CENTRIFUGATION OUTLINE n n n Basic theory Applications
CENTRIFUGATION

OUTLINE n n n Basic theory Applications Instrumentation lseidman@matcmadison. edu

SEPARATIONS n n This begins new topic: separations Prior to this, talked about measurements and solution making lseidman@matcmadison. edu

BIOSEPARATIONS n n n Separating and purifying biological materials Filtration and centrifugation Chromatography and electrophoresis also common methods lseidman@matcmadison. edu

PRINCIPLE Rate of settling of a particle, or the rate of separation of two immiscible liquids, is increased many times by the application of a centrifugal field (force) many times that of gravity. lseidman@matcmadison. edu

MANY APPLICATIONS n n Separate two immiscible liquids Isolate cellular organelles Isolate DNA, RNA, and proteins Isolate small particles including q q q Bacteria Viruses Cells lseidman@matcmadison. edu

SUPERNATANT AND A PELLET n n Supernatant is the liquid at the top Pellet is particles at the bottom lseidman@matcmadison. edu

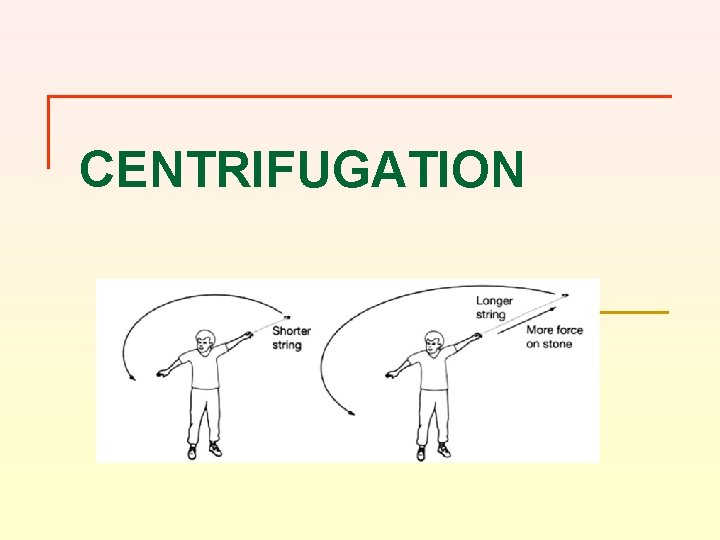
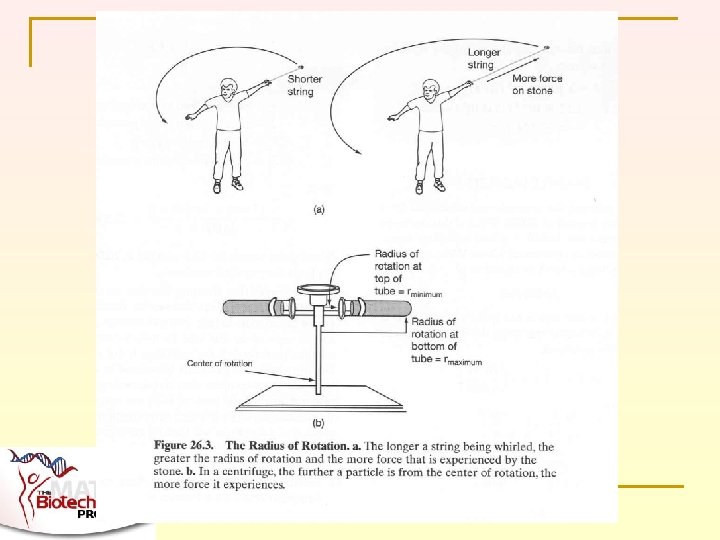
FORCE IN A CENTRIFUGE IS PROPORTIONAL TO TWO THINGS n n First, it depends on how fast the centrifuge spins Second, it depends on the radius of rotation – think about “crack the whip” lseidman@matcmadison. edu
lseidman@matcmadison. edu

RELATIVE CENTRIFUGAL FORCE, RCF n n Also = Xg RCF = 11. 17(r)(n/1000)2 Where r = radius in cm from centerline n = rotor speed in RPM, revolutions/minute lseidman@matcmadison. edu

CALCULATING RCF Suppose rmin = 3. 84 cm n raverage = 6. 47 cm n rmax = 9. 10 cm n N = 30, 000 RPM n Then, what is the RCF on a particle at rmin, rave, and rmax? n lseidman@matcmadison. edu

ANSWERS n n 38, 600 X g 65, 043 X g 91, 482 X g Don’t report RPM, report RCF because everyone’s centrifuge is different lseidman@matcmadison. edu

HOW FAST DOES A PARTICLE SEDIMENT? n It depends on: q q q RCFs in the centrifuge Size of particle Particle density Liquid viscosity lseidman@matcmadison. edu

n It turns out that if: q q q A particle is the same density as the liquid around it, the particle doesn’t move A particle is more dense than the liquid, it moves down the tube A particle is less dense than the liquid, it moves up! lseidman@matcmadison. edu

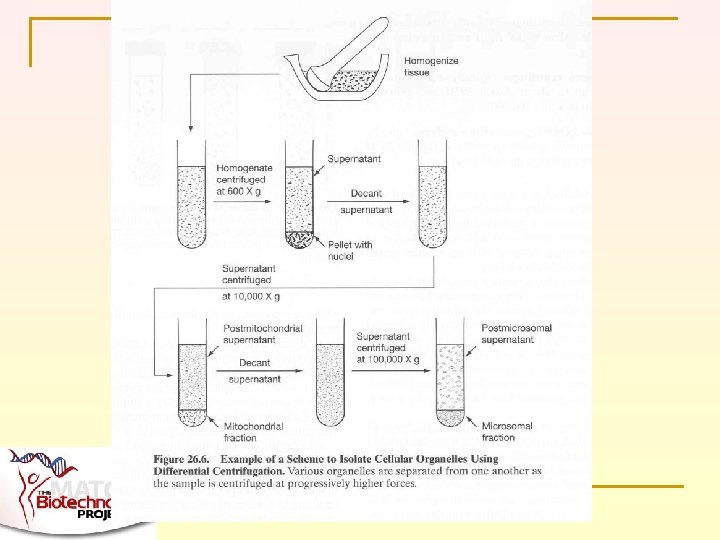
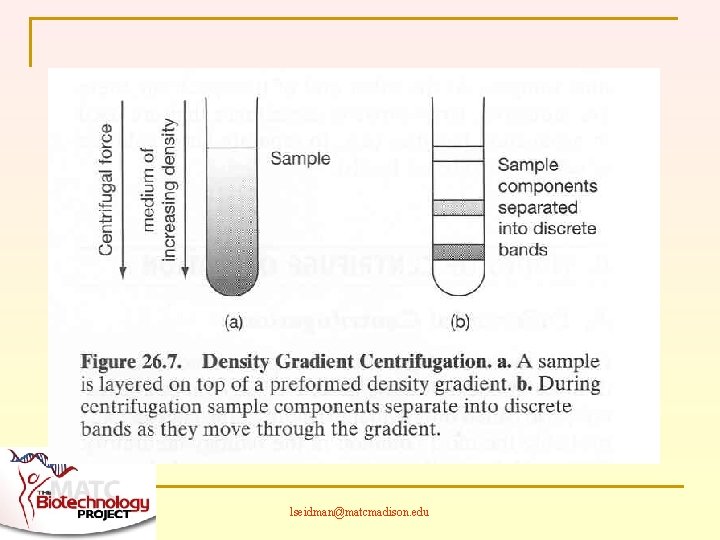
TWO BASIC MODES OF CENTRIFUGATION n n Most familiar is differential centrifugation Also density gradient centrifugation lseidman@matcmadison. edu
lseidman@matcmadison. edu
lseidman@matcmadison. edu

n On the next slide there is an excerpt from a research article. Explain how centrifugation is used in this research project. lseidman@matcmadison. edu

How Did Scientists Find Cytochrome C? Preparation of Mitochondria from mouse liver The mouse livers were removed after sacrifice and dounce homogenized in ice-cold mitochondria isolation buffer (MIB) containing 250 m. M mannitol, 0. 5 m. M EGTA, 5 m. M HEPES, and 0. 1% (w/v) BSA (p. H 7. 2) supplemented with the protease inhibitors of leupeptin (1 mg/ml), pepstatin A (1 mg/ml), antipain (50 mg/ml), and PMSF (0. 1 m. M). Unbroken cells and nuclei were pelleted by centrifugation at 600 g for 5 min at 4 o. C. The supernatants were further centrifuged at 10, 000 g for 10 min at 4 o. C to pellet the mitochondria. The mitochondria pellet was resuspended in 4 ml MIB and loaded onto a continuous Percoll gradient consisted of 30% (v/v) Percoll (Sigma), 225 m. M mannitol, 25 m. M HEPES, 0. 5 m. M EGTA, and 0. 1% (w/v) BSA (p. H 7. 2). The suspension/gradient was centrifuged at 40, 000 g for 1 hr. The mitochondria were removed from the brownish band at 1. 10 g/ml with a transfer pipette. The mitochondrial pellets were washed with MIB by centrifuging for 10 min at 6300 g at 4 o. C. The mitochondria were then resuspended gently in mitochondria resuspension buffer containing 400 m. M mannitol, 10 m. M KH 2 PO 4, and 50 m. M Tris-HCl (p. H 7. 2) with 5 mg/ml BSA and stored on ice for up to 4 hr. http: //www. swmed. edu/home_pages/wanglab/Wanglab-pic/protocols. htm lseidman@matcmadison. edu

INSTRUMENTATION n n n Lots of types Some go faster and some slower Some can take a lot of volume, others little Some allow temperature control Some allow you to add sample as centrifuge is running Some are specific for pathogens or whenever aerosols must be avoided lseidman@matcmadison. edu

TERMINOLOGY n n n Desktop, or clinical centrifuges <10, 000 RPM Superspeeds, 10, 000 – 30, 000 RPMs (around 50, 000 Xg) Ultracentrifuges up to 80, 000 RPM and 500, 000 Xg lseidman@matcmadison. edu
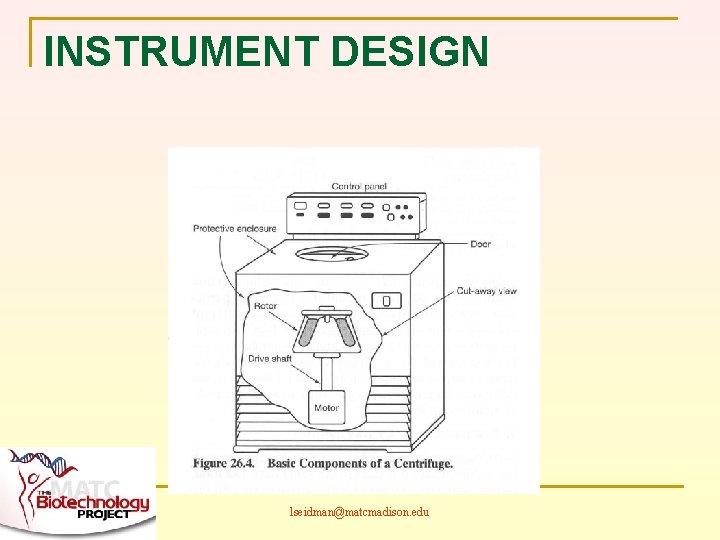
INSTRUMENT DESIGN lseidman@matcmadison. edu

SAFETY!! n n Centrifuges look sturdy, sort of like washing machines But, they are probably the most dangerous instrument any of you will use Also surprisingly easy to damage BE CAREFUL!!!!! lseidman@matcmadison. edu

TWO MAIN SAFETY CONCERNS n First is rotor coming off shaft - disaster lseidman@matcmadison. edu

ROTORS ARE FRAGILE n n Must withstand huge forces In an ultracentrifuge, a 1 gram particle “weighs” 0. 65 tons Any imperfection will weaken rotor Therefore: lseidman@matcmadison. edu

n n Expert design Proper use Retire at correct time Derate (run slower) when necessary lseidman@matcmadison. edu

PROPER USE OF ROTORS n n n Every rotor has a maximum speed As high speed rotors age, derate With ultrarotors retire them after certain age or number of revolutions Log books record every use and revolution Overspeed discs on bottom lseidman@matcmadison. edu

n n Purchase the correct rotors for your application Derate them as necessary – follow manufacturer’s directions Balance, balance Check your textbook, p. 561 for guidelines lseidman@matcmadison. edu

BIGGEST CHALLENGES IS TO READ THE CATALOGS n n n Rotor and tubes must match application Rotor and tubes must match centrifuge Rotor and tubes each have maximum RPMs at which can use them – and may be different q Use the slower value lseidman@matcmadison. edu

n May need adaptors to fit certain tubes into certain rotors q q This is because rotors are engineered to take varying size and styles of tubes Makes them versatile, but also requires complex combinations of adaptors and tubes lseidman@matcmadison. edu

GENERAL RULES n Protect the rotors from: q q Scratches Moisture Spills Alkaline detergents (like Countoff, for radioisotopes) lseidman@matcmadison. edu

n n n Follow manufacturer’s directions and NEVER! spin faster than is supposed to go Select tubes that can handle the samples and speeds you are using Keep your hands and hair out of centrifuges! Keep your hands out of rotors Wear glasses Make sure you know what you are doing lseidman@matcmadison. edu

MUCH MORE SUBTLE, BUT ALSO DANGEROUS, n n Aerosols Inevitable with normal centrifuge and very high levels are released if accident occurs Special centrifuges use special rotors, caps, and seals that prevent leakage Containment lseidman@matcmadison. edu
- Slides: 33