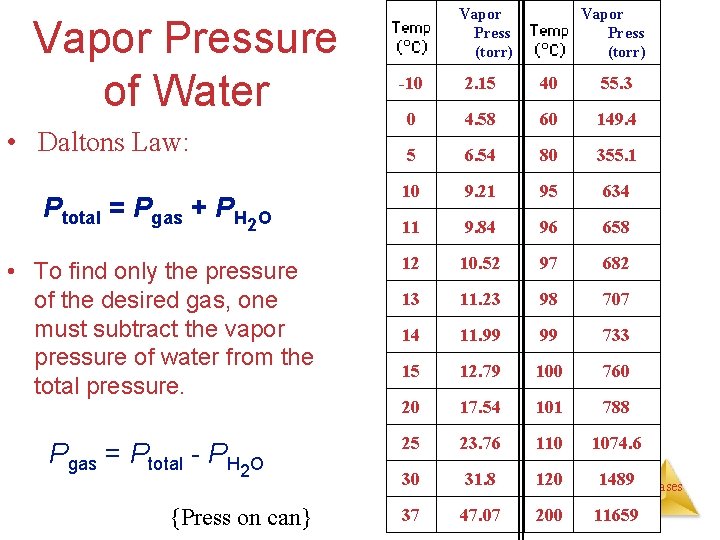
Partial Pressures • When one collects a gas over water, there is water vapor mixed in with the gas. P of gas P of atm • To find only the pressure of the desired gas, one must subtract Gases the vapor pressure of water from the total pressure.

Vapor Pressure of Water • Daltons Law: Ptotal = Pgas + PH 2 O • To find only the pressure of the desired gas, one must subtract the vapor pressure of water from the total pressure. Pgas = Ptotal - PH 2 O {Press on can} Vapor Press (torr) -10 2. 15 40 55. 3 0 4. 58 60 149. 4 5 6. 54 80 355. 1 10 9. 21 95 634 11 9. 84 96 658 12 10. 52 97 682 13 11. 23 98 707 14 11. 99 99 733 15 12. 79 100 760 20 17. 54 101 788 25 23. 76 110 1074. 6 30 31. 8 120 1489 37 47. 07 200 11659 Gases

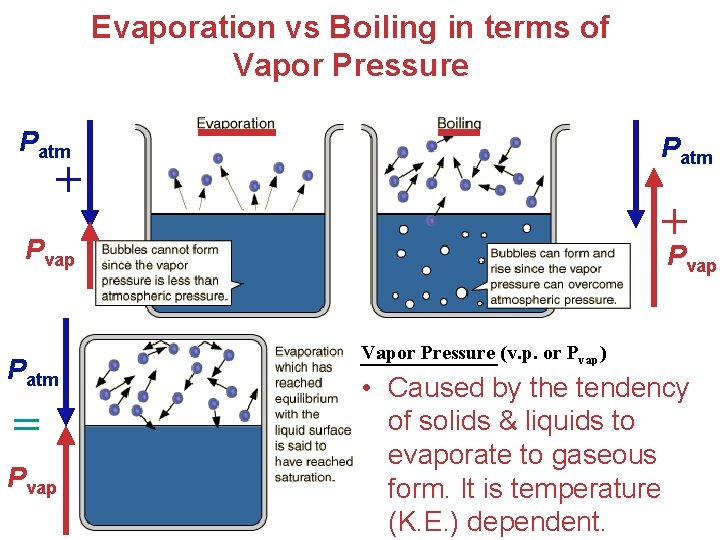
Evaporation vs Boiling in terms of Vapor Pressure Patm + + Pvap Patm = Pvap Vapor Pressure (v. p. or Pvap) • Caused by the tendency of solids & liquids to evaporate to gaseous form. It is temperature. Gases (K. E. ) dependent.

Stoichiometry with Gases Mg (s) + 2 HCl (aq) Mg. Cl 2 (aq) + H 2 (g) Problem: If 2. 0 g of Mg are reacted with excess HCl, what volume of H 2 will be produced at 250 C and 775 torr? At STP? PV = n. RT Gases

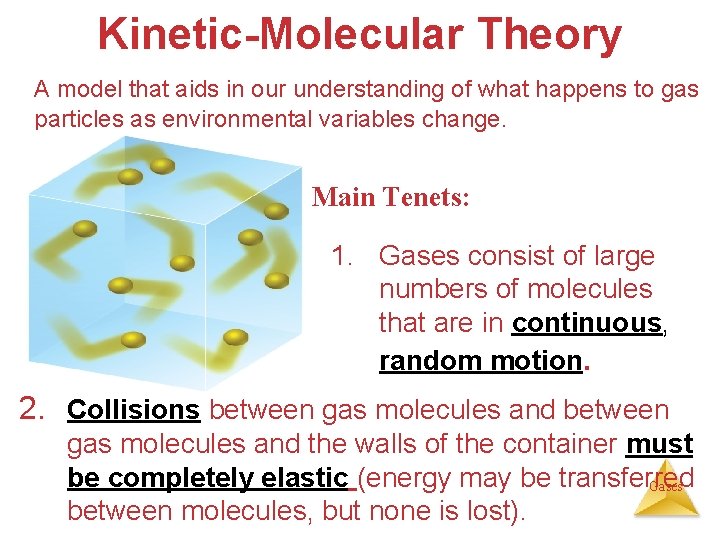
Kinetic-Molecular Theory A model that aids in our understanding of what happens to gas particles as environmental variables change. Main Tenets: 1. Gases consist of large numbers of molecules that are in continuous, random motion. 2. Collisions between gas molecules and the walls of the container must be completely elastic (energy may be transferred Gases between molecules, but none is lost).

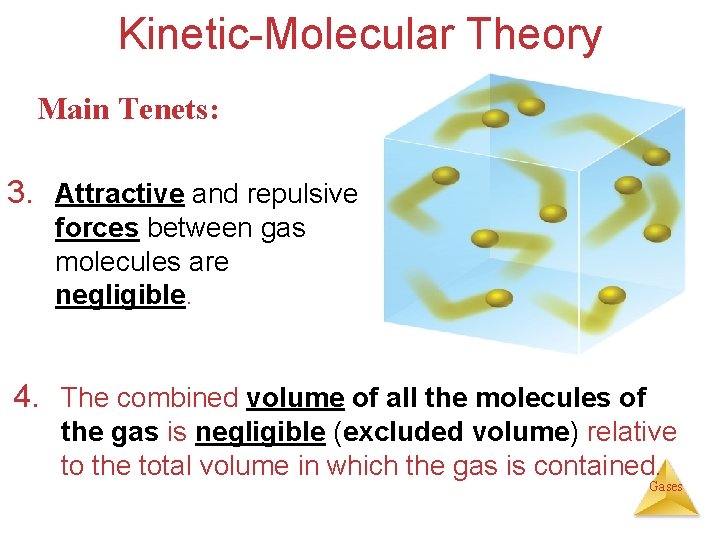
Kinetic-Molecular Theory Main Tenets: 3. Attractive and repulsive forces between gas molecules are negligible. 4. The combined volume of all the molecules of the gas is negligible (excluded volume) relative to the total volume in which the gas is contained. Gases

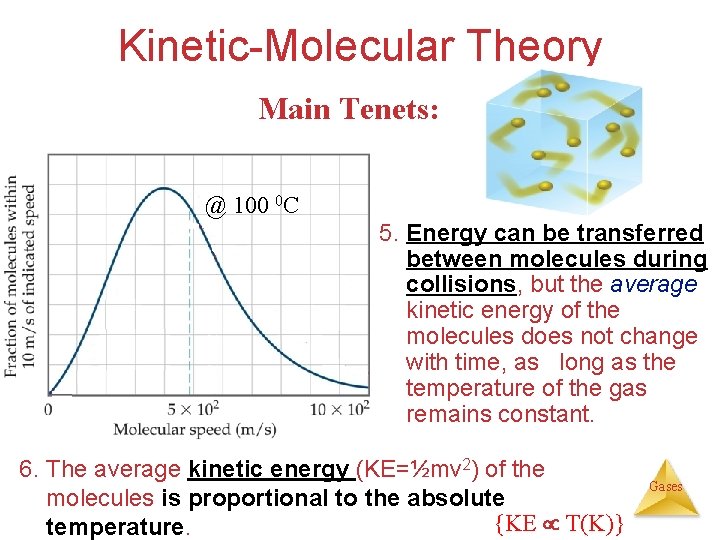
Kinetic-Molecular Theory Main Tenets: @ 100 0 C 5. Energy can be transferred between molecules during collisions, but the average kinetic energy of the molecules does not change with time, as long as the temperature of the gas remains constant. 6. The average kinetic energy (KE=½mv 2) of the molecules is proportional to the absolute {KE T(K)} temperature. Gases

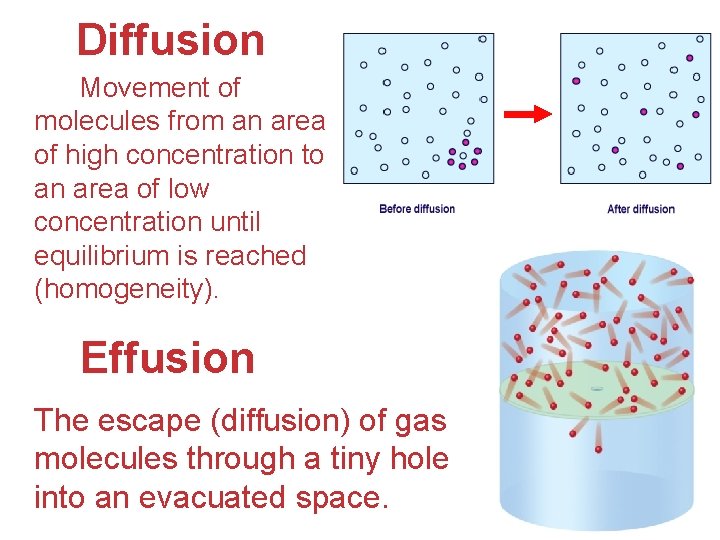
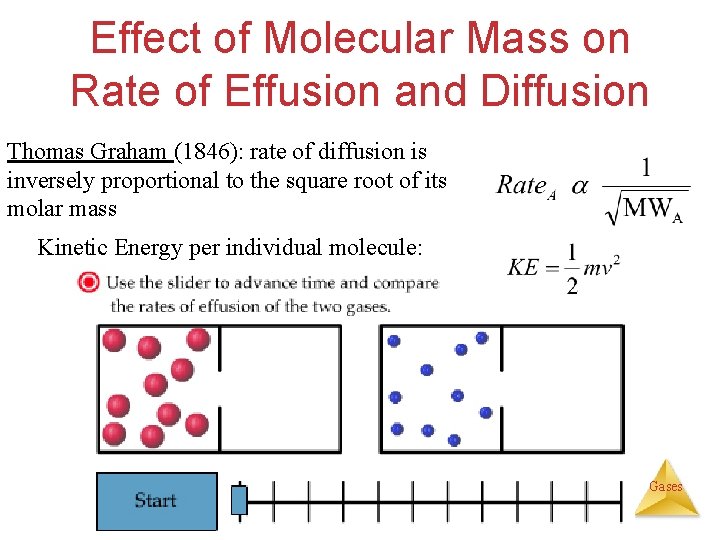
Diffusion Movement of molecules from an area of high concentration to an area of low concentration until equilibrium is reached (homogeneity). Effusion The escape (diffusion) of gas molecules through a tiny hole into an evacuated space. Gases

Effect of Molecular Mass on Rate of Effusion and Diffusion Thomas Graham (1846): rate of diffusion is inversely proportional to the square root of its molar mass Kinetic Energy per individual molecule: Gases

Rate of Diffusion & Effusion Dropper with Br (l) Thomas Graham (1846): rate of diffusion is inversely proportional to the square root of its molar mass {Br. Diffusion} Comparing the rates of two gases: Graham’s Law of Diffusion and Effusion of Gases {Gas. Diff}

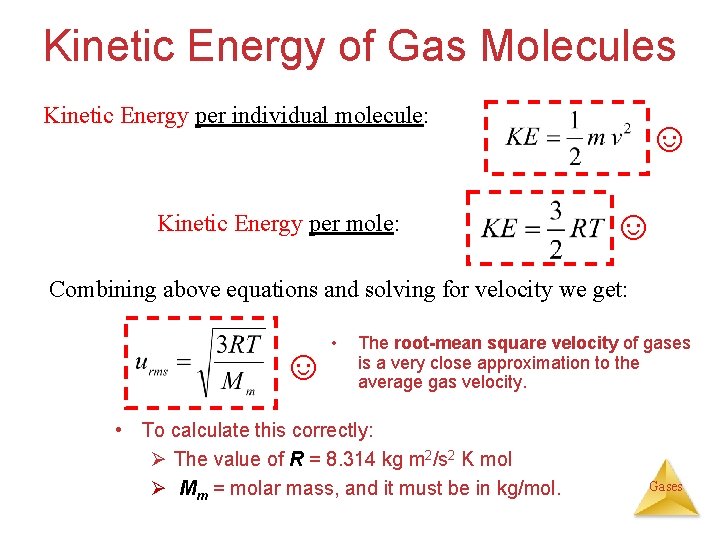
Kinetic Energy of Gas Molecules Kinetic Energy per individual molecule: Kinetic Energy per mole: ☺ ☺ Combining above equations and solving for velocity we get: ☺ • The root-mean square velocity of gases is a very close approximation to the average gas velocity. • To calculate this correctly: Ø The value of R = 8. 314 kg m 2/s 2 K mol Ø Mm = molar mass, and it must be in kg/mol. Gases

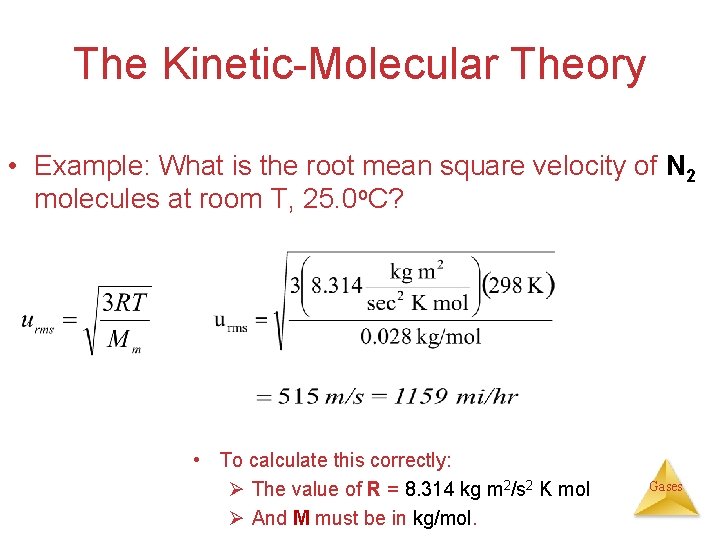
The Kinetic-Molecular Theory • Example: What is the root mean square velocity of N 2 molecules at room T, 25. 0 o. C? • To calculate this correctly: Ø The value of R = 8. 314 kg m 2/s 2 K mol Ø And M must be in kg/mol. Gases

The Kinetic-Molecular Theory Problem: What is the root mean square velocity of He atoms at room T, 25. 0 o. C? You do it! • To calculate this correctly: Ø The value of R = 8. 314 kg m 2/s 2 K mol Ø And M must be in kg/mol. • Can you think of a physical situation that proves He molecules have a velocity that is so much greater than N 2 molecules? • What happens to your voice when you breathe He(g) or SF 6 (g)? Gas MW He. Gases 4 N 2 28 SF 6 146

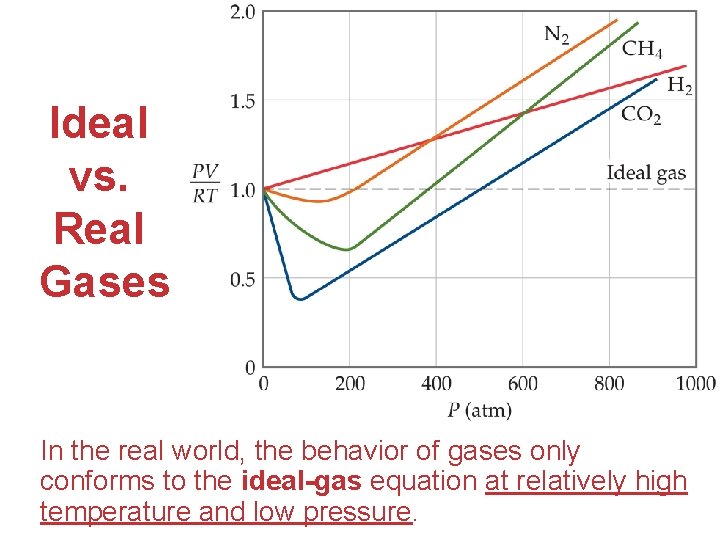
Ideal vs. Real Gases In the real world, the behavior of gases only conforms to the ideal-gas equation at relatively high Gases temperature and low pressure.

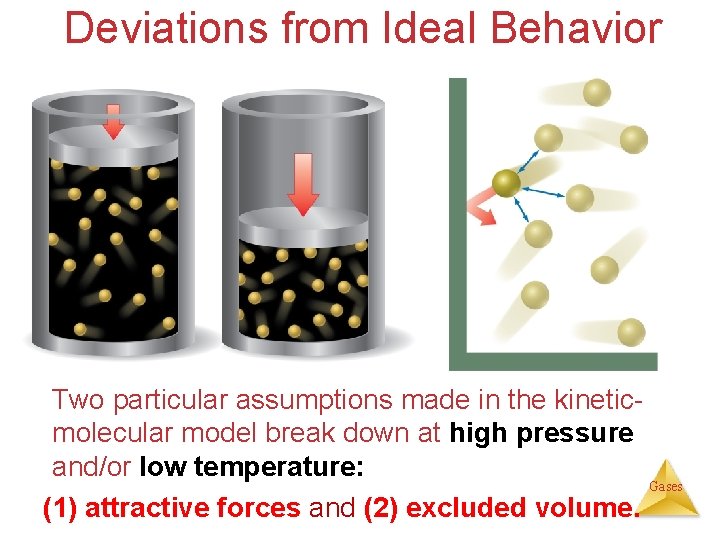
Deviations from Ideal Behavior Two particular assumptions made in the kineticmolecular model break down at high pressure and/or low temperature: Gases (1) attractive forces and (2) excluded volume.

2003 A Gases

Corrections for Non-ideal Gas Behavior • The ideal-gas equation can be adjusted to take these deviations from ideal behavior into account. • The corrected ideal-gas equation is known as the van der Waals equation. Gases

The van der Waals Equation n 2 a P + 2 (V − nb) = n. RT V ( ) Gases

Real Gases: Deviations from Ideality • van der Waals’ equation accounts for the behavior of real gases at low temperatures and high pressures. a b • The van der Waals constants a and b take into account two things: 1. a accounts for intermolecular attraction 2. b accounts for volume of gas molecules • At large volumes a and b are relatively small and van der Waal’s equation reduces to ideal Gases gas law at high temperatures and low pressures.

Real Gases: Deviations from Ideality • Example: Calculate the pressure exerted by 84. 0 g of ammonia, NH 3, in a 5. 00 L container at 200. o. C using the ideal gas law. You do it! Gases

Real Gases: Deviations from Ideality • Example: Calculate the pressure exerted by 84. 0 g of ammonia, NH 3, in a 5. 00 L container at 200. o. C using the van der Waal’s equation You do it! Gases