CENTAUR Study cenicriviroc in NASH phase 2 b
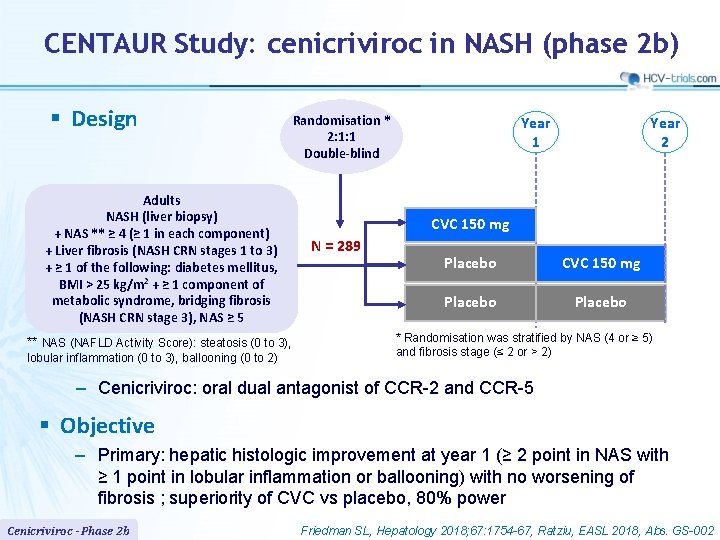
CENTAUR Study: cenicriviroc in NASH (phase 2 b) § Design Adults NASH (liver biopsy) + NAS ** ≥ 4 (≥ 1 in each component) + Liver fibrosis (NASH CRN stages 1 to 3) + ≥ 1 of the following: diabetes mellitus, BMI > 25 kg/m 2 + ≥ 1 component of metabolic syndrome, bridging fibrosis (NASH CRN stage 3), NAS ≥ 5 ** NAS (NAFLD Activity Score): steatosis (0 to 3), lobular inflammation (0 to 3), ballooning (0 to 2) Randomisation * 2: 1: 1 Double-blind Year 1 Year 2 CVC 150 mg N = 289 Placebo CVC 150 mg Placebo * Randomisation was stratified by NAS (4 or ≥ 5) and fibrosis stage (≤ 2 or > 2) ‒ Cenicriviroc: oral dual antagonist of CCR-2 and CCR-5 § Objective – Primary: hepatic histologic improvement at year 1 (≥ 2 point in NAS with ≥ 1 point in lobular inflammation or ballooning) with no worsening of fibrosis ; superiority of CVC vs placebo, 80% power Cenicriviroc - Phase 2 b Friedman SL, Hepatology 2018; 67: 1754 -67, Ratziu, EASL 2018, Abs. GS-002

CENTAUR Study: cenicriviroc in NASH (phase 2 b) Baseline characteristics and disposition CVC, N = 145 Placebo, N = 144 55 54 Female 50% 55% Mean BMI, kg/m 2 33. 6 34. 1 Diabetes mellitus 57% Mean age, years ≥ 3 criteria of metabolic syndrome, % Liver histology NASH CRN fibrosis stage 1 / 2 / 3 NAS Mean ≥ 5, % Mean steatosis Mean lobular inflammation Mean ballooning Discontinued before end of Year 1, N For adverse event Withdrew consent Entered Year 2 of the study, N Early withdrawal, N / for adverse event, N Cenicriviroc - Phase 2 b 44% 72 72 32% / 29% / 39% 33% / 28% / 38% 5. 3 73. 1% 1. 4 2. 4 1. 5 5. 4 75. 0% 1. 4 2. 4 1. 5 20 9 10 18 8 7 121 12 / 5 CVC = 61 2 /1 PCB = 60 2 /0 Friedman SL, Hepatology 2018; 67: 1754 -67, Ratziu, EASL 2018, Abs. GS-002
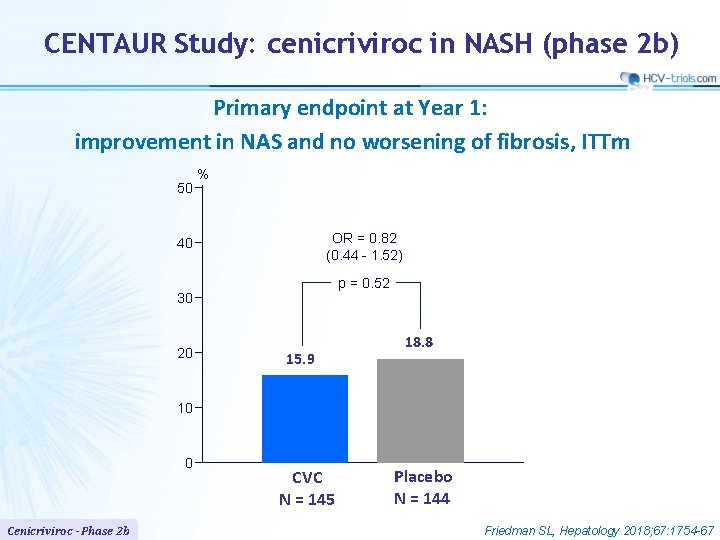
CENTAUR Study: cenicriviroc in NASH (phase 2 b) Primary endpoint at Year 1: improvement in NAS and no worsening of fibrosis, ITTm 50 % OR = 0. 82 (0. 44 - 1. 52) 40 p = 0. 52 30 20 15. 9 18. 8 10 0 Cenicriviroc - Phase 2 b CVC N = 145 Placebo N = 144 Friedman SL, Hepatology 2018; 67: 1754 -67
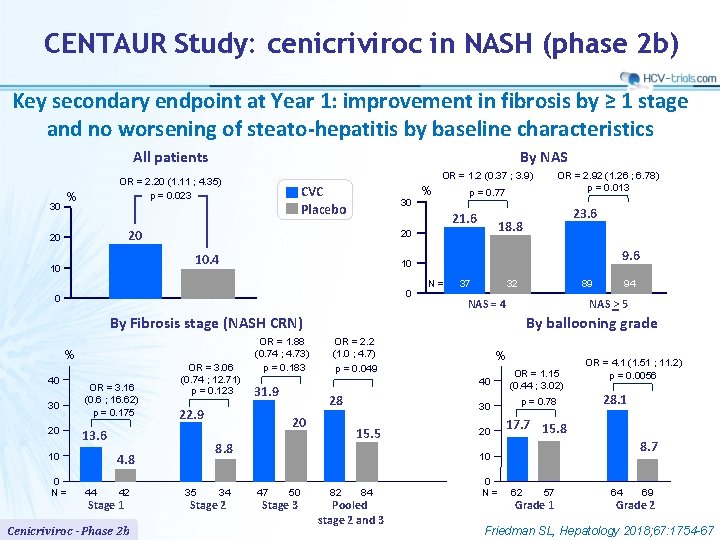
CENTAUR Study: cenicriviroc in NASH (phase 2 b) Key secondary endpoint at Year 1: improvement in fibrosis by ≥ 1 stage and no worsening of steato-hepatitis by baseline characteristics All patients 30 By NAS OR = 1. 2 (0. 37 ; 3. 9) OR = 2. 20 (1. 11 ; 4. 35) p = 0. 023 % CVC Placebo 30 9. 6 10 0 0 N= 37 32 89 NAS = 4 40 30 20 OR = 3. 16 (0. 6 ; 16. 62) p = 0. 175 42 Stage 1 Cenicriviroc - Phase 2 b By ballooning grade OR = 2. 2 (1. 0 ; 4. 7) p = 0. 049 28 20 15. 5 8. 8 44 31. 9 22. 9 13. 6 10 0 N= OR = 3. 06 (0. 74 ; 12. 71) p = 0. 123 OR = 1. 88 (0. 74 ; 4. 73) p = 0. 183 35 34 Stage 2 94 NAS > 5 By Fibrosis stage (NASH CRN) % 23. 6 18. 8 20 10. 4 10 p = 0. 77 21. 6 20 20 % OR = 2. 92 (1. 26 ; 6. 78) p = 0. 013 % 40 OR = 1. 15 (0. 44 ; 3. 02) 30 p = 0. 78 20 17. 7 15. 8 OR = 4. 1 (1. 51 ; 11. 2) p = 0. 0056 28. 1 8. 7 10 47 50 Stage 3 82 84 Pooled stage 2 and 3 0 N= 62 57 Grade 1 64 69 Grade 2 Friedman SL, Hepatology 2018; 67: 1754 -67

CENTAUR Study: cenicriviroc in NASH (phase 2 b) Improvement in fibrosis by ≥ 1 stage and no worsening of steato-hepatitis Favors placebo Overall m. ITT population NAS Fibrosis stage (NASH CRN) 29/126 (23%) 15/126 (12%) NAS = 4 8/37 (22%) 6/32 (19%) NAS > 5 21/89 (24%) 9/94 (10%) Stage 1 6/44 (14%) 2/42 (5%) Stage 2 Stage 3 8/35 (23%) 15/47 (32%) 3/34 (9%) 10/50 (20%) Grade 1 (few ballon cells) 11/62 (18%) 9/57 (16%) Grade 2 (prominent ballooning) 18/64 (28%) 6/69 (9%) Grade 2 (2 -4 foci/200 x) 17/66 (26%) 7/61 (12%) Grade 3 (> 4 foci/200 x) 11/54 (20%) 7/61 (12%) Grade 1 (5 -33 %) 21/89 (24%) 10/81 (12%) Grade 2 (> 33 -66 %) 8/31 (26%) 4/41 (10%) < 30 kg/m² > 30 - < 35 kg/m² > 35 - < 40 kg/m² > 40 kg/m² 7/32 (22%) 8/51 (16%) 6/22 (27%) 7/19 (37%) 3/34 (9%) 5/45 (11%) 4/28 (14%) 3/19 (16%) Present Not present 17/74 (23%) 15/52 (23%) 10/57 (18%) 5/69 (7%) Favors CVC Hepatocellular ballooning Lobular inflammation Steatosis BMI Type 2 diabetes status Cenicriviroc - Phase 2 b Friedman SL, Hepatology 2018; 67: 1754 -67

CENTAUR Study: cenicriviroc in NASH (phase 2 b) Change in liver biopsy at year 1 (IPP population) Placebo N = 123 CVC N = 123 Improved No change Worsened Steatosis 19. 5 74. 0 6. 5 25. 2 61. 8 13. 0 Lobular inflammation 31. 7 43. 9 24. 4 27. 6 49. 6 22. 8 Ballooning 26. 8 54. 5 18. 7 35. 0 50. 4 14. 6 § Biomarkers of inflammation – Marked reductions in circulating biomarkers of systemic inflammation (high-sensitivity C-reactive protein, interleukin-6, interleukin-8, fibrinogen, and IL-1ß) and of monocyte activation (s. CD 14) were observed with CVC (vs placebo). Persistence of systmeic anti-inflamamtory activity at Year 2, without no metabolic disturbances Cenicriviroc - Phase 2 b Friedman SL, Hepatology 2018; 67: 1754 -67, Ratziu, EASL 2018, Abs. GS-002

CENTAUR Study: cenicriviroc in NASH (phase 2 b) § Main outcomes at Year 2 – Improvement in fibrosis (≥ 1 stage) – Improvement in fibrosis (≥ 1 or ≥ 2 stages) and no worsening of NASH – Maintenance of antifibrotic response from Year 1 to Year 2 (groups with 2 years CVC vs 2 years of placebo) – Effects of 2 years of CVC treatment (groups with 2 years CVC vs 2 years of placebo) – Effects of 1 year of CVC treatment (immediate or deferred CVC vs placebo) – Safety and tolerability of CVC vs placebo during Year 2 Cenicriviroc - Phase 2 b Ratziu, EASL 2018, Abs. GS-002
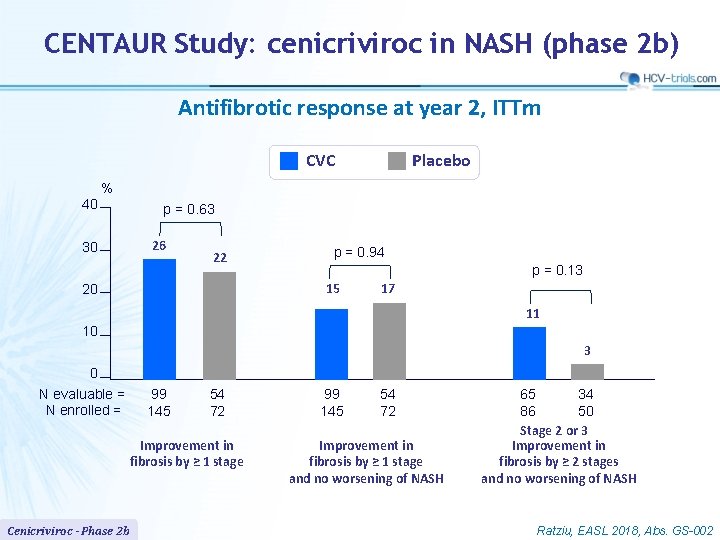
CENTAUR Study: cenicriviroc in NASH (phase 2 b) Antifibrotic response at year 2, ITTm CVC Placebo % 40 p = 0. 63 26 30 22 p = 0. 94 p = 0. 13 15 20 17 11 10 3 0 N evaluable = N enrolled = 99 145 54 72 Improvement in fibrosis by ≥ 1 stage Cenicriviroc - Phase 2 b 99 145 54 72 Improvement in fibrosis by ≥ 1 stage and no worsening of NASH 65 34 86 50 Stage 2 or 3 Improvement in fibrosis by ≥ 2 stages and no worsening of NASH Ratziu, EASL 2018, Abs. GS-002

CENTAUR Study: cenicriviroc in NASH (phase 2 b) Adverse events and laboratory abnormalities, % CVC 1 st year N = 144 Placebo 1 st year N = 145 CVC 2 nd year N = 182 Grade 3 -4 drug-related adverse event 8. 3 6. 3 12. 1 Serious adverse event 11. 1 6. 9 NA Discontinuation for adverse event 6. 3 6. 9 2. 7 Most common adverse events of grade ≥ 2 Fatigue Diarrhea Headache ALT increase ≥ grade 2 2. 8 2. 1 1. 4 NA 0. 7 3. 5 NA NA 7. 1 Grade 3 -4 laboratory abnormalities Fasting serum glucose > 250 mg/d. L ALT > 5 x ULN AST > 5 x ULN Triglycerides grade 3 / grade 4 Gamma-GT grade 3 / grade 4 Creatine kinase grade 3 / grade 4 Uric acid grade 3 / grade 4 Amylase > 2 -5 x ULN Phosphorus < 2 -1 mg/d. L Absolute neutrophil grade 3 / grade 4 11. 9 11. 8 4. 9 3. 5 / 2. 1 5. 6 / 0. 7 4. 2 / 1. 4 6. 3 / 7. 6 4. 2 3. 5 1. 4 / 1. 4 9. 2 11. 8 6. 9 4. 9 / 2. 1 4. 2 / 0. 7 4. 9 / 1. 4 6. 3 / 4. 2 0. 7 1. 4 2. 1 / 0. 7 Cenicriviroc - Phase 2 b NA Friedman SL, Hepatology 2018; 67: 1754 -67, Ratziu, EASL 2018, Abs. GS-002

CENTAUR Study: cenicriviroc in NASH (phase 2 b) § Summary – CVC showed a significant antifibrotic benefit at year 1 and was well tolerated – Although the primary endpoint of the study was not met, the fact that the CENTAUR year 1 study results showed that CVC provided clinically meaningful benefits and resulted in twice as many subjects achieving improvement in fibrosis by 1 stage and no worsening of steato-hepatitis as compared to placebo suggests that the study did, in fact, show proof of concept, warranting phase 3 development of CVC – Year 2 analyses corroborate CVC antifibrotic activity in adults with NADH and liver fibrosis • Effect more pronounced in stage 3 fibrosis Cenicriviroc - Phase 2 b Friedman SL, Hepatology 2018; 67: 1754 -67, Ratziu, EASL 2018, Abs. GS-002
- Slides: 10