CENTRAL MEDICAL STORES Merapelo Baruti CMS Deputy Manager

CENTRAL MEDICAL STORES Merapelo Baruti CMS Deputy Manager

Topics covered • • • CMS overview Drug Supply Chain Management Product Sourcing Contract Management Challenges

CENTRAL MEDICAL STORES (CMS) q CMS is public sector entity which is responsible for Procurement, storage and distribution of Pharmaceuticals and other health commodities in public sector q 4 departments – Procurement, Warehouse (Technical Operation), Quality Assurance and Corporate services. q Staffing-166 staff, q 700 customers (public facilities, Mission and Mine Hospitals) q Process approx. 10, 000 items per month (Schedule Orders & Emergency Orders) q ISO 9001 certification with Botswana Bureau of Standards (BOBS) in November 2012. q Fully automated warehouse using WMS (Pulse) in inventory processing, from receipt, to the service delivery point (SDP).

CMS Conti. . . • >1, 500 products on the CMS catalogue + Special Orders (Non. Stockable) • Budgets 2012 -13 – BWP 700 m ( USD 95 m) v. P 408 million for Essential Meds and Dressings v. P 238 million for ARVs and Lab commodities (NACA) + Merck Donation v. P 17 million for ARVs (PEPFAR) & Training
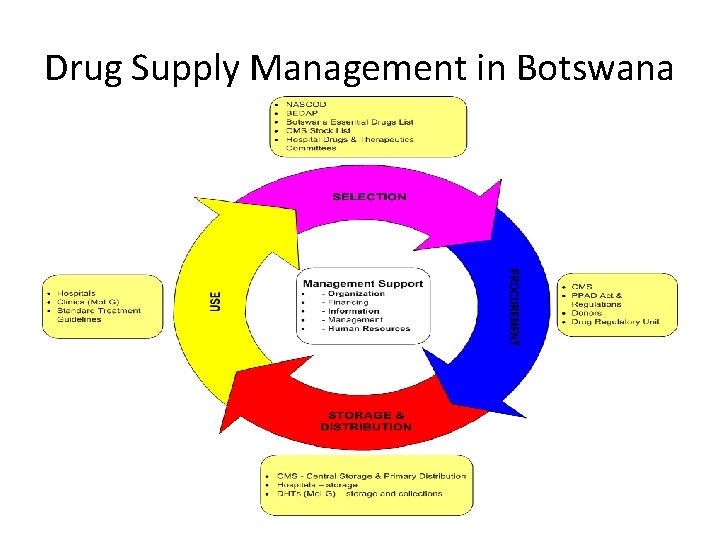
Drug Supply Management in Botswana

Product Sourcing Procurement Plan Ø All products (642 product lines) planned to be on a 2 years framework contracts Ø 10 drugs tenders planed for 2012/2013 financial year (450 products on framework) Ø Remaining 350 will be covered 2013/14 financial (including non stockable with high demand but no decision to declassify them)

Product Sourcing Ø Product sourcing is by generic description Ø Commonly used methods of procurement – Open domestic tender- default method of procurement – Restricted – Direct procurement – Quotations proposal method – Micro-procurement method Ø ARVs - Restricted method of tendering is preferred from Manufacturers who are o WHO or US-FDA prequalified o CHAI (price benchmarking)

Contract Management Contract agreement for purchases above 50, 000 pula 1. Supply Relationship Management (SRM) • Product availability and consumption data • Product visibility and pipeline information • Supplier relationship management on the key suppliers • Monitor over due orders (stock safety levels at 6 months)

Contract Management 2. End of Activity Reports for all the procurement performed 3. Payment • Letter of credit • 30 day payment (EFT)

Challenges High cost of drugs Lack of manufacturers Products in the list not DRU registered Which products in the essential list are protected by valid patent? • Access to patent data base? ? • •

Ke a Leboga Thank you
- Slides: 11