Central Vein in the Clinic Next Steps Daniel













- Slides: 19

Central Vein in the Clinic Next Steps Daniel Ontaneda MD MSc Cleveland Clinic Mellen Center for Multiple Sclerosis

Disclosures • Grant Support: NIH, Race to Erase, Patient Centered Outcome Research Institute, National Multiple Sclerosis Society, Genentech, Genzyme, Novartis. Consulting Biogen Idec, Genentech, Novartis. • Work presented was supported by Race to Erase MS Foundation

Challenges in diagnosis and misdiagnosis • Costs associated with MS total over $48. 4 billion annually, and 40% of direct care expense is related to disease modifying therapy (DMT) cost. 1 • Up to 20% of individuals referred for a diagnosis of MS are incorrectly diagnosed with MS. 2 • >2/3 of misdiagnosed patients are exposed to unnecessary and sometimes life-threatening risks associated with DMTs 2 1. Gooch CL. The burden of neurological disease in the United States: A summary report and call to action. Ann Neurol 2017; 81: 479– 84. 2. Solomon AJ, The contemporary spectrum of multiple sclerosis misdiagnosis: A multicenter study. Neurology 2016; 87: 1393– 9.

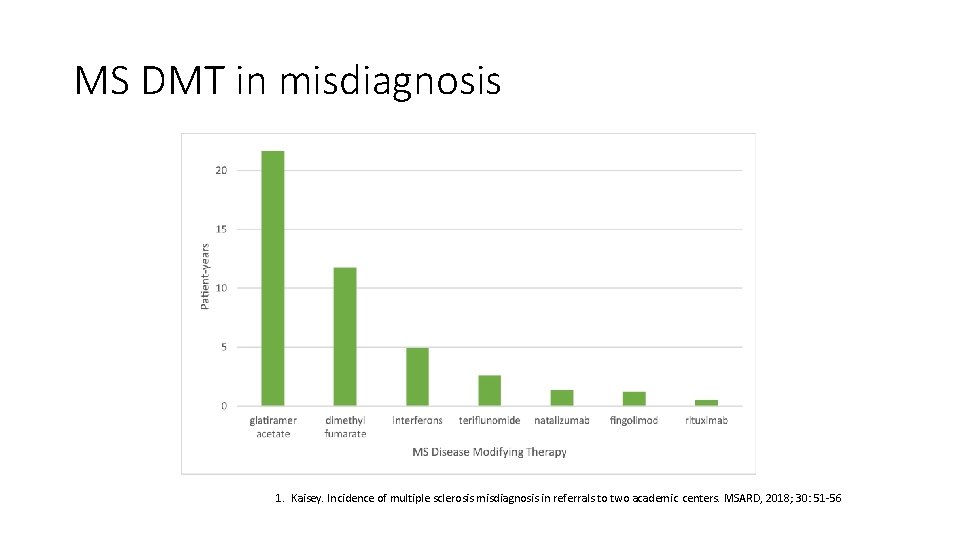
MS DMT in misdiagnosis 1. Kaisey. Incidence of multiple sclerosis misdiagnosis in referrals to two academic centers. MSARD, 2018; 30: 51 -56

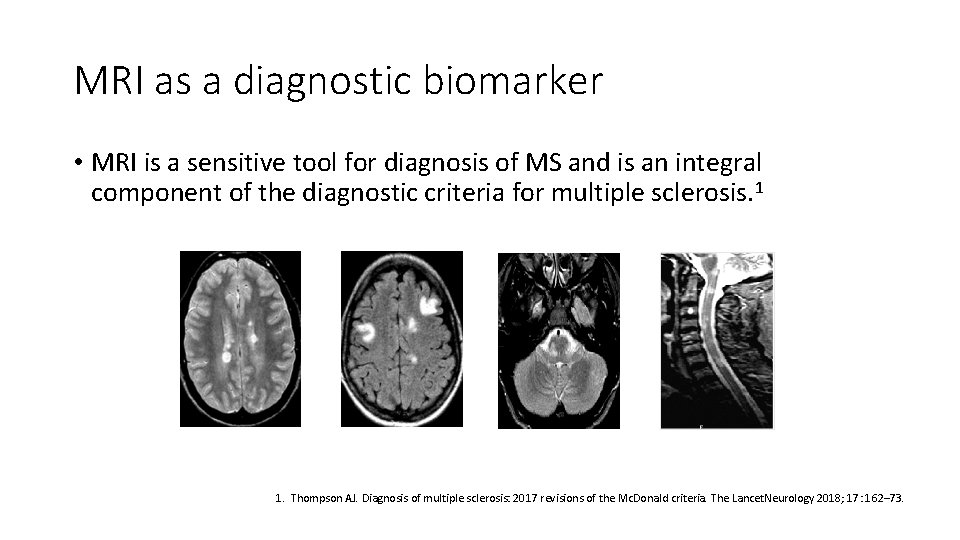
MRI as a diagnostic biomarker • MRI is a sensitive tool for diagnosis of MS and is an integral component of the diagnostic criteria for multiple sclerosis. 1 1. Thompson AJ. Diagnosis of multiple sclerosis: 2017 revisions of the Mc. Donald criteria. The Lancet. Neurology 2018; 17: 162– 73.

Problems with MRI implementation • Approximately half of individuals referred to an MS clinic, present with atypical syndromes (not typical clinically isolated syndrome). 1 • Increasing diagnostic sensitivity may have come at the price of decreased specificity. 2 • MRI criteria have specificity of 32% for dissemination in space (DIS) and 42% for dissemination in time (DIT). 3 1. Kelly SB. Using atypical symptoms and red flags to identify non-demyelinating disease. J Neurol Neurosurg Psychiatry 2012; 83: 44– 8 2. Rommer PS. Applying the 2017 Mc. Donald diagnostic criteria for multiple sclerosis. Lancet Neurol 2018; 17: 497– 8. 3. Filippi M, Prediction of a multiple sclerosis diagnosis in patients with clinically isolated syndrome using the 2016 MAGNIMS and 2010 Mc. Donald criteria: a retrospective study. Lancet Neurol 2018; 17: 133– 42.

MRI/Central Vein • Misdiagnosis appears to be mainly due to over interpretation of abnormal MRI findings • CVS may differentiate: • • 85% of white matter lesions in MS 12 8% small vessel ischemic disease 13 34 % Migraine 14 14% Other inflammatory or autoimmune diseases 12 Sati, Nature Reviews Neurology 2016 1. Maggi P. Central vein sign differentiates Multiple Sclerosis from central nervous system inflammatory vasculopathies. Ann Neurol 2018; 83: 283– 94. 2. Mistry N. Imaging central veins in brain lesions with 3 -T T 2*-weighted magnetic resonance imaging differentiates multiple sclerosis from microangiopathic brain lesions. Mult Scler 2016; 22: 1289– 96. 3. Solomon AJ. ‘Central vessel sign’ on 3 T FLAIR* MRI for the differentiation of multiple sclerosis from migraine. Ann Clin Transl Neurol 2015; 3: 82– 7.

Central Vein: Next Steps • There is a significant unmet need for more specific and accurate diagnostic tests to facilitate early confirmation of a diagnosis of MS. • We propose a prospective evaluation of the central vein sign (CVS) — which we hypothesize will reduce misdiagnosis, hasten early diagnosis, and simplify clinical decision-making.

Centr. Al Vein Sign in MS (CAVS-MS) • Multi-center prospective observational study • Cross-sectional initial pilot study across 10 sites • Establish feasibility of CVS implementation in a multi-site study

Aims Primary Objective: • Establish the CNR of lesion-to-NAWM and central vein-to-lesion across the 10 different sites using 3 T FLAIR* imaging in subjects with a clinical or radiological suspicion of MS. Secondary Objectives: • Investigate the difference in CNR identified in the primary objective between precontrast FLAIR* imaging and post contrast FLAIR* imaging to identify whether gadolinium injection is required for central vein detection. • Determine the reproducibility of different methods for detection of positive CVS across sites. • Determine the sensitivity and specificity of the different methods for the diagnosis of MS compared to the Mc. Donald 2010 MS criteria.

Study Population • Individuals referred to an MS center based on a clinical or radiological suspicion of MS • Broad inclusion criteria, total sample size 30 • Sites • • • Cleveland Clinic Johns Hopkins University of California San Francisco University of Texas Houston University of Toronto (St. Michael’s Hospital) University of Vermont • • University of Southern California Cedars Sinai Yale University of Pennsylvania

Progress • Study funded by Race to Erase MS Foundation • 30 subjects enrolled across the different sites • Development of software platform for rating of central veins through imaging software partner: QMENTA • Study coordination Cleveland Clinic • Central image analysis at NIH (Reich/Sati) • Statistical analysis University of Pennsylvania (Shinohara)

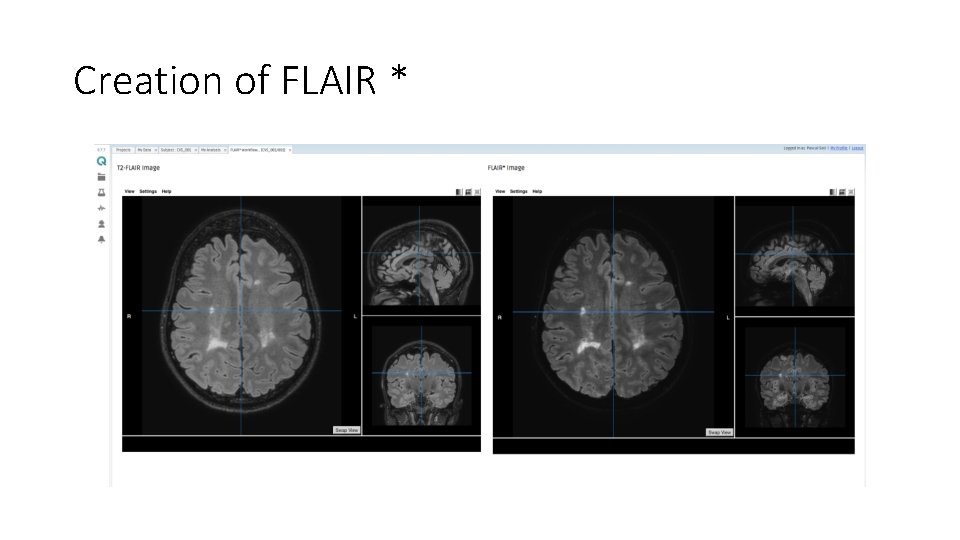
Creation of FLAIR *

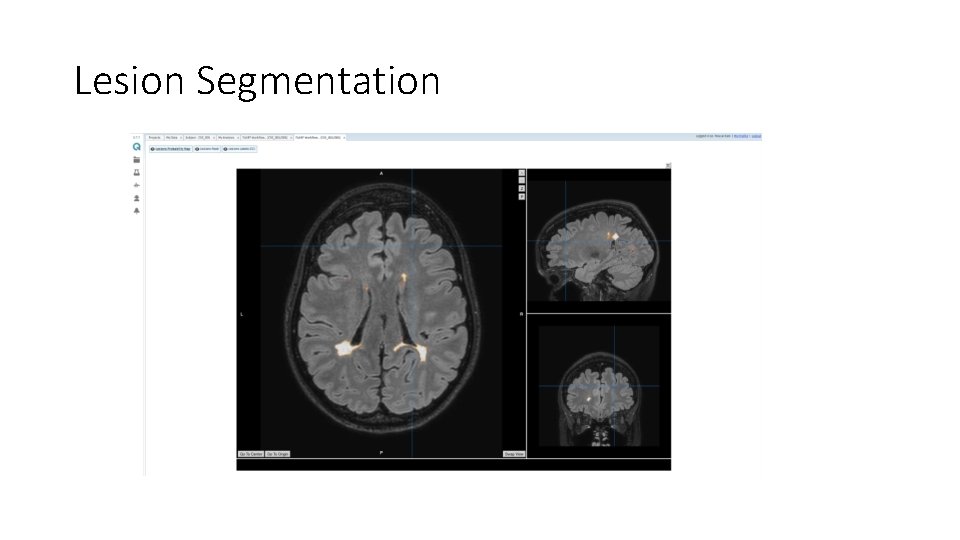
Lesion Segmentation

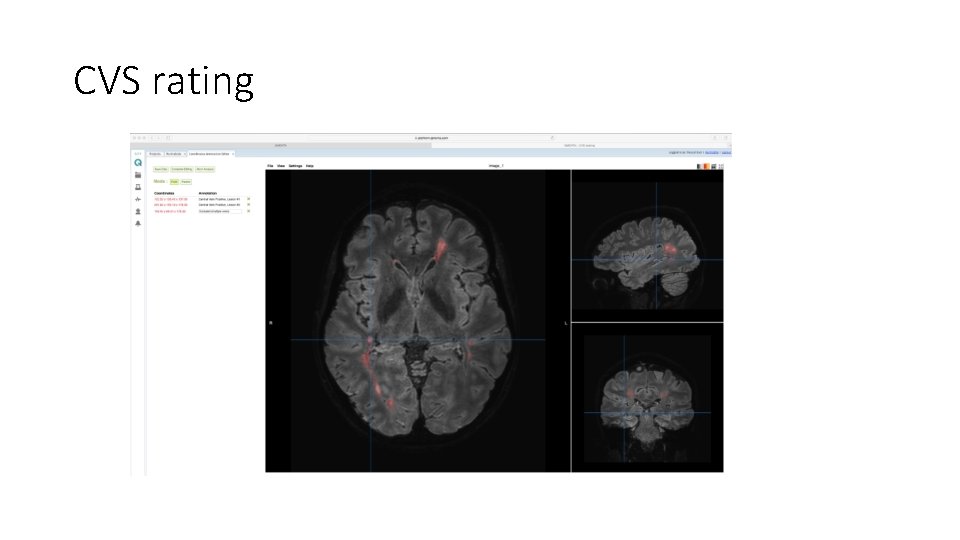
CVS rating

Prospective Study AIMS • Aim 1: To determine if incorporation of CVS for the diagnosis of MS improves diagnostic accuracy and hastens diagnosis in individuals presenting with typical first clinical events. • Aim 2: To determine if incorporation of CVS for the diagnosis of MS improves specificity among individuals presenting with atypical syndromes. • Aim 3: Central vein as a predictor of clinical/MRI disease activity associated with disability in MS.

Conclusions • Low specificity and misuse of diagnostic criteria may expose mis diagnosed patients to risk and increase health care costs • Central vein sign is a tool that offers promise both for increasing specificity, and perhaps enabling earlier diagnosis of MS. • Studies will determine if central vein sign can be incorporated into the diagnostic criteria • NIH is working with MRI manufacturers to make sequences available for disseminated clinical use

Acknowledgements • NAIMS • NIH • Pascal Sati, Danny Reich • University of Pennsylvania • Taki Shinohara • University of Vermont • Andy Solomon • Cedars Sinai • Nancy Sicotte

QUESTIONS