Ch 10 Chemical Reactions III Types of Chemical















- Slides: 22

Ch. 10 – Chemical Reactions III. Types of Chemical Reactions C. Johannesson

A. Combustion • the burning of any substance in O 2 to produce heat A + O 2 B CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) C. Johannesson

A. Combustion • Products: – contain oxygen in reactants – hydrocarbons form CO 2 + H 2 O 4 Na(s)+ O 2(g) 2 Na 2 O(s) C 3 H 8(g)+ 5 O 2(g) 3 CO 2(g)+4 H 2 O(g) C. Johannesson

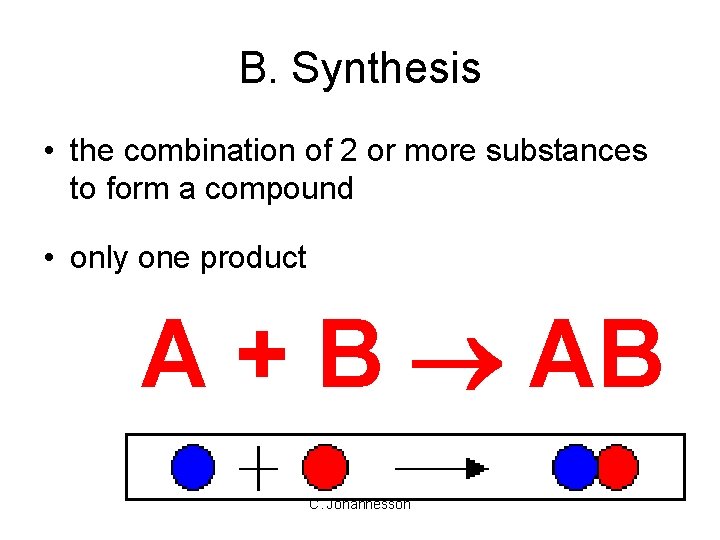
B. Synthesis • the combination of 2 or more substances to form a compound • only one product A + B AB C. Johannesson

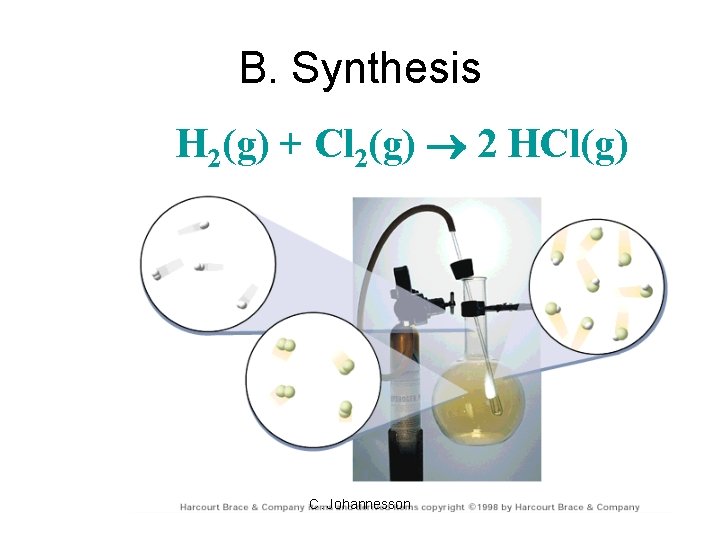
B. Synthesis H 2(g) + Cl 2(g) 2 HCl(g) C. Johannesson

B. Synthesis • Products: – ionic - cancel charges – covalent - hard to tell 2 Al(s)+ 3 Cl 2(g) 2 Al. Cl 3(s) C. Johannesson

C. Decomposition • a compound breaks down into 2 or more simpler substances • only one reactant AB A + B C. Johannesson

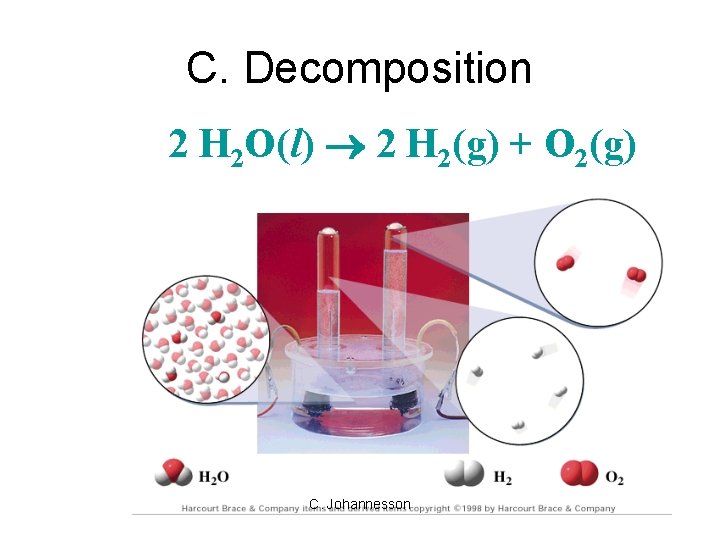
C. Decomposition 2 H 2 O(l) 2 H 2(g) + O 2(g) C. Johannesson

C. Decomposition • Products: – binary - break into elements – others - hard to tell (look at reference table) 2 KBr(l) 2 K(s) + C. Johannesson Br 2(l)

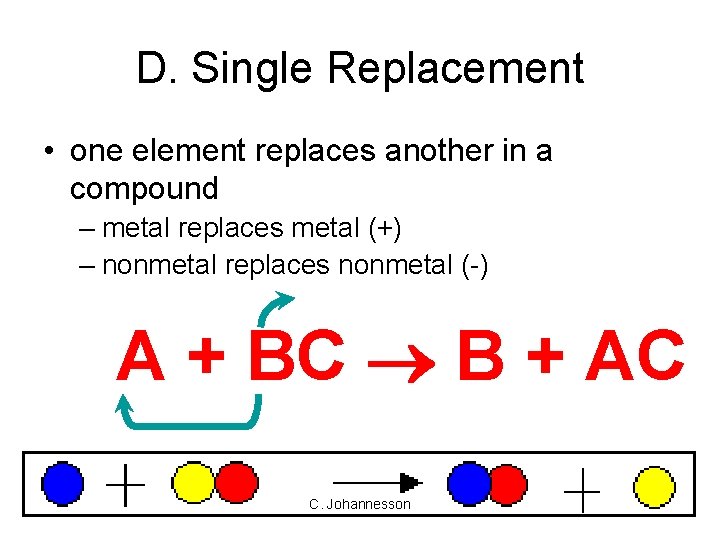
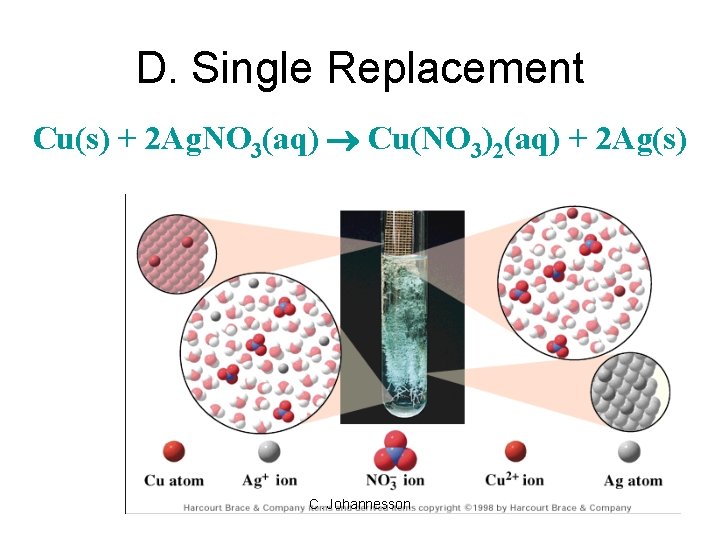
D. Single Replacement • one element replaces another in a compound – metal replaces metal (+) – nonmetal replaces nonmetal (-) A + BC B + AC C. Johannesson

D. Single Replacement Cu(s) + 2 Ag. NO 3(aq) Cu(NO 3)2(aq) + 2 Ag(s) C. Johannesson

D. Single Replacement • Products: – metal (+) – nonmetal (-) – free element must be more active (check activity series) Fe(s)+ Cu. SO 4(aq) Cu(s)+ Fe. SO 4(aq) Br 2(l)+ Na. Cl(aq) C. Johannesson N. R.

C. Johannesson

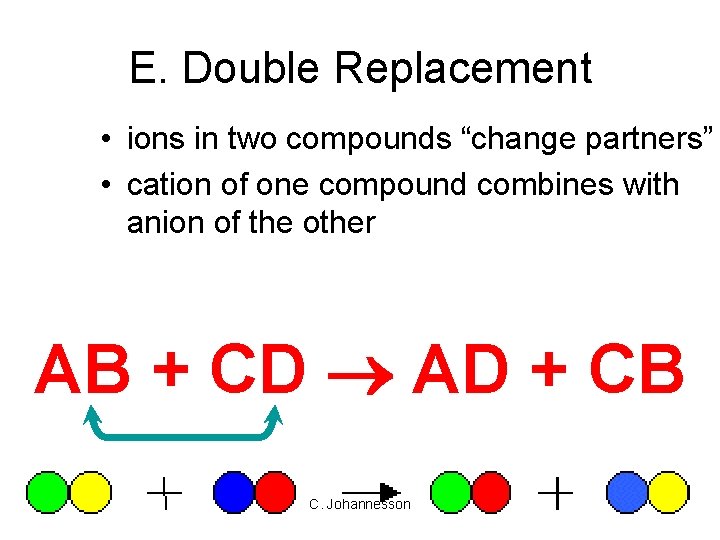
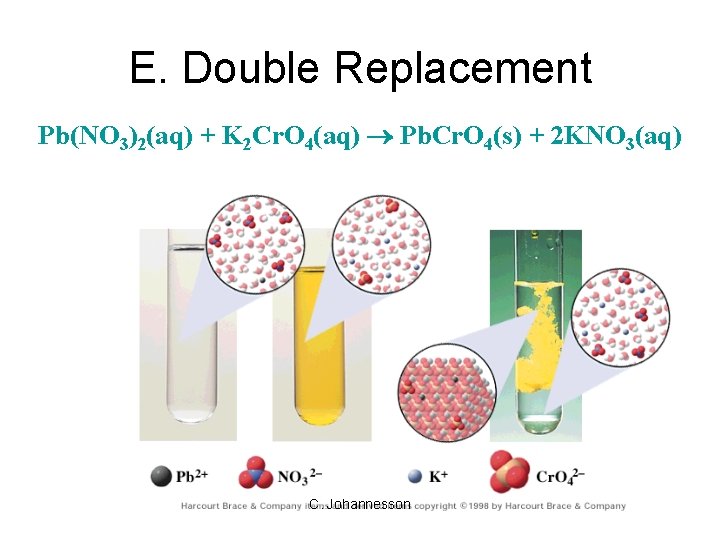
E. Double Replacement • ions in two compounds “change partners” • cation of one compound combines with anion of the other AB + CD AD + CB C. Johannesson

E. Double Replacement • Products: – switch negative ions – one product must be insoluble (check solubility table) C. Johannesson

E. Double Replacement Pb(NO 3)2(aq) + K 2 Cr. O 4(aq) Pb. Cr. O 4(s) + 2 KNO 3(aq) C. Johannesson

Writing Net Ionic Equations for Double Replacement Reactions • A net ionic equation only shows the ions that were used to make the precipitate. • Some ions were always dissolved in water. These are called “spectator ions”. (They don’t do anything, so we can ignore them. ) Example: Ca. Cl 2 (aq) + 2 Ag. NO 3 (aq) Ca(NO 3)2 (aq) + 2 Ag. Cl (s) Ionic Equation Written as Ions Dissolved in Water: +2 − + Ca 2 Cl 2 Ag ___ (aq) + • − − +2 2 NO Ca 2 NO 3 3 _____ (aq) + _____ (aq) + 2 Ag. Cl _____ (s) Cancel out the spectator ions, and you are left with the Net Ionic Equation! − + 2 Cl 2 Ag. Cl(s) (aq) ____ + _____ C. Johannesson

Writing Net Ionic Equations for Double Replacement Reactions Practice Problem: Write the net ionic equation for the following reaction. K 2 CO 3 (aq) + KNO 3 (aq)+ _____ Ba. CO 3 Ba(NO 3)2 (aq) _____ CO 3 − 2 (aq) + Ba+2 (aq) Ba. CO 3 Net Ionic Equation = _____________ C. Johannesson

In Summary… • To write the complete and net ionic equations… 1. Write formulas* of reactants and use solubility rules to determine state (aq or s) 2. Determine the products (switch the negative ions) -write formulas* and use solubility rules to determine state (aq or s) *don’t forget to use the periodic table and list of Johannesson polyatomic ions to. C. determine charge

3. Balance equation (this is a balanced chemical equation) 4. Split anything that is aq into ions -leave the s alone (this is the complete ionic equation) 5. Cross out spectator ions 6. Write what is left (this is the net ionic equation) C. Johannesson

Activity Series (Single Replacement reactions) • Higher up = more reactive • Elements from Li to Na can displace hydrogen in water to form a metallic hydroxide and H 2 gas. C. Johannesson

Single Replacement Reactions Examples: + F 2 Na. F + _____ Cl 2 _____ Fe. Cl 2 + K KCl + _____ Fe _____ Na. Cl HCl + H 2 Zn Zn. Cl _____ 2 + _____ HCl + Au no reaction _____ + _____ H 2 O + H(OH) H 2 O + Na H 2 _____ Na. OH + _____ Fe no reaction _____ + _____ Ag. NO 3 + Cu Cu. NO Ag _____3 + _____ C. Johannesson