Ch 1 Introduction Optoelectronic devices devices deal with







- Slides: 19

Ch. 1 Introduction • Optoelectronic devices: - devices deal with interaction of electronic and optical processes • Solid-state physics: - study of solids, through methods such as quantum mechanics, crystallography, electromagnetism and metallurgy • Elemental semiconductors: - Si, Ge, . . etc. - indirect bandgap, low electric-optics conversion efficiency • Compound semiconductors - III-V (e. g. Ga. N, Ga. As), II-VI - direct bandgap, high electric-optics conversion efficiency • Ga. As, In. P - higher mobility than Si, Ge, - energy band gap, Eg: 1. 43 (Ga. As), 1. 35 (In. P) - most common substrate, used to grow up compound semiconductors

Periodic Table

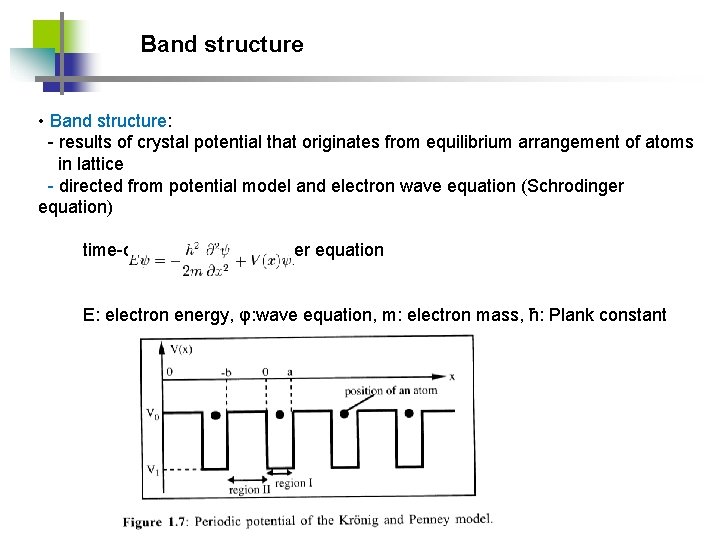
Band structure • Band structure: - results of crystal potential that originates from equilibrium arrangement of atoms in lattice - directed from potential model and electron wave equation (Schrodinger equation) time-dependent Schrodinger equation E: electron energy, φ: wave equation, m: electron mass, ħ: Plank constant

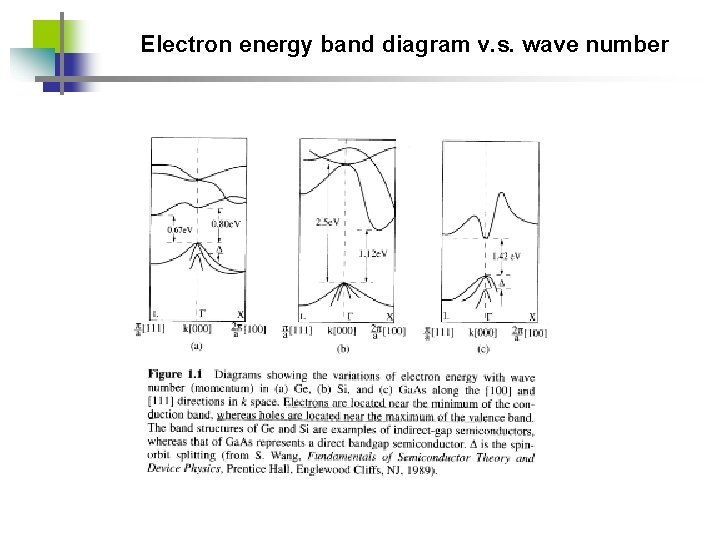
Electron energy band diagram v. s. wave number

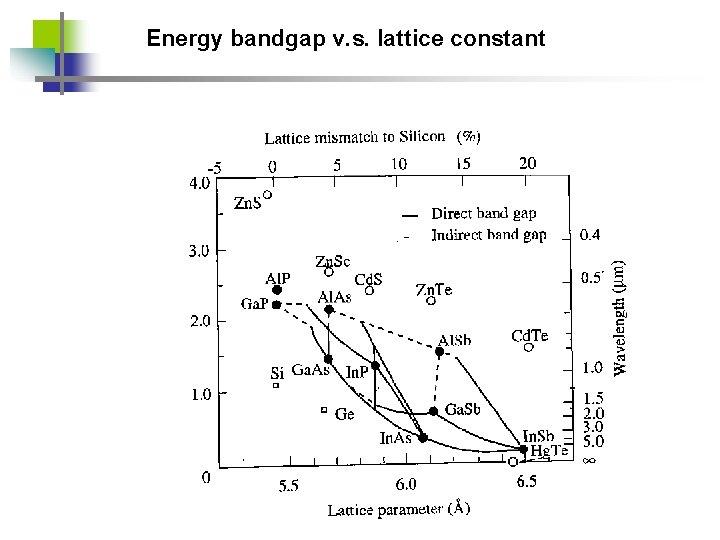
Energy bandgap v. s. lattice constant

Bonding in solids • Van der Waals bonding: formation of dipoles between atoms and their electrons e. g. : inert gas, like Ar • Ionic bonding: electron exchange between atoms produces positive and negative ions which attract each other by Coulomb-type interactions e. g. Na. Cl, KCl • covalent bonding sharing of electrons between neighboring atoms e. g. : elemental and compound semiconductors • Metallic bonding: valence electrons are shared by many atoms (bonding not directional, electron free or nearly free contributed to conductivity) e. g. : Zn

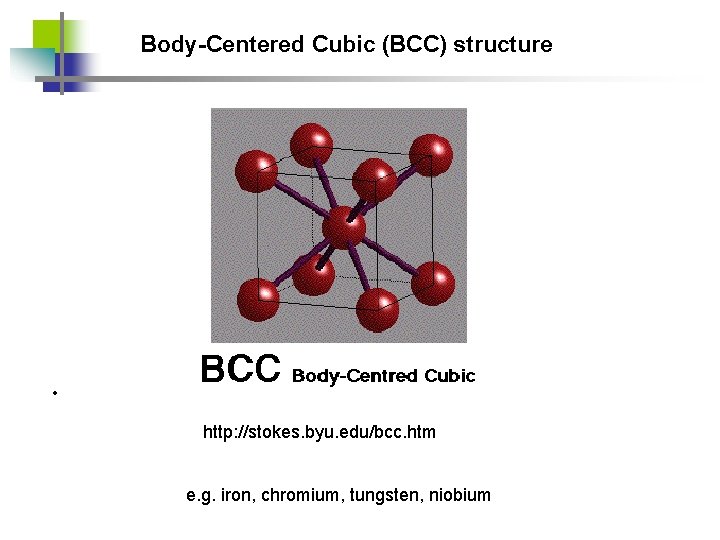
Body-Centered Cubic (BCC) structure • http: //stokes. byu. edu/bcc. htm e. g. iron, chromium, tungsten, niobium

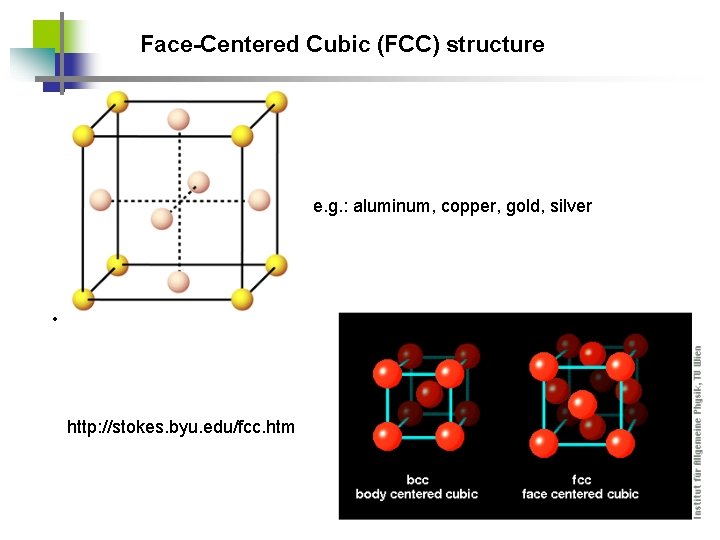
Face-Centered Cubic (FCC) structure e. g. : aluminum, copper, gold, silver • http: //stokes. byu. edu/fcc. htm

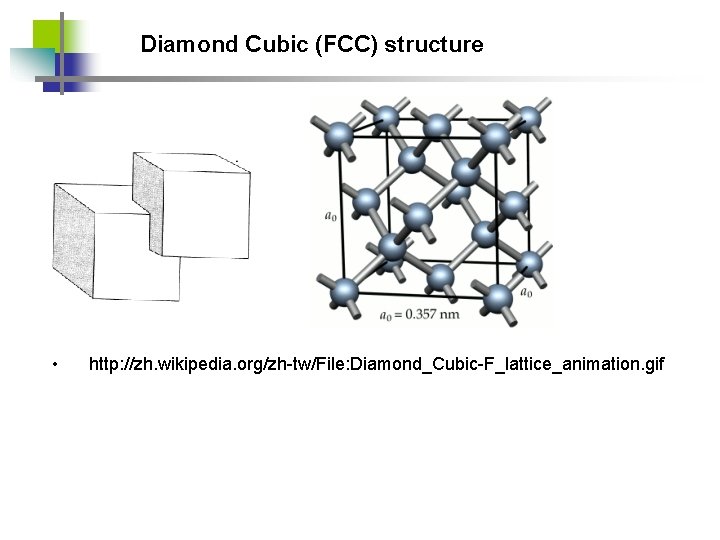
Diamond Cubic (FCC) structure • http: //zh. wikipedia. org/zh-tw/File: Diamond_Cubic-F_lattice_animation. gif

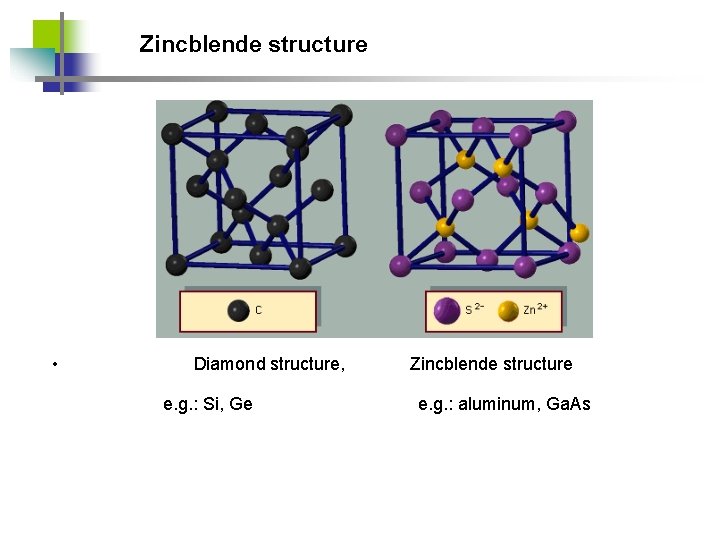
Zincblende structure • Diamond structure, e. g. : Si, Ge Zincblende structure e. g. : aluminum, Ga. As

Atomic arrangement in different solids

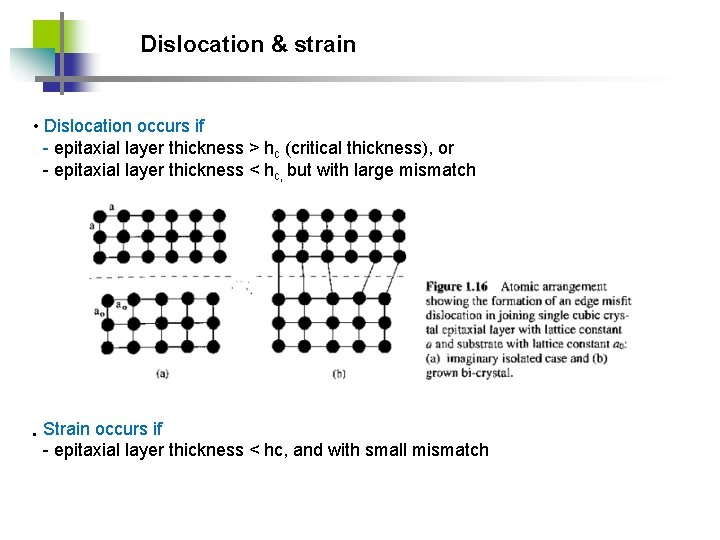
Dislocation & strain • Dislocation occurs if - epitaxial layer thickness > hc (critical thickness), or - epitaxial layer thickness < hc, but with large mismatch • Strain occurs if - epitaxial layer thickness < hc, and with small mismatch

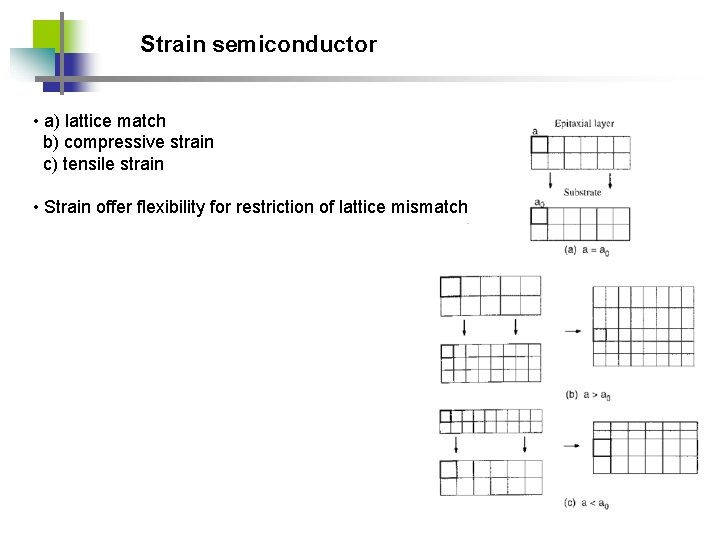
Strain semiconductor • a) lattice match b) compressive strain c) tensile strain • Strain offer flexibility for restriction of lattice mismatch

Crystal Growth • Bulk growth: - furnace growth - pulling technique • Epitaxial growth: - Liquid Phase Epitaxy (LPE) - Vapor Phase Epitaxy (VPE), or termed Chemical Vapor Deposition (CVD) - Molecular Beam Epitaxy (MBE)

Epitaxy • epi means “above” taxis means “in order manner” epitaxy can be translated to “to arrange upon” • with controlled thickness and doping • subtract acts as a seed crystal, deposited film takes on a lattice structure and orientation identical to the subtract • different from thin film deposition that deposit polycrystalline or amorphous film • - homoepitaxy: epi and subtract are with the same material epi layer more pure than subtract and have different doping level - hetroepitaxy: • used for - Si-based process for BJT and CMOS, or - compound semiconductors, such as Ga. As

Epitaxy Material Growth Methods • Liquid Phase Epitaxy • Vapor Phase Epitaxy (VPE), or termed Chemical Vapor Deposition (CVD) - formation of condensed phase from gas of different chemical composition - distinct from physical vapor deposition (PVD) such as sputtering, e-beam deposition, MBE (condensation occurs without chemical change) - gas stream through a reactor and interact on a heated subtract to grow epi layer • Molecular Beam Epitaxy

Doping of Semiconductors • Intrinsic materials: undoped - Undoped materials by epitaxy technology have more carriers than in intrinsic material. e. g. Ga. As: 1013 /cm 3 (instrinsic carrier concentration: 1. 8 x 106 /cm 3) - impurity comes from source materials, carrier gases, process equipment, or subtract handle • Extrinsic materials: - n-type: III sub-lattice of III-V compound is substituted by V elements: impurity terms “donor” - p-type: V sub-lattice of III-V compound is substituted by III elements: impurity terms “acceptor” http: //www. siliconfareast. com/sigegaas. htm

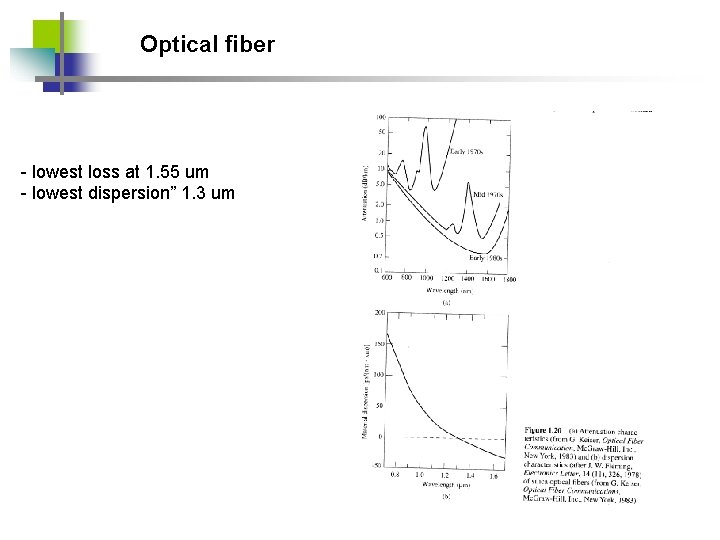
Optical fiber - lowest loss at 1. 55 um - lowest dispersion” 1. 3 um

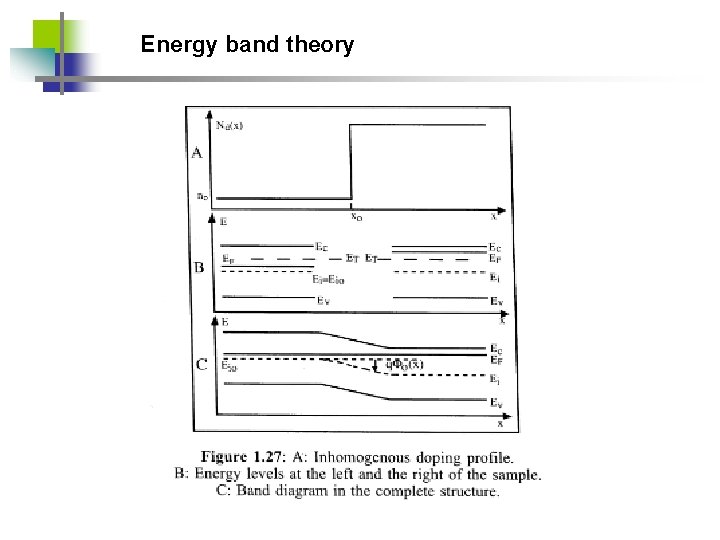
Energy band theory
 Deal or no deal machine
Deal or no deal machine Rudolf vizental
Rudolf vizental The great depression vocabulary
The great depression vocabulary New deal alphabet soup
New deal alphabet soup Preference for specific design control for procedural bias
Preference for specific design control for procedural bias Trumans fair deal
Trumans fair deal Watermelon short story
Watermelon short story The new deal affects many groups
The new deal affects many groups Apush new deal
Apush new deal Aruba deal registration
Aruba deal registration Motivul salcamului sara pe deal
Motivul salcamului sara pe deal Ruckus partner program
Ruckus partner program Aaa new deal purpose
Aaa new deal purpose Political cartoons fdr
Political cartoons fdr The new deal rrr
The new deal rrr Benjamin west contributed a great deal to american art:
Benjamin west contributed a great deal to american art: Modelo de bolman y deal
Modelo de bolman y deal Impacto de la gran depresion en chile
Impacto de la gran depresion en chile Ebra new deal
Ebra new deal Benefit2key
Benefit2key





































