Central Intravenous Line Placement Wendy Blount DVM Practical
Central Intravenous Line Placement Wendy Blount, DVM
Practical Hematology 1. Anemia 101 2. Blood Loss Anemia 3. Hemolysis 4. Non-Regenerative Anemia 5. Transfusion Medicine 6. Polycythemia 7. Bone Marrow Disease 8. Coagulopathy 9. Central IV Lines 10. Leukophilia 11. Leukopenias 12. Splenic Disease

Why Place a Central Line? • When serial blood values are needed. – Avoids pain, trauma and bruising – Can get blood from a fractious cat without actually touching it – Makes repeated blood draws from difficult animals possible – Maintains integrity of the veins for easy placement of the next IV catheter

Why Place a Central Line? • When do we need serial values? – Diabetic ketoacidosis – Renal failure requiring diuresis – Liver failure – hypoproteinemia – “mean as a snake” diabetics who need a glucose curve – Hemolytic anemia

Why Place a Central Line? • When you need central venous access – Drugs that are caustic • Doxycycline IV • Diazepam CRI fro severe seizures – Total Parenteral Nutrition (TPN) – Partial Parenteral Nutrition (PPN) – Monitoring Central Venous Pressure (CVP) • CVP = pressure in the RA • When giving IV fluids to dogs with right congestive heart failure

Why Place a Central Line? • Central Venous Pressure – Normal • 5 -8 cm H 20 in dogs • 2 -3 cm H 20 in cats – Increased CVP can result in signs of right heart failure • >10 cm in dogs • >8 cm in cats – Trends are probably more important than absolute values – Approximated by caval bounce on AFAST® DH view
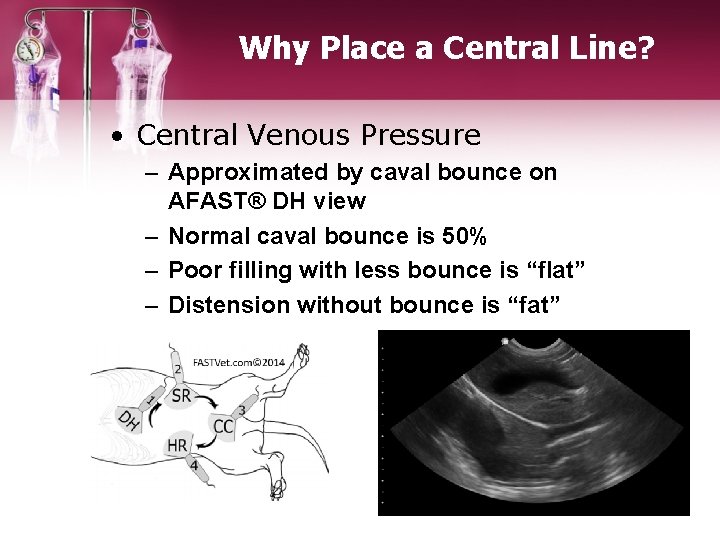
Why Place a Central Line? • Central Venous Pressure – Approximated by caval bounce on AFAST® DH view – Normal caval bounce is 50% – Poor filling with less bounce is “flat” – Distension without bounce is “fat”
Why Place a Central Line? • Central Venous Pressure

Videos Medial saphenous in a cat Through the needle catheter w/ clam shell

Stephen Garner Nacogdoches TX

Videos Jugular vein in the dog Seldinger Guidewire Technique

Videos Jugular vein in the dog Seldinger Guidewire Technique • • J 1040 – 20 g x 8 cm J 1040 A – 20 g x 13 cm J 1041 – 18 g x 8 cm J 1041 A – 18 g x 13 cm J 1042 – 16 g x 13 cm J 1042 A – 16 g x 20 cm J 1043, J 1043 A Also Double Lumen and Triple Lumen caths

Videos Jugular vein in the dog Tear Away Introducer Technique

Videos Jugular vein in the dog Tear Away Introducer Technique • • J 1437 – 20 g x 5 inch J 1437 A – 18 g x 6 cm J 1437 B – 18 g x 8 cm J 1437 C – 18 g x 12 cm J 1437 D – 16 g x 8 cm J 1437 E – 16 g x 12 cm Also Double Lumen caths
Around the Needle Catheter • Angio. Cath® style • Standard Teflon large bore IV catheter – 14 -18 gauge x 3 -4 inches – Smaller for kittens, puppies, toy breeds • J 0457 A – Pebax® 14 g x 5. 5 inch • J 0457 B – Pebax® 10 g 5. 5 inch
Around the Needle Catheter • Angio. Cath® style • • • J 0458 A – polyurethane 14 g x 5. 25 inch J 0458 B – polyurethane 16 g x 5. 25 inch J 0458 C – polyurethane 16 g x 3 inch J 0458 D – polyurethane 14 g x 3. 5 inch J 0458 E – polyurethane 12 g x 5. 25 inch J 0458 F – polyurethane 18 g x 2. 5 inch
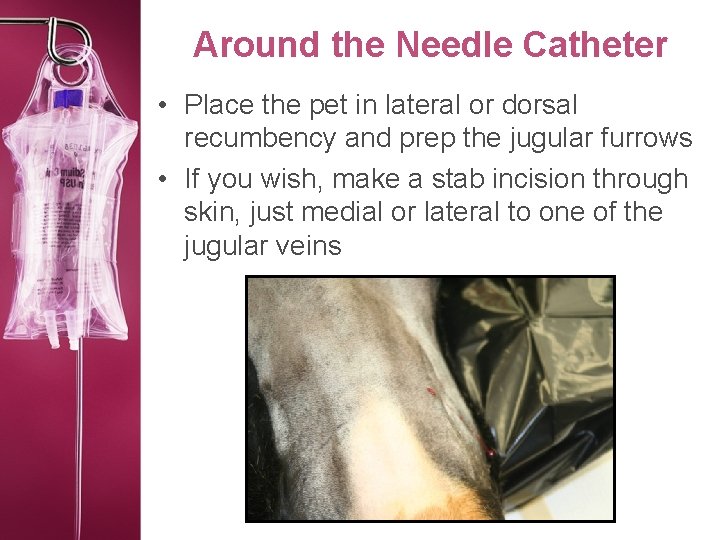
Around the Needle Catheter • Place the pet in lateral or dorsal recumbency and prep the jugular furrows • If you wish, make a stab incision through skin, just medial or lateral to one of the jugular veins
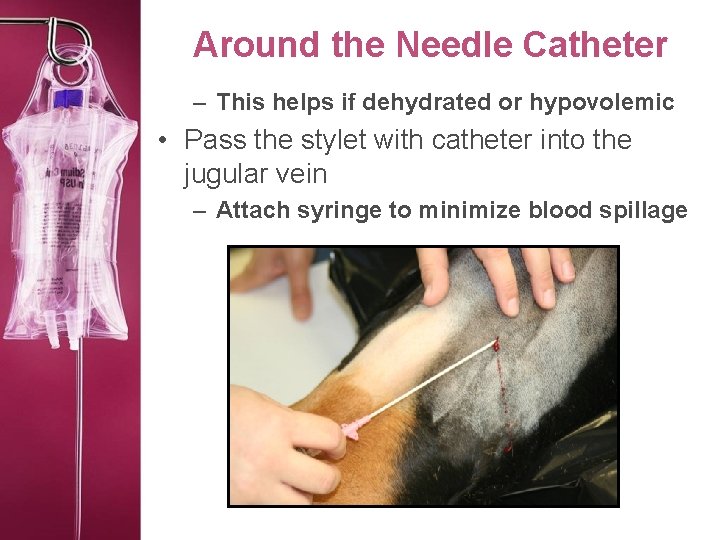
Around the Needle Catheter – This helps if dehydrated or hypovolemic • Pass the stylet with catheter into the jugular vein – Attach syringe to minimize blood spillage
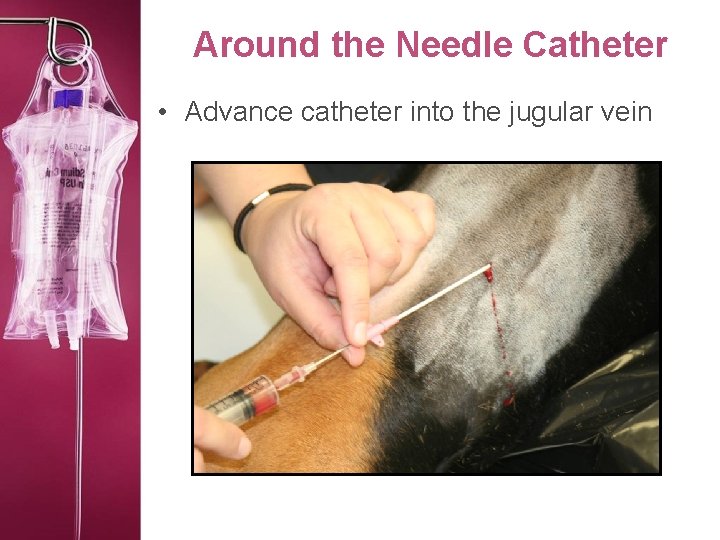
Around the Needle Catheter • Advance catheter into the jugular vein
Around the Needle Catheter
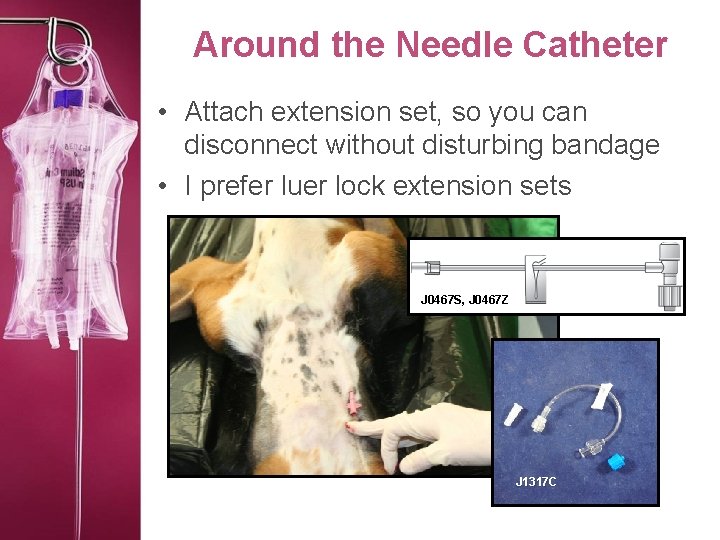
Around the Needle Catheter • Attach extension set, so you can disconnect without disturbing bandage • I prefer luer lock extension sets J 0467 S, J 0467 Z J 1317 C

Around the Needle Catheter • Tape or suture catheter to the skin • Recover from sedation • Bandage – – Antibiotic dressing Cast padding Roll gauze Vetrap
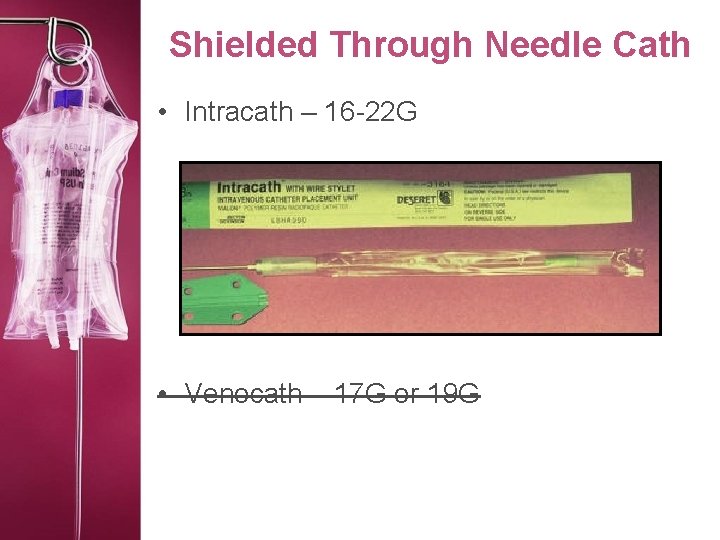
Shielded Through Needle Cath • Intracath – 16 -22 G • Venocath – 17 G or 19 G
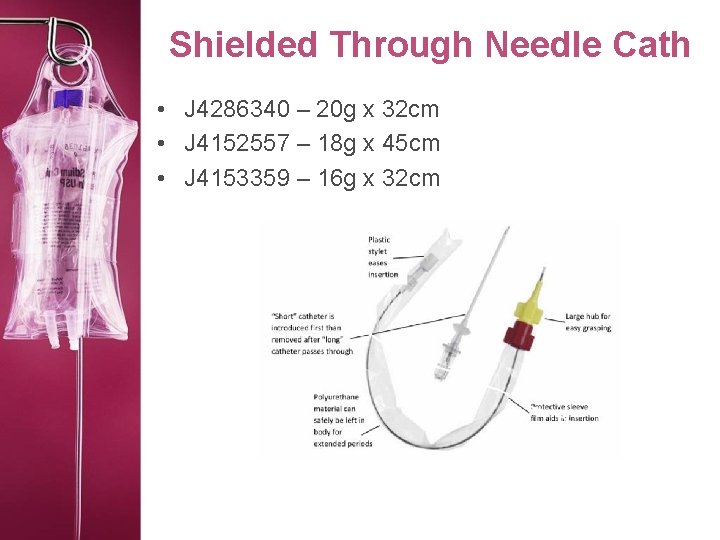
Shielded Through Needle Cath • J 4286340 – 20 g x 32 cm • J 4152557 – 18 g x 45 cm • J 4153359 – 16 g x 32 cm

Shielded Through Needle Cath • Three layers: – Guide wire stylet (inner most) – Polypropylene catheter – Needle stylet (outer most) • Needle Guard – Clamps around the needle like a clamshell, to keep the needle from cutting the catheter in half
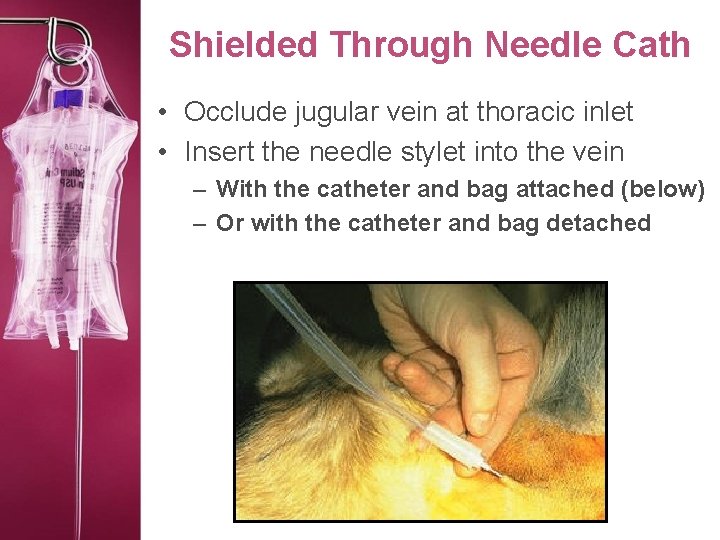
Shielded Through Needle Cath • Occlude jugular vein at thoracic inlet • Insert the needle stylet into the vein – With the catheter and bag attached (below) – Or with the catheter and bag detached
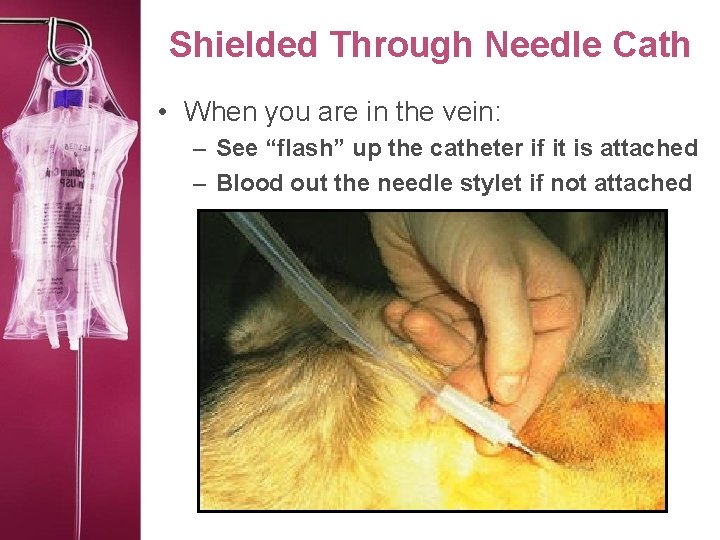
Shielded Through Needle Cath • When you are in the vein: – See “flash” up the catheter if it is attached – Blood out the needle stylet if not attached

Shielded Through Needle Cath • Stop occluding the jugular vein – Decreases flow of blood out the hub if bag not attached • Thread the catheter by “milking it” through the sterile bag, or threading it into the open needle hub – Thread in at least 3 -4 inches in a small dog or cat – to the 4 th ICS – All the way in if dog is big enough – To the right atrium, or least well into the thoracic inlet

Shielded Through Needle Cath • Place a 4 x 4 gauze at the venipuncture site an apply gentle pressure • Withdraw the needle, leaving the catheter in place. • Remove the protective bag • Seat the catheter hub firmly into the needle stylet hub – I like to use a drop of tissue glue to secure them together – If there is movement here, the catheter can be sheared off the hub

Shielded Through Needle Cath • Apply the needle guard – Secure closed with white tape • Remove the wire stylet – MAKE SURE the cath & needle hubs are attached (b attached to c) to each other before the stylet (a) is removed – If not, you can remove the catheter (b) with the stylet (a) by mistake

Shielded Through Needle Cath • attach a 10 -12 cc syringe filled with saline or LRS – Flush to make sure the catheter is patent – Aspirate to make sure catheter is patent – flush and aspirate about every minute, to make sure catheter is still patent and not kinked, while wrapping
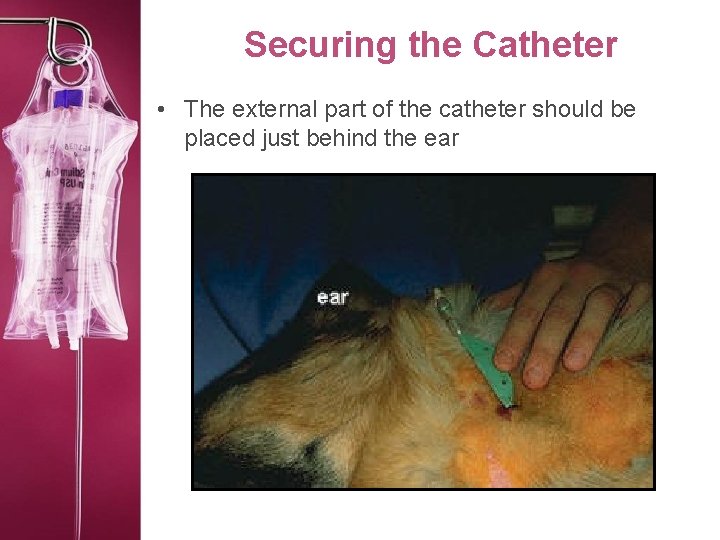
Securing the Catheter • The external part of the catheter should be placed just behind the ear

Securing the Catheter • Place a white tape butterfly on the needle guard – Secures the needle guard closed – Used to suture the guard to the skin – I don’t use the suture holes in the needle guard – Vetafil 2 -0 or other Nonabsorbable suture

Securing the Catheter
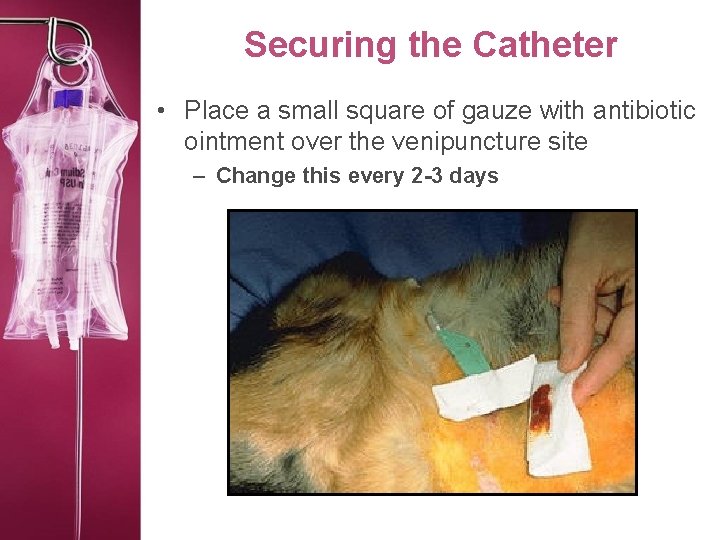
Securing the Catheter • Place a small square of gauze with antibiotic ointment over the venipuncture site – Change this every 2 -3 days

Securing the Catheter • Place ¼-1/2 inch padding between the needle guard and the skin – Cotton, gauze or cast padding - prevents sores – Be careful not to kink the catheter here
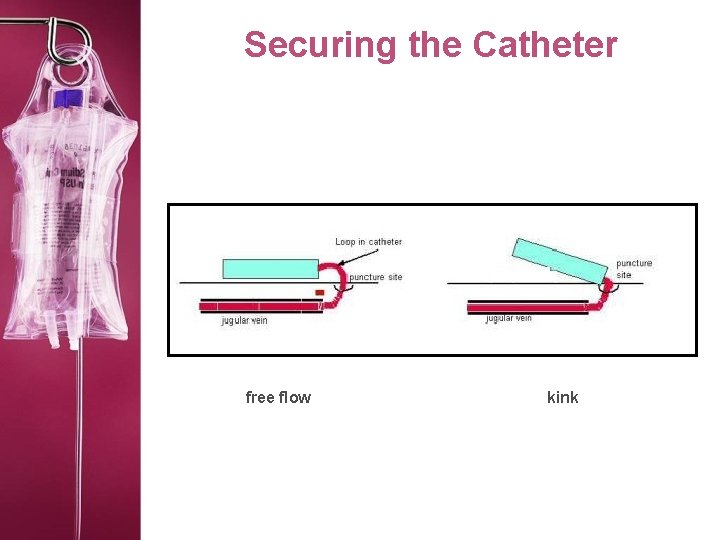
Securing the Catheter free flow kink

Securing the Catheter • REMEMBER, you are flushing and aspirating every minute or so as you go, with syringe full of saline attached to the catheter hub • Secure catheter to the neck with full circle white tape – Clip a “bridle path” in the fur if needed

Securing the Catheter • Attach the catheter hub to the outer layer of roll gauze with “split” white tape

Securing the Catheter • Outer layer of Vetrap – Cut hole over catheter – Can completely cover catheter with another loop of Vetrap when not in use.
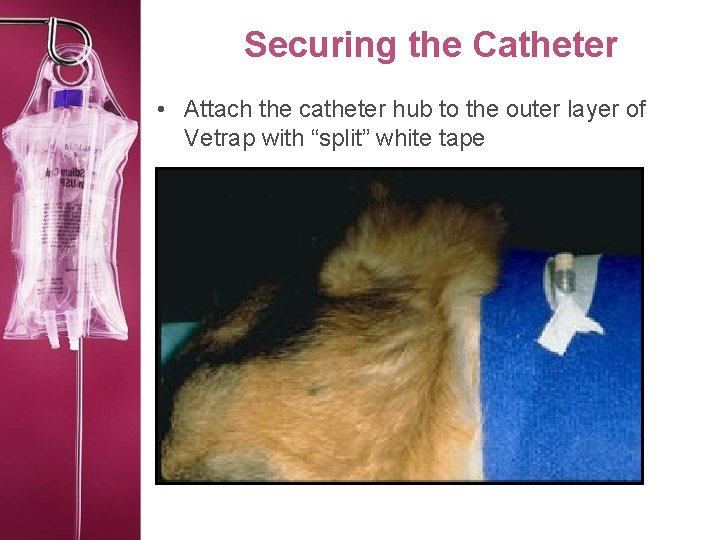
Securing the Catheter • Attach the catheter hub to the outer layer of Vetrap with “split” white tape

Securing the Catheter • These photos show bandaging while anesthetized. • If sedation is required, I prefer to tape while sedated, then finish the bandage after awake and sitting sternal • This prevents a bandage that is too tight
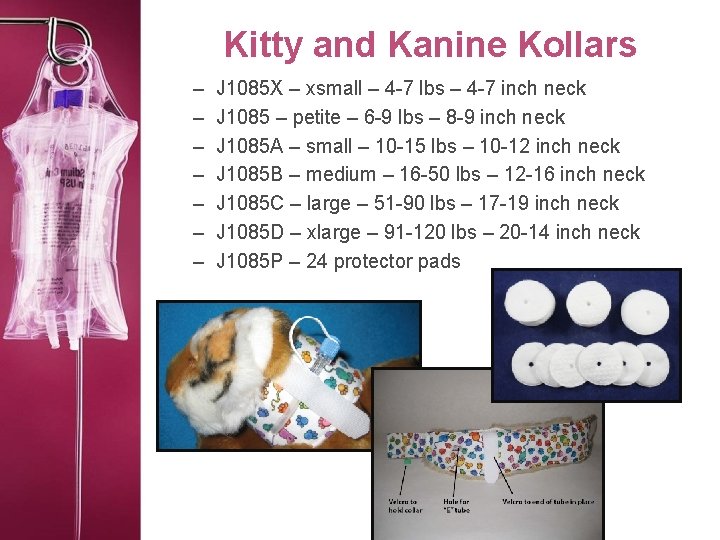
Kitty and Kanine Kollars – – – – J 1085 X – xsmall – 4 -7 lbs – 4 -7 inch neck J 1085 – petite – 6 -9 lbs – 8 -9 inch neck J 1085 A – small – 10 -15 lbs – 10 -12 inch neck J 1085 B – medium – 16 -50 lbs – 12 -16 inch neck J 1085 C – large – 51 -90 lbs – 17 -19 inch neck J 1085 D – xlarge – 91 -120 lbs – 20 -14 inch neck J 1085 P – 24 protector pads

Susan Johnson Mc. Dade TX
Taking Blood Samples • • Disconnect IV fluid line and cap Flush the catheter with heparinized 3 -5 cc saline Gently withdraw 5 -6 cc of blood (dump syringe) Gently withdraw needed sample – All values except platelets will be accurate • Gently replace 5 -6 cc of blood in “dump syringe” • Flush the catheter with 3 -5 cc saline

CVP Measurement • Equipment Needed – – – Bag of fluids Fluid administration set IV extension set Three-way stopcock Manometer If you don’t have a manometer, you can tape IV tubing to a ruler with cm marks on it • Flush patient’s IV catheter • Fill the IV extension set with fluid • Connect the IV extension set – To patient jugular catheter at one end – To 3 -way stopcock at the other end (side) – Stopcock off to patient

CVP Measurement

CVP Measurement • Connect manometer to 3 -way stop-cock (top) – This connection is the weakest point – Support the manometer when turning the stopcock • Connect the IV set and fill the line with fluids – One end to the fluid bag (one side) – The other end to the stopcock (other side)
CVP Measurement • Turn the stopcock off to manometer, and make sure fluid flows freely into the patient • Turn stopcock off to patient, and fill manometer with fluid – to at least 15 -20 cm – Make sure no air bubbles, which could cause vapor lock
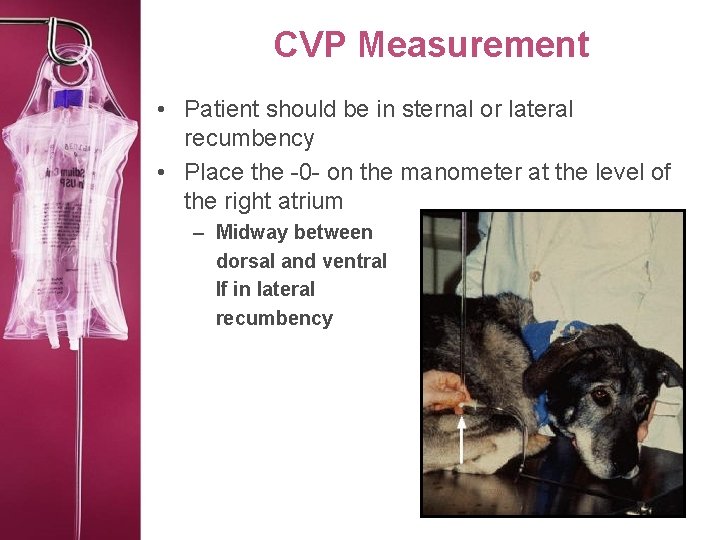
CVP Measurement • Patient should be in sternal or lateral recumbency • Place the -0 - on the manometer at the level of the right atrium – Midway between dorsal and ventral If in lateral recumbency

CVP Measurement • Turn stopcock off to fluids, and allow fluid to fall until it rests at the patient’s CVP – The meniscus will oscillate up and down as the heart beats and the patient breathes

Acknowledgements Maeckelbergh VA, Acierno MJ. Comparison of prothrombin time, activated partial thromboplastin time, and fibrinogen concentration in blood samples collected via an intravenous catheter versus direct venipuncture in dogs. Am J Vet Res 2008; 69: 868– 873.
- Slides: 52