Center for Global Health MER and WHO TBHIV

Center for Global Health MER and WHO TB/HIV Indicator Reference Guide: TB/HIV Diagnostic and Care Cascade Division of Global HIV and TB TB Preventive Treatment Unit

Intended End-User(s) : CDC or other USG agency headquarters-based staff providing technical assistance, CDC or other USG agency country team programmatic staff, CDC or other USG agency country team strategic information staff, and other donor agencies and implementing partners Goals of Tool : 1. Provide TB/HIV cascade visual mapped with relevant PEPFAR and WHO indicators, potential data sources, and country example 2. Provide definitions for all TB/HIV cascade indicators 3. Provide details for recommended custom indicators 4. Provide data entry screen visuals for PEPFAR TB/HIV indicators 5. Provide detailed indicator guidance for relevant PEPFAR/WHO indicators Description : This document was developed as a reference for all TB/HIV PEPFAR Monitoring, Evaluation, and Reporting (MER) and WHO indicators to support understanding for TPT program reporting. This document includes details for how to define, collect, and report on these indicators. It also provides comparisons to show changes in the indicators from FY 17 to FY 18, as well as provides explanations of the disaggregates of the indicators. Instruction to end-users: Use these slides as a reference and/or training tool.
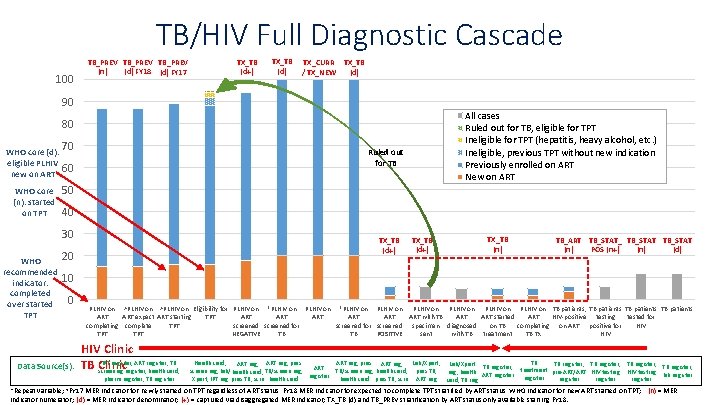
TB/HIV Full Diagnostic Cascade 100 TB_PREV (n) (d) FY 18 (d) FY 17 TX_TB (d+) TX_TB (d) TX_CURR / TX_NEW TX_TB (d) 90 All cases Ruled out for TB, eligible for TPT Ineligible for TPT (hepatitis, heavy alcohol, etc. ) Ineligible, previous TPT without new indication Previously enrolled on ART New on ART 80 70 WHO core (d): eligible PLHIV 60 new on ART WHO core (n): started on TPT Ruled out for TB 50 40 30 WHO recommended indicator: completed over started TPT TX_TB (d+) 20 TX_TB (d+) TX_TB (n) TB_ART TB_STAT_ TB_STAT POS (n+) (d) (n) 10 0 Data Source(s): PLHIV on ^PLHIV on Eligibility for PLHIV on *PLHIV on PLHIV on TB patients, TB patients ART expect ART starting TPT ART ART ART with TB ART started ART HIV-positive testing tested for completing complete TPT screened for screened specimen diagnosed on TB completing on ART positive for HIV TPT NEGATIVE TB TB POSITIVE sent with TB treatment TB Tx HIV Clinic IPT register, ART register, TB TB Clinic screening register, health card, pharm register, TB register Health card, ART reg, pres. ART reg, Lab/Xpert ART reg, pres TB register, ART screen reg, lab/ health card, TB/screen reg, health card, pres TB, reg, health ART register Xpert, IPT reg pres TB, scrn health card pres TB, scrn ART reg card, TB reg TB TB register, treatment pre-ART/ART HIV testing lab register register *Repeat variable; ^FY 17 MER indicator for newly started on TPT regardless of ART status, FY 18 MER indicator for expected to complete TPT stratified by ART status, WHO indicator for new ART started on TPT; (n) = MER indicator numerator; (d) = MER indicator denominator; (+) = captured via disaggregated MER indicator; TX_TB (d) and TB_PREV stratification by ART status only available starting FY 18.

Data Sources • PLHIV on ART (TX_CURR/TX_NEW) • Facility ART registers/databases, Program monitoring tools, Drug supply management systems, Pharmacies • PLHIV on ART screened for TB (TX_TB denominator) • ART registers, Facility-based TB screening registers, Presumptive TB registers, Patient passports/health cards • PLHIV on ART screened negative for TB (TX_TB denominator NEG) • ART registers, Facility-based TB screening registers, Presumptive TB registers, Patient passports/health cards • Eligibility for TPT (depends on country guidelines, contraindications) • Patient passports/health cards, Facility-based TB screening registers, lab registers, Gene. Xpert data collection systems, IPT registers • PLHIV on ART started on TPT (WHO numerator) • ART register or from separate TB screening or IPT register, Patient passports/health cards, Pharmacy registers; Screen positive/test negative for TB (TB specimen registers, lab registers, TB microscopy registers, Gene. Xpert data collection systems) • PLHIV on ART expected to complete TPT (TB_PREV denominator) • ART register or from separate TB screening or IPT register, Patient passports/health cards, Pharmacy registers; Screen positive/test negative for TB (TB specimen registers, lab registers, TB microscopy registers, Gene. Xpert data collection systems) • PLHIV on ART completing a course of TPT (TB_PREV numerator) • ART register or from separate TB screening or IPT register, Patient passports/health cards, Pharmacy registers
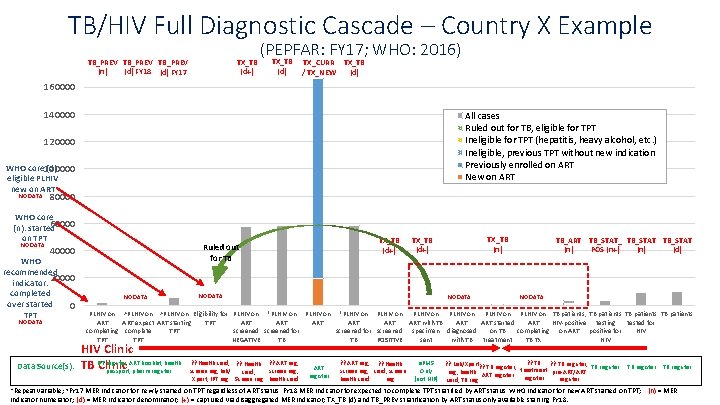
TB/HIV Full Diagnostic Cascade – Country X Example TB_PREV (n) (d) FY 18 (d) FY 17 TX_TB (d+) (PEPFAR: FY 17; WHO: 2016) TX_TB (d) TX_CURR / TX_NEW TX_TB (d) 160000 140000 All cases Ruled out for TB, eligible for TPT Ineligible for TPT (hepatitis, heavy alcohol, etc. ) Ineligible, previous TPT without new indication Previously enrolled on ART New on ART 120000 WHO core 100000 (d): eligible PLHIV new on ART NO DATA 80000 WHO core 60000 (n): started on TPT NO DATA WHO recommended indicator: 20000 completed over started 0 TPT NO DATA Data Source(s): TX_TB (d+) Ruled out for TB 40000 NO DATA TX_TB (n) TX_TB (d+) NO DATA TB_ART TB_STAT_ TB_STAT POS (n+) (d) (n) NO DATA PLHIV on ^PLHIV on Eligibility for PLHIV on *PLHIV on PLHIV on TB patients, TB patients ART expect ART starting TPT ART ART ART with TB ART started ART HIV-positive testing tested for completing complete TPT screened for screened specimen diagnosed on TB completing on ART positive for HIV TPT NEGATIVE TB TB POSITIVE sent with TB treatment TB Tx HIV Clinic register, ART booklet, health TB IPT Clinic passport, pharm register ? ? Health card, ? ? Health ? ? ART reg, screen reg, lab/ screen reg, card, Xpert, IPT reg Screen reg health card ART register ? ? ART reg, ? ? Health screen reg, card, screen health card reg e. PMS Only (not NIP) ? ? TB register, ? ? Lab/Xpert? ? TB register, TB register reg, health ART register treatment pre-ART/ART register card, TB register *Repeat variable; ^FY 17 MER indicator for newly started on TPT regardless of ART status, FY 18 MER indicator for expected to complete TPT stratified by ART status, WHO indicator for new ART started on TPT; (n) = MER indicator numerator; (d) = MER indicator denominator; (+) = captured via disaggregated MER indicator; TX_TB (d) and TB_PREV stratification by ART status only available starting FY 18.

Current Indicator Cheat Sheets

TB_STAT § Description – New and relapse TB cases with documented HIV status within reporting period – Reported semi-annually (Q 2 and Q 4) – Sum across Q 2 and Q 4 for annual total Description Disaggregates Numerator The number of new and relapse TB cases with documented HIV status Known HIV positives: (<15 F, <15 M, 15+F, 15+M); Newly tested HIV positives: (<15 F, <15 M, 15+F, 15+M); New negatives: (<15 F, <15 M, 15+F, 15+M) Denominator The number of new and relapse TB cases <15 F, <15 M, 15+F, 15+M

TB_ART § Description – HIV-positive new and relapse TB cases on ART during reporting period – Reported semi-annually (Q 2 and Q 4) – Sum across Q 2 and Q 4 for annual total Description Numerator The number of TB cases with documented HIV-positive status who start or continue ART Disaggregates New on ART; previous on ART <1, 1 -9, 10 -14 F, 10 -14 M, 15 -19 F, 15 -19 M, 20 -24 F, 20 -24 M, 25 -29 F, 25 -29 M, 30 -34 F, 30 -34 M, 35 -39 F, 35 -39 M, 40 -49 F, 40 -49 M, 50+F, 50+M

TX_CURR § Description – Adults and children currently receiving ART – Reported quarterly – Snapshot indicator (do not sum quarters, use Q 4 for annual total) Numerator FY 17 and FY 18 Data Disaggregates The number of adults and children currently receiving ART <1, 1 -9, 10 -14 F, 10 -14 M, 15 -19 F, 15 -19 M, 2024 F, 20 -24 M, 25 -29 F, 25 -29 M, 30 -34 F, 3034 M, 35 -39 F, 35 -39 M, 40 -49 F, 40 -49 M, 50+F, 50+M

TX_NEW § Description – Adults and children newly enrolled on ART – Reported quarterly – Sum across all quarters for annual total FY 17 and FY 18 Data Numerator The number of adults and children newly enrolled on ART Disaggregates Pregnant; Breastfeeding Confirmed diagnosis of TB <1, 1 -9, 10 -14 F, 10 -14 M, 15 -19 F, 15 -19 M, 20 -24 F, 20 -24 M, 25 -29 F, 25 -29 M, 30 -34 F, 30 -34 M, 3539 F, 35 -39 M, 40 -49 F, 40 -49 M, 50+F, 50+M
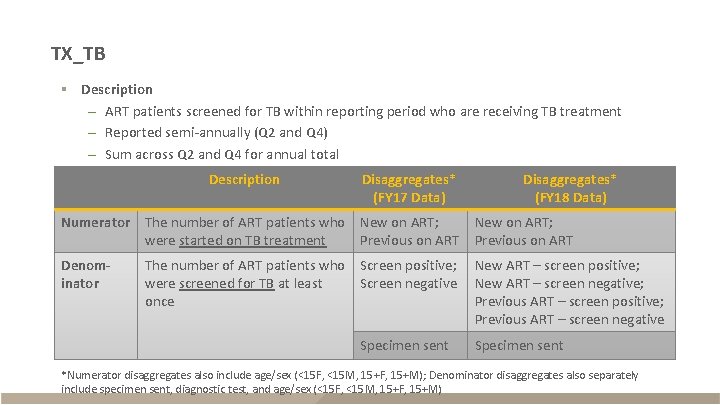
TX_TB § Description – ART patients screened for TB within reporting period who are receiving TB treatment – Reported semi-annually (Q 2 and Q 4) – Sum across Q 2 and Q 4 for annual total Description Disaggregates* (FY 17 Data) Disaggregates* (FY 18 Data) Numerator The number of ART patients who New on ART; were started on TB treatment Previous on ART New on ART; Previous on ART Denominator New ART – screen positive; New ART – screen negative; Previous ART – screen positive; Previous ART – screen negative The number of ART patients who Screen positive; were screened for TB at least Screen negative once Specimen sent *Numerator disaggregates also include age/sex (<15 F, <15 M, 15+F, 15+M); Denominator disaggregates also separately include specimen sent, diagnostic test, and age/sex (<15 F, <15 M, 15+F, 15+M)
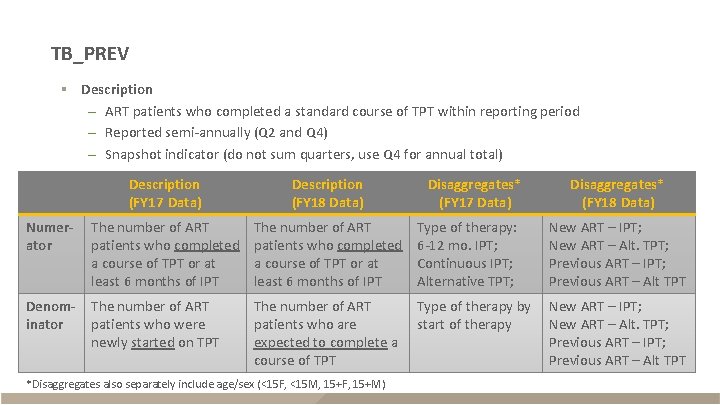
TB_PREV § Description – ART patients who completed a standard course of TPT within reporting period – Reported semi-annually (Q 2 and Q 4) – Snapshot indicator (do not sum quarters, use Q 4 for annual total) Description (FY 17 Data) Description (FY 18 Data) Numerator The number of ART patients who completed a course of TPT or at least 6 months of IPT Type of therapy: 6 -12 mo. IPT; Continuous IPT; Alternative TPT; New ART – IPT; New ART – Alt. TPT; Previous ART – IPT; Previous ART – Alt TPT Denominator The number of ART patients who were newly started on TPT The number of ART patients who are expected to complete a course of TPT Type of therapy by start of therapy New ART – IPT; New ART – Alt. TPT; Previous ART – IPT; Previous ART – Alt TPT *Disaggregates also separately include age/sex (<15 F, <15 M, 15+F, 15+M) Disaggregates* (FY 17 Data) Disaggregates* (FY 18 Data)

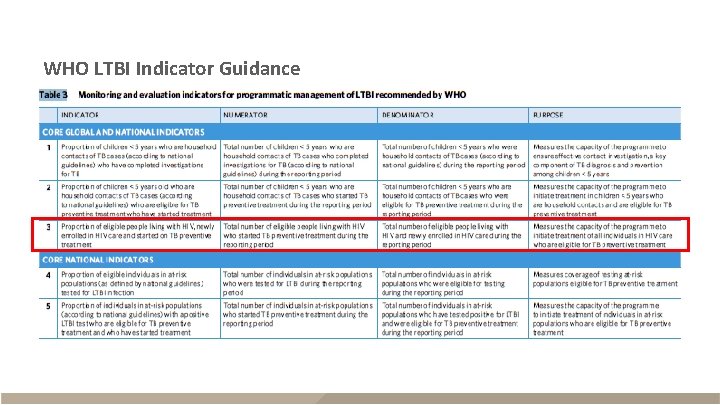
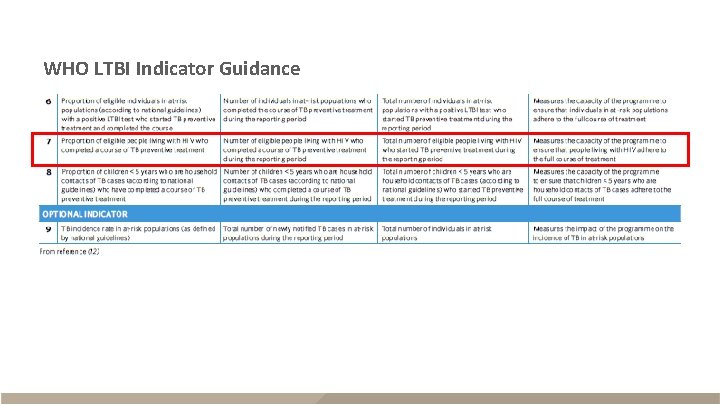
WHO: PLHIV starting TPT § Description – Eligible PLHIV newly enrolled in care started on TPT during the reporting period – Required core global indicator – Recorded daily, reported quarterly, consolidated annually for WHO Data since 2015 Numerator The number of eligible PLHIV newly enrolled in HIV care who started TPT Denominator The number of eligible PLHIV newly enrolled in HIV care

WHO: PLHIV completing TPT § Description – Eligible PLHIV who completed a course of TPT during the reporting period – Recommended core national indicator – Recorded daily, reported quarterly to national or sub-national level Data since 2015 Numerator The number of eligible PLHIV who completed a course of TPT Denominator The number of eligible PLHIV who started TPT § Information that affects recording, reporting, and interpretation of this indicator – Individuals who have adhered to and completed at least 80% of the dose within the recommended time frame are classified as completed

Recommended Custom Indicators

Eligibility for TPT § Description – ART patients eligible for initiation of TPT – Report semi-annually (Q 2 and Q 4) Description Numerator Denominator The number of ART patients eligible for TPT The total number of ART patients both eligible and ineligible for TPT Disaggregates New ART Eligible for TPT; Previous ART Eligible for TPT; <15 F/M, 15+F/M Ineligible for TPT (active TB, <15 F/M, 15+F/M contraindications) <15 F/M, 15+F/M Ineligible for TPT (active TB, <15 F/M, 15+F/M contraindications, previous TPT completion) New ART (<15 F, <15 M, 15+F, 15+M); Previous ART (<15 F, <15 M, 15+F, 15+M)

TPT initiation § Description – ART patients who initiated a standard course of TPT within reporting period – Report semi-annually (Q 2 and Q 4) Numerator Description Disaggregates The number of ART patients who were newly started on TPT New ART (<15 F, <15 M, 15+F, 15+M); Previous ART (<15 F, <15 M, 15+F, 15+M)

Cumulative PLHIV with at least 6 months TPT § Description – ART patients who have ever completed a standard course of TPT as of the reporting period – Report semi-annually (Q 2 and Q 4) Description Numerator The number of ART patients who ever completed at least 6 months of TPT Disaggregates New ART (<15 F, <15 M, 15+F, 15+M); Previous ART (<15 F, <15 M, 15+F, 15+M)
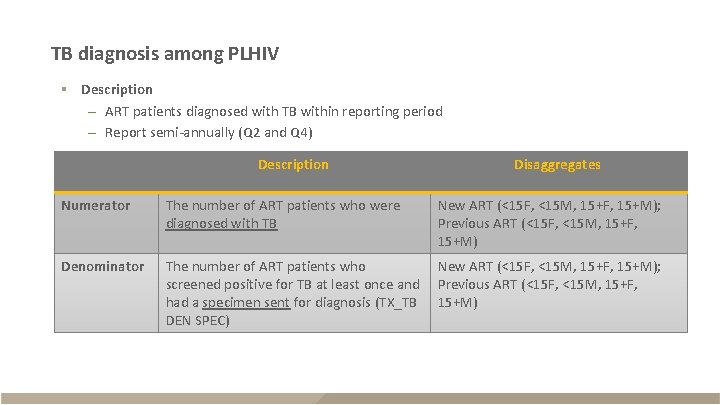
TB diagnosis among PLHIV § Description – ART patients diagnosed with TB within reporting period – Report semi-annually (Q 2 and Q 4) Description Disaggregates Numerator The number of ART patients who were diagnosed with TB New ART (<15 F, <15 M, 15+F, 15+M); Previous ART (<15 F, <15 M, 15+F, 15+M) Denominator The number of ART patients who screened positive for TB at least once and had a specimen sent for diagnosis (TX_TB DEN SPEC) New ART (<15 F, <15 M, 15+F, 15+M); Previous ART (<15 F, <15 M, 15+F, 15+M)

MER Data Entry Screens
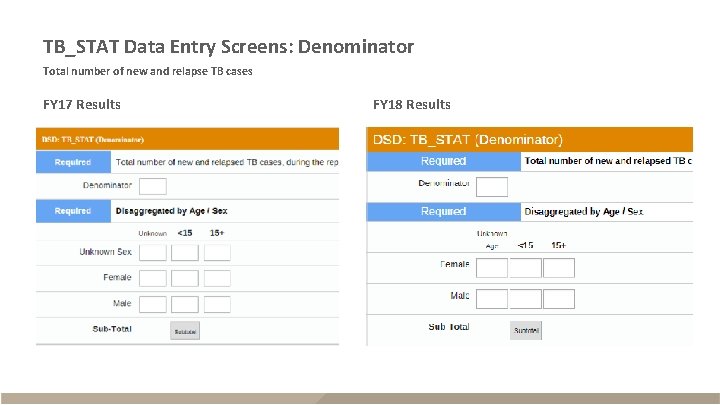
TB_STAT Data Entry Screens: Denominator Total number of new and relapse TB cases FY 17 Results FY 18 Results
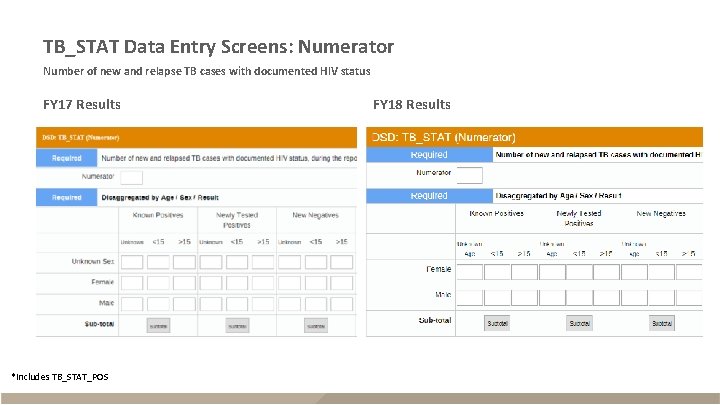
TB_STAT Data Entry Screens: Numerator Number of new and relapse TB cases with documented HIV status FY 17 Results *Includes TB_STAT_POS FY 18 Results
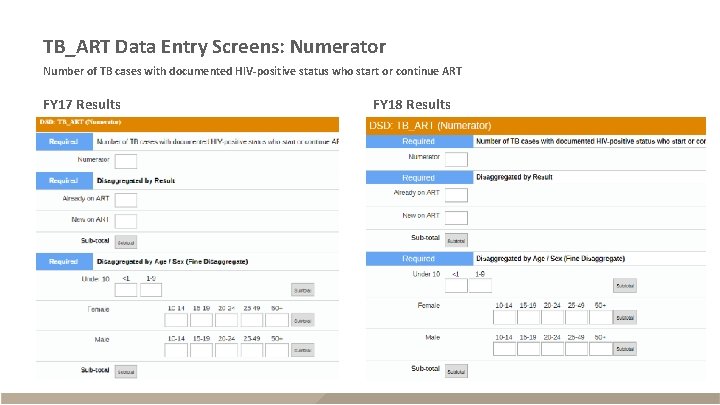
TB_ART Data Entry Screens: Numerator Number of TB cases with documented HIV-positive status who start or continue ART FY 17 Results FY 18 Results
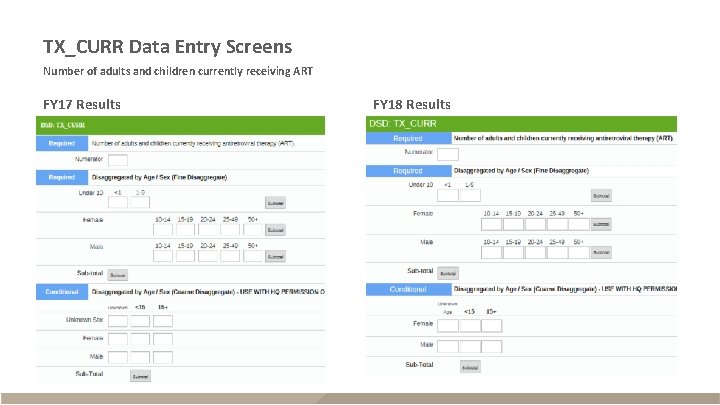
TX_CURR Data Entry Screens Number of adults and children currently receiving ART FY 17 Results FY 18 Results
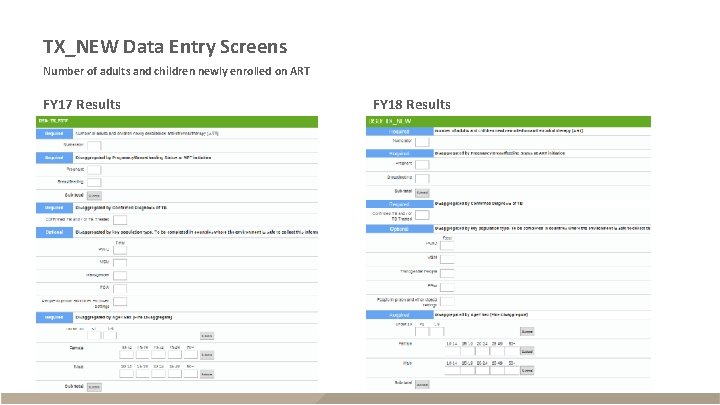
TX_NEW Data Entry Screens Number of adults and children newly enrolled on ART FY 17 Results FY 18 Results
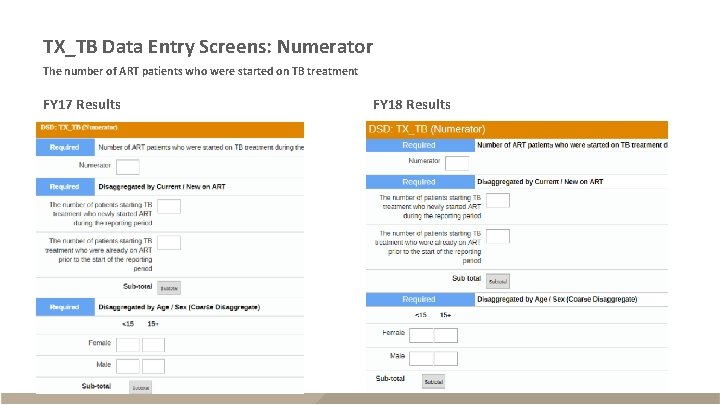
TX_TB Data Entry Screens: Numerator The number of ART patients who were started on TB treatment FY 17 Results FY 18 Results
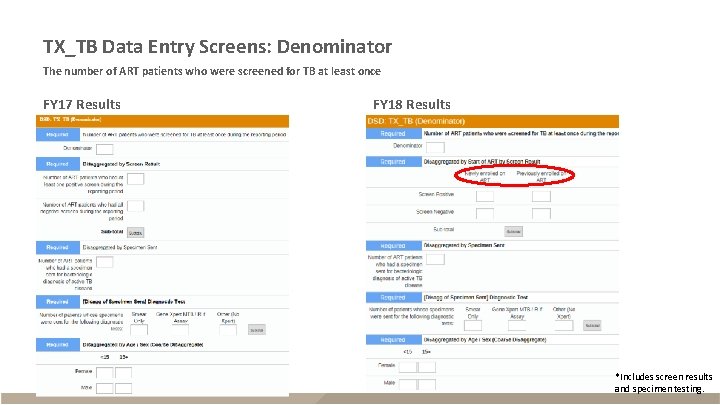
TX_TB Data Entry Screens: Denominator The number of ART patients who were screened for TB at least once FY 17 Results FY 18 Results *Includes screen results and specimen testing.
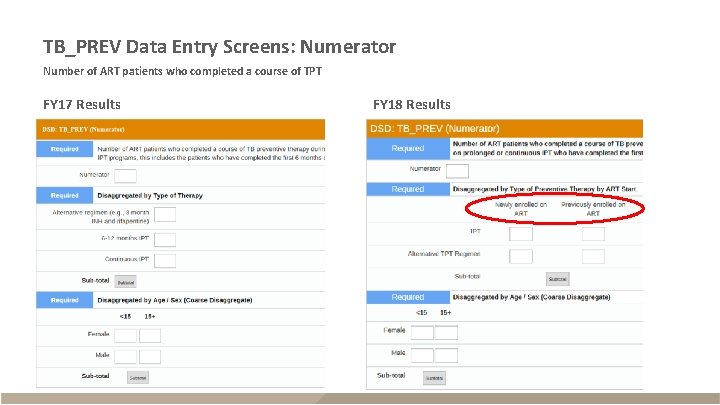
TB_PREV Data Entry Screens: Numerator Number of ART patients who completed a course of TPT FY 17 Results FY 18 Results
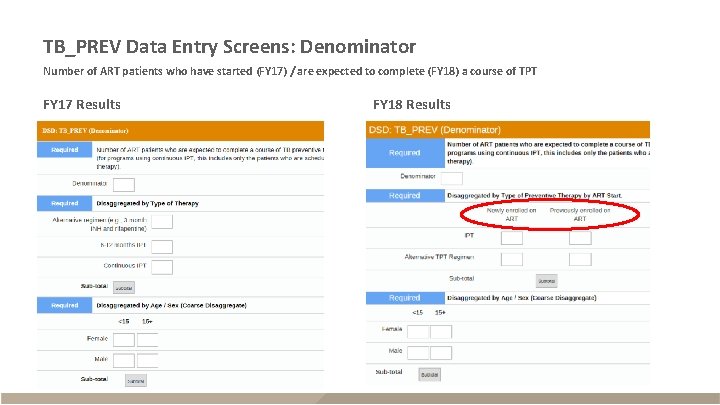
TB_PREV Data Entry Screens: Denominator Number of ART patients who have started (FY 17) / are expected to complete (FY 18) a course of TPT FY 17 Results FY 18 Results

Indicator Guidance Details
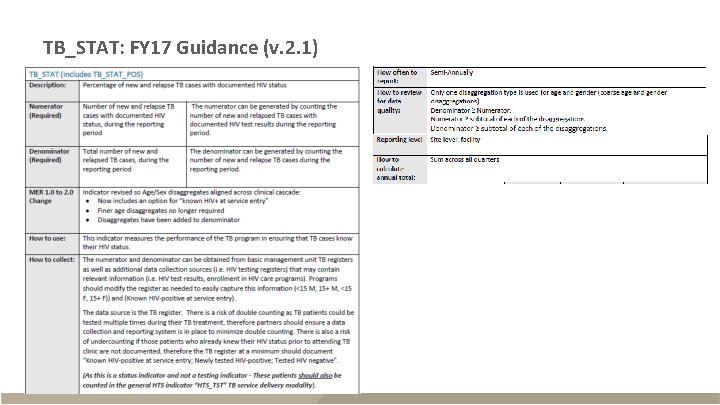
TB_STAT: FY 17 Guidance (v. 2. 1)
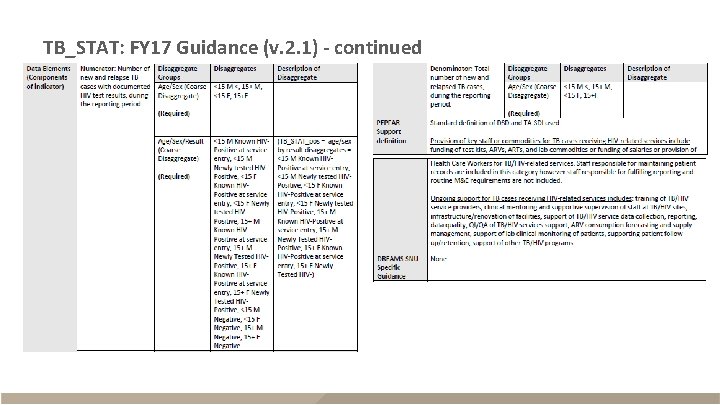
TB_STAT: FY 17 Guidance (v. 2. 1) - continued
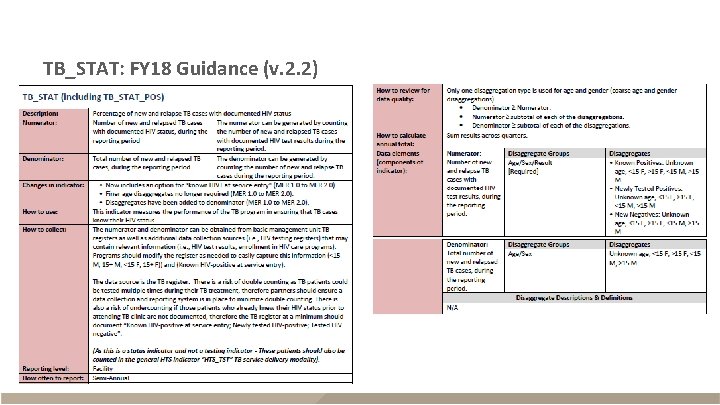
TB_STAT: FY 18 Guidance (v. 2. 2)

TB_STAT: FY 18 Guidance (v. 2. 2) - continued
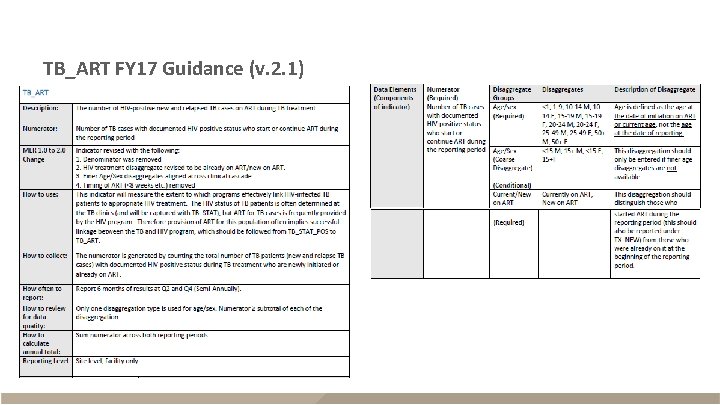
TB_ART FY 17 Guidance (v. 2. 1)

TB_ART FY 17 Guidance (v. 2. 1) - continued
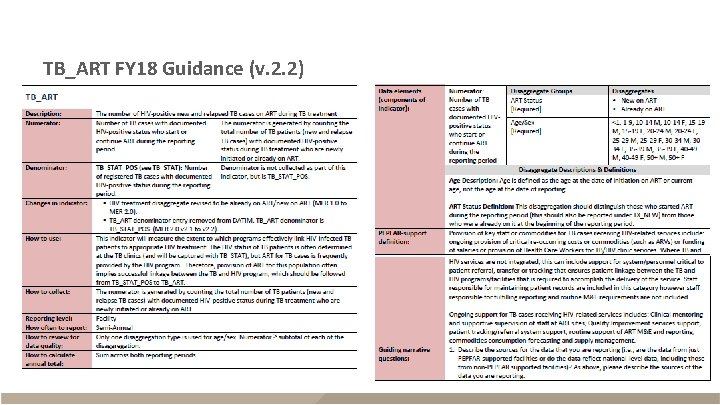
TB_ART FY 18 Guidance (v. 2. 2)

TX_CURR FY 17 Guidance (v. 2. 1)
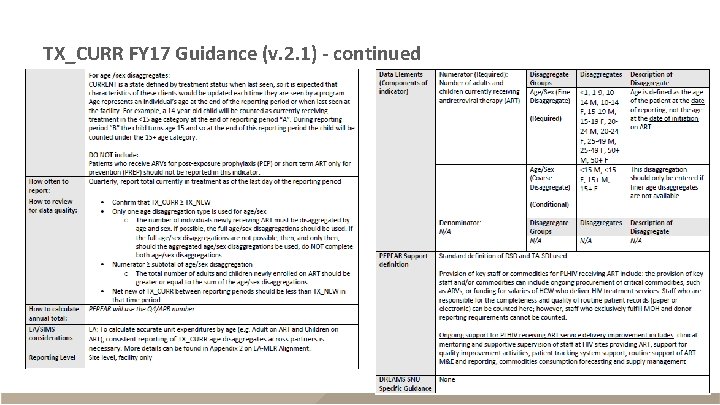
TX_CURR FY 17 Guidance (v. 2. 1) - continued
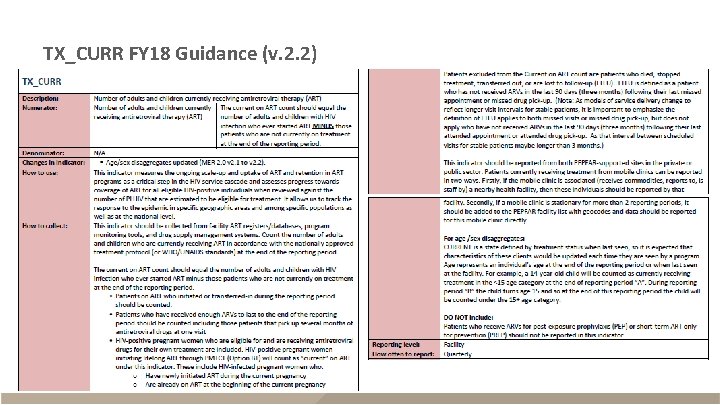
TX_CURR FY 18 Guidance (v. 2. 2)

TX_CURR FY 18 Guidance (v. 2. 2) - continued
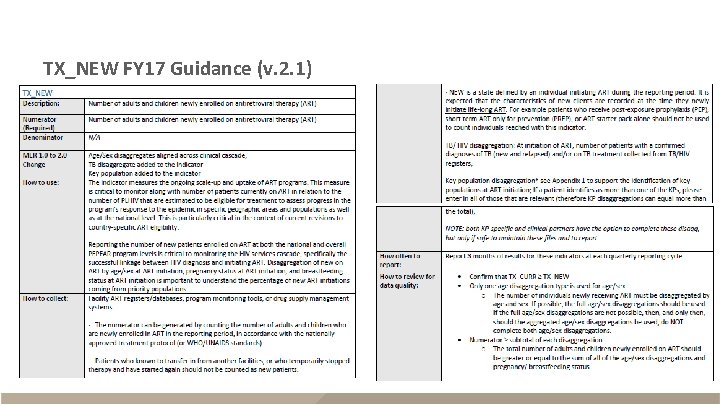
TX_NEW FY 17 Guidance (v. 2. 1)
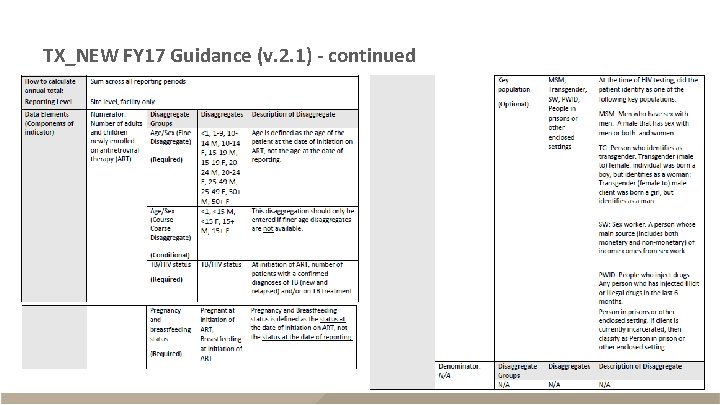
TX_NEW FY 17 Guidance (v. 2. 1) - continued

TX_NEW FY 17 Guidance (v. 2. 1) - continued
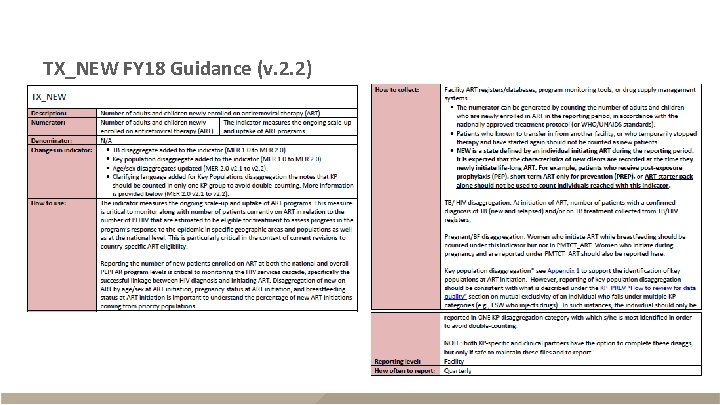
TX_NEW FY 18 Guidance (v. 2. 2)
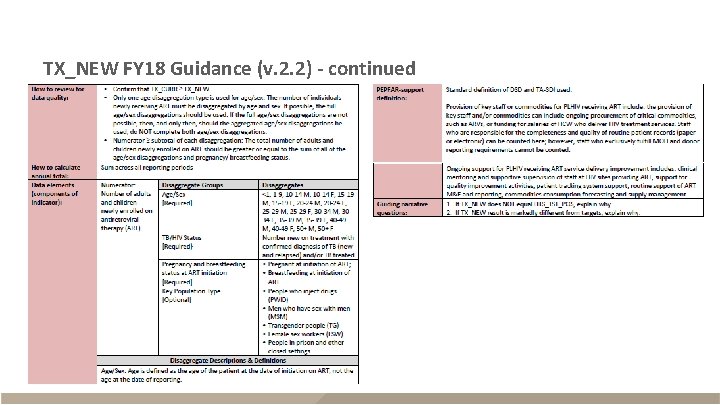
TX_NEW FY 18 Guidance (v. 2. 2) - continued
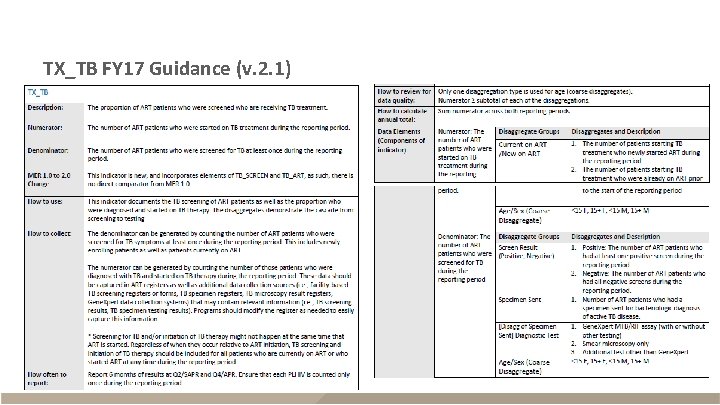
TX_TB FY 17 Guidance (v. 2. 1)

TX_TB FY 17 Guidance (v. 2. 1) - continued
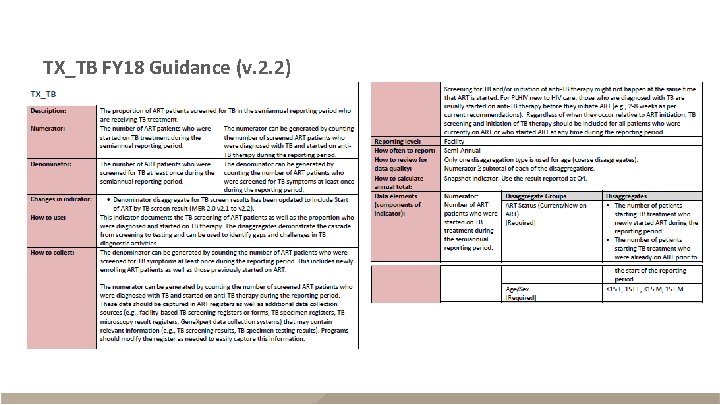
TX_TB FY 18 Guidance (v. 2. 2)

TX_TB FY 18 Guidance (v. 2. 2) - continued

TB_PREV FY 17 Guidance (v. 2. 1)

TB_PREV FY 17 Guidance (v. 2. 1) - continued

TB_PREV FY 18 Guidance (v. 2. 2)
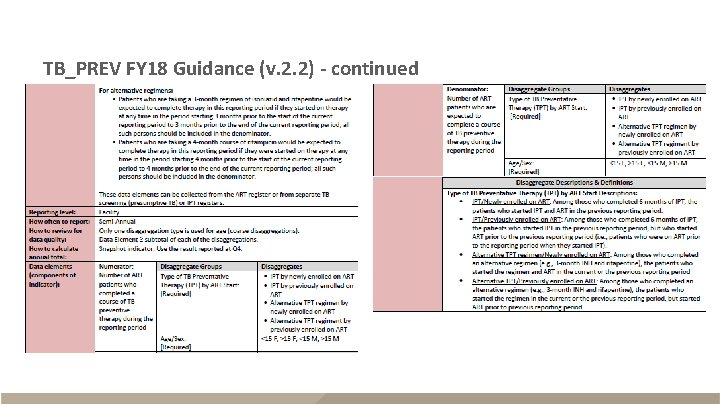
TB_PREV FY 18 Guidance (v. 2. 2) - continued

TB_PREV FY 18 Guidance (v. 2. 2) - continued
WHO LTBI Indicator Guidance
WHO LTBI Indicator Guidance
- Slides: 57