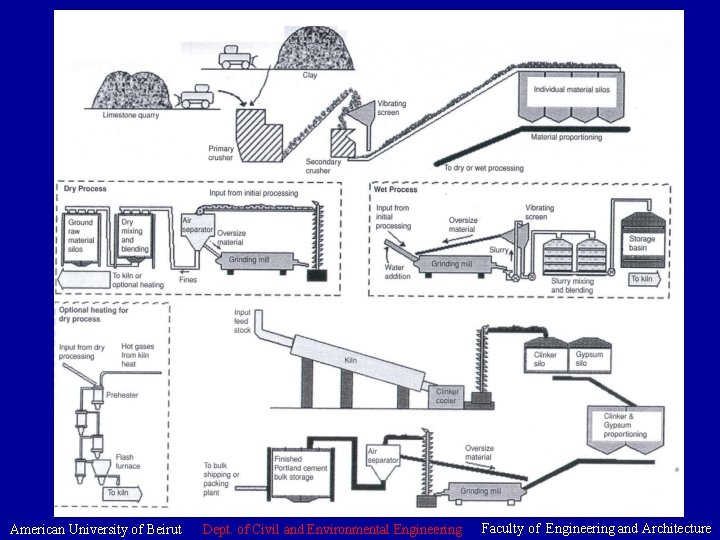
Cement Portland Cement Production Two basic raw ingredients

Cement • Portland Cement Production: Two basic raw ingredients: calcareous material (limestone, chalk, or oyster shells) + argillaceous material (silica and alumina from clay, shale and blast furnace slag) American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

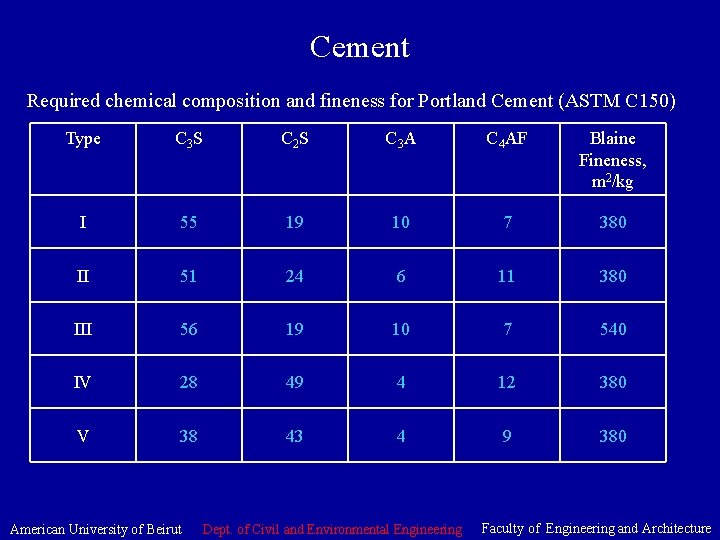
Cement • Chemical composition of Portland Cement: Compound Chemical formula Common formula Usual range. %wt. Tricalcium silicate 3 Ca. O Si. O 2 C 3 S 45 -60 Dicalcium Silicate 2 Ca. O Si. O 2 C 2 S 15 -30 Tricalcium Aluminate 3 Ca. O Al 2 O 3 C 3 A 6 -12 Tetracalcium Aluminoferrite 4 Ca. O Al 2 O 3 Fe 2 O 3 C 4 AF 6 -8 American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • • • Cement Properties and Characteristics Oxides Sources Si. O 2 (silicon dioxide) - cap rock (Sandstone) Ca. O (calcium oxide) - limestone Oxides used to calculate theoretical cementitious compounds: • Al 2 O 3 (aluminum oxide) - clay • C 3 S, C 2 S, C 3 A and C 4 AF • Fe 2 O 3 (ferric oxide) American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

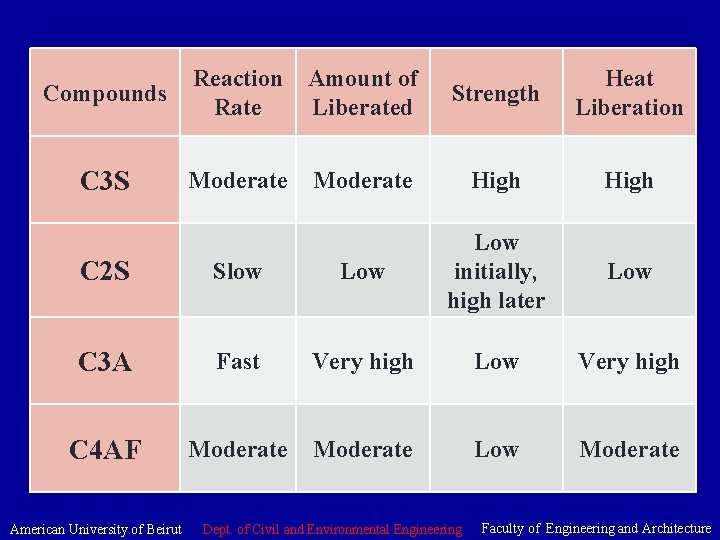
Cement • Compounds • Tricalcium Silicate (C 3 S) hardens rapidly and is largely responsible for initial set and early strength. • In general, the early strength of portland cement concrete is higher with increased percentages of C 3 S. • Dicalcium Silicate (C 2 S) hardens slowly and contributes largely to strength increases at ages beyond 7 days. • Tricalcium Aluminate (C 3 A) liberates a large amount of heat during the first few days of hardening and, together with C 3 S • and C 2 S may somewhat increase the early strength of the hardening cement (this effect being due to the considerable heat of hydration that this compound evolves). It does affect set times. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Tetracalcium Aluminoferrite (C 4 AF) contributes very slightly to strength gain. However, acts as a flux during manufacturing. Contributes to the color effects that makes cement grey. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Hydration of cement • Hydration of cement • When Portland cement is mixed with water its chemical compound constituents undergo a series of chemical reactions that cause it to harden. This chemical reaction with water is called "hydration". Each one of these reactions occurs at a different time and rate. Together, the results of these reactions determine how Portland cement hardens and gains strength. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Hydration of cement • OPC hydration • Hydration starts as soon as the cement and water are mixed. • The rate of hydration and the heat liberated by the reaction of each compound is different. • Each compound produces different products when it hydrates. • American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

• Tricalcium silicate (C 3 S). Hydrates and hardens rapidly and is largely responsible for initial set and early strength. Portland cements with higher percentages of C 3 S will exhibit higher early strength. • Tricalcium aluminate (C 3 A). Hydrates and hardens the quickest. Liberates a large amount of heat almost immediately and contributes somewhat to early strength. Gypsum is added to Portland cement to retard C 3 A hydration. Without gypsum, C 3 A hydration would cause Portland cement to set almost immediately after adding water. • American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

• Dicalcium silicate (C 2 S). Hydrates and hardens slowly and is largely responsible for strength increases beyond one week. • Tetracalcium aluminoferrite (C 4 AF). Hydrates rapidly but contributes very little to strength. Its use allows lower kiln temperatures in Portland cement manufacturing. Most Portland cement color effects are due to C 4 AF. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
Compounds Reaction Rate Amount of Liberated Strength Heat Liberation C 3 S Moderate High Low C 2 S Slow Low initially, high later C 3 A Fast Very high Low Very high C 4 AF Moderate Low Moderate American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
Cement American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Main Types of Portland Cement: Type Name I Normal II Moderate Sulfate resistance Protection against moderate sulfate exposure. 0. 1 -0. 2% wt. Water soluble sulfate in soil or 150 -1500 ppm sulfate in water (seawater). Can be specified with a moderate heat of hydration making it suitable for large piers, heavy abutments, and retaining walls. Also for concrete in warm weather. III High early strength Used for fast track construction, forms need to be removed as soon as possible, or structure need to be put in service as soon as possible. In cold weather reduces time for controlled curing. IV Low Heat of Hydration Used when mass of structure, careful control of heat of hydration, such as large dams. V High Sulfate resistance Protection from severe sulfate exposure 0. 2 -2. 0% wt. Watersoluble sulfate in soils or 1500 -10, 800 ppm sulfate in water. Application General concrete work when the special properties of other types are not needed. Suitable for floors, reinforced structures, pavements, …etc American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
Cement Required chemical composition and fineness for Portland Cement (ASTM C 150) Type C 3 S C 2 S C 3 A C 4 AF Blaine Fineness, m 2/kg I 55 19 10 7 380 II 51 24 6 11 380 III 56 19 10 7 540 IV 28 49 4 12 380 V 38 43 4 9 380 American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
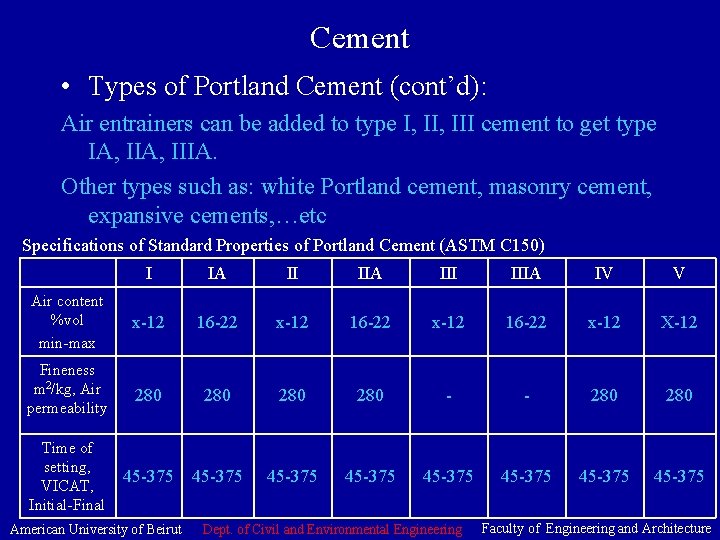
Cement • Types of Portland Cement (cont’d): Air entrainers can be added to type I, III cement to get type IA, IIIA. Other types such as: white Portland cement, masonry cement, expansive cements, …etc Specifications of Standard Properties of Portland Cement (ASTM C 150) I IA II IIA IIIA IV V Air content %vol min-max x-12 16 -22 x-12 X-12 Fineness m 2/kg, Air permeability 280 280 - - 280 Time of setting, VICAT, Initial-Final 45 -375 45 -375 American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Hydration of Portland Cement (cont’d): Primary Chemical reaction during Cement Hydration 2 (3 Ca. O Si. O 2) + 6 H 2 O = 3 Ca. O 2 Si. O 2 3 H 2 O + 3 Ca(OH)2 Tricalcium silicate Water Calcium silicate Hydrates Calcium hydroxide 2 (2 Ca. O Si. O 2) + 4 H 2 O = 3 Ca. O 2 Si. O 2 3 H 2 O + 3 Ca(OH)2 Dicalcium silicate Calcium silicate Hydrates Water 3 Ca. O Al 2 O 3 + 12 H 2 O + Ca(OH)2 Tricalcium aluminate Water Calcium hydroxide 4 Ca. O Al 2 O 3 Fe 2 O 3 + 10 H 2 O + 2 Ca(OH)2 Tetracalcium aluminoferrite Water Calcium hydroxide 3 Ca. O Al 2 O 3 + 10 H 2 O + Ca. SO 4 2 H 2 O Tricalcium aluminate water American University of Beirut Gypsum Calcium hydroxide = 3 Ca. O AL 2 O 3 Ca(OH)2 12 H 2 O Calcium aluminate hydrate = 6 Ca. O Al 2 O 3 Fe 2 O 3 12 H 2 O Calcium aliminoferrite hydrate = 3 Ca. O Al 2 O 3 Ca. SO 4 12 H 2 O Calcium monosulfoaluminate hydrate Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

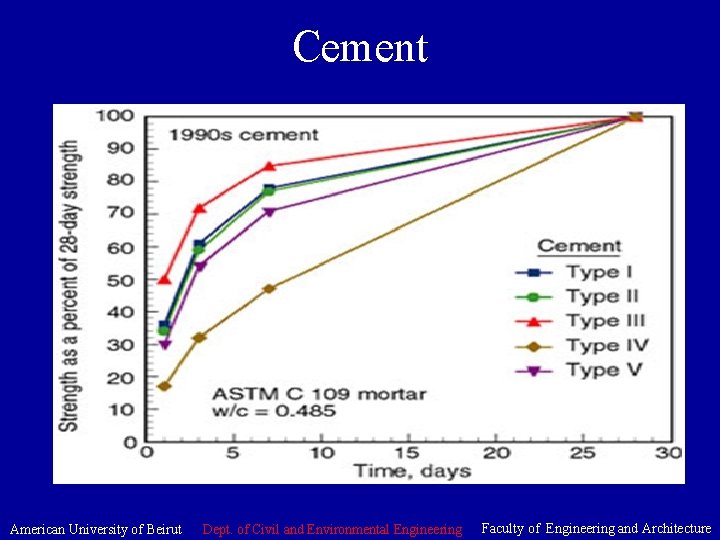
Cement • Fineness of Portland Cement: Fineness is important. Since hydration starts at the surface of cement particles. The larger the surface area the faster the hydration. i. e finer material results in faster strength development. Finesses is related to productivity cost and detrimental to concrete quality. Blaine air permeability Test (ASTM C 204) or % passing the 0. 045 mm sieve (No. 325) (ASTM C 430) • Hydration of Portland Cement: Hydration is a chemical reaction between cement particles and water. The solidification of the cement paste relates to the time of setting. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Properties of Hydrated Cement: Quality control issue – Setting: stiffening of the cement paste or change from plastic to solid state. • Setting = Final - Initial setting levels • Vicat Test (ASTM C 191) or Gillmore (ASTM C 206) – Soundness: ability of cement paste to retain its volume after setting. • Le Châtelier Test – Compressive Strength: measured by preparing 50 mm cubes and subject them to compression (ASTM C 109). Minimum values of compressive strength in ASTM C 150 American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Water-Cement Ratio: (Abrams 1918) w/c affects the concrete quality (strength and other desirable properties). Hydration requires 0. 22 – 0. 25 kg water per 1 kg cement. Concrete require excess moisture, beyond hydration, for workability. As w/c increases. Concrete porosity and permeability increase, the strength decreases. Low w/c increases the resistance to weathering, provides good bond between concrete layers, and between concrete and steel reinforcement, and limits volume change due to wetting and drying. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
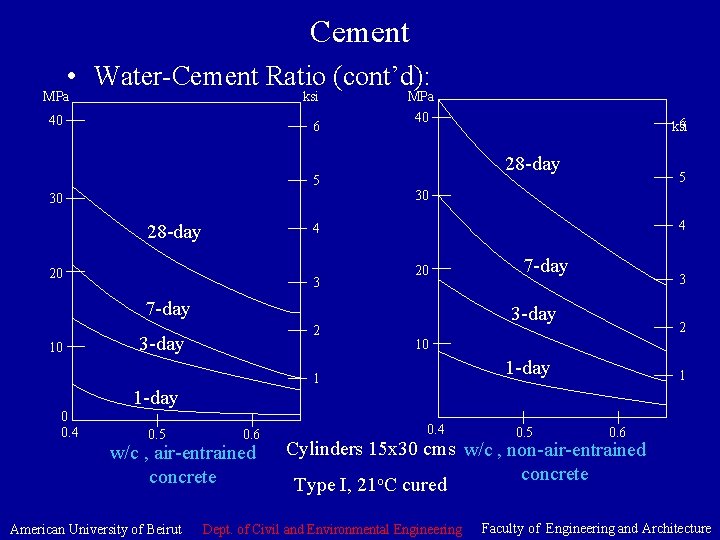
Cement • Water-Cement Ratio (cont’d): MPa ksi 40 6 5 30 28 -day MPa 40 28 -day 30 4 3 20 7 -day 0 0. 4 2 3 -day 0. 6 w/c , air-entrained concrete American University of Beirut 7 -day 3 3 -day 2 10 1 -day 1 1 -day 0. 5 5 4 20 10 6 ksi 0. 4 0. 5 1 0. 6 Cylinders 15 x 30 cms w/c , non-air-entrained concrete Type I, 21 o. C cured Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
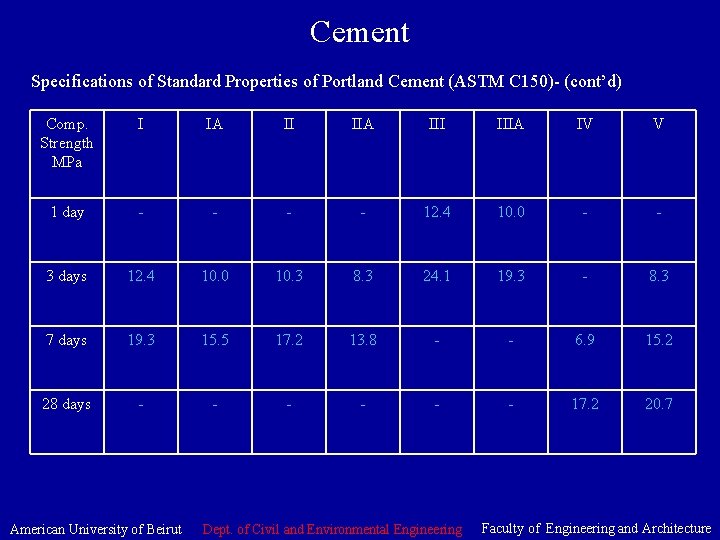
Cement Specifications of Standard Properties of Portland Cement (ASTM C 150)- (cont’d) Comp. Strength MPa I IA II IIA IIIA IV V 1 day - - 12. 4 10. 0 - - 3 days 12. 4 10. 0 10. 3 8. 3 24. 1 19. 3 - 8. 3 7 days 19. 3 15. 5 17. 2 13. 8 - - 6. 9 15. 2 28 days - - - 17. 2 20. 7 American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Admixtures for Concrete: – – – – Air entrainers Water reducers High-range water reducers—Superplasticizers Retarders Accelerators Fine minerals Specialty admixtures American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Admixtures for Concrete (cont’d): – Air entrainers: produce tiny air bubbles in hardened concrete to provide space for water to expand upon freezing. ( salts of wood, synthetic…) – Water Reducers: minimize the amount of water required for workability. Increase the mobility of cement particles in the plastic mix. (no effect on slump, increase strength, decrease cost using less cement) – Superplasticizers: increase the flow of fresh concrete or reduce the amount of water required. – Retarders: delay the initial set of concrete ( offset the effect of hot weather, allow for long distances placement, provide time for special finishes) American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture

Cement • Admixtures for Concrete (cont’d): – Accelerators: develop early strength of concrete at a faster rate than that developed in normal concrete. The ultimate strength, however, is the same as in normal concrete. (reduce curing time, increase rate of strength, plug leaks under hydraulic pressure efficiently). – Fine minerals: (waste from a production process) improve the characteristics of both plastic and hardened concrete ( cementitious, pozzolanic, …) – Specialty Admixtures: workability agents, corrosion inhibitators, damp proofing agents, permeability reducing agents, pumping aids, bonding agents, coloring agents, …etc. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
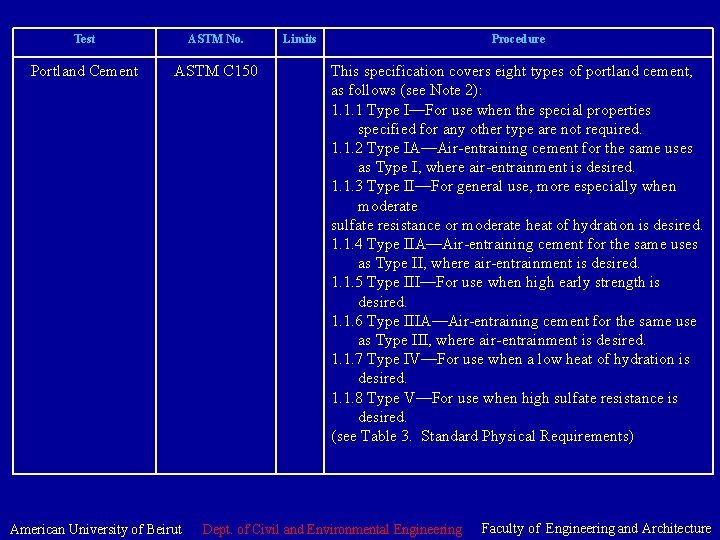
Test ASTM No. Portland Cement ASTM C 150 American University of Beirut Limits Procedure This specification covers eight types of portland cement, as follows (see Note 2): 1. 1. 1 Type I—For use when the special properties specified for any other type are not required. 1. 1. 2 Type IA—Air-entraining cement for the same uses as Type I, where air-entrainment is desired. 1. 1. 3 Type II—For general use, more especially when moderate sulfate resistance or moderate heat of hydration is desired. 1. 1. 4 Type IIA—Air-entraining cement for the same uses as Type II, where air-entrainment is desired. 1. 1. 5 Type III—For use when high early strength is desired. 1. 1. 6 Type IIIA—Air-entraining cement for the same use as Type III, where air-entrainment is desired. 1. 1. 7 Type IV—For use when a low heat of hydration is desired. 1. 1. 8 Type V—For use when high sulfate resistance is desired. (see Table 3. Standard Physical Requirements) Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
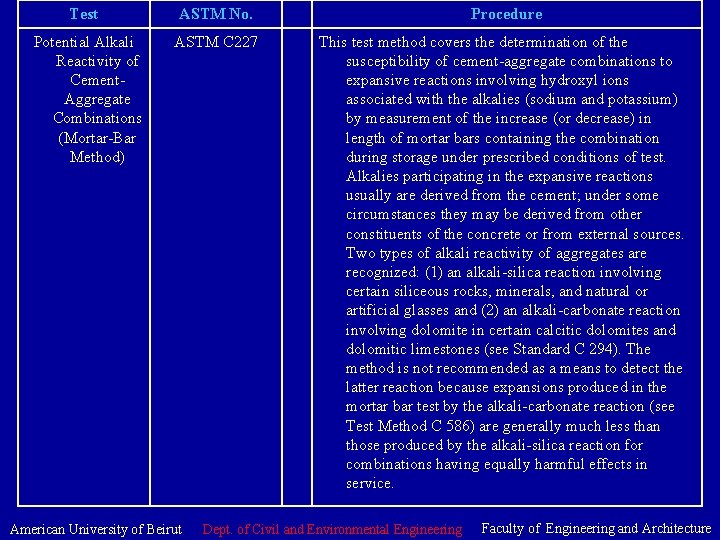
Test ASTM No. Procedure Potential Alkali Reactivity of Cement. Aggregate Combinations (Mortar-Bar Method) ASTM C 227 This test method covers the determination of the susceptibility of cement-aggregate combinations to expansive reactions involving hydroxyl ions associated with the alkalies (sodium and potassium) by measurement of the increase (or decrease) in length of mortar bars containing the combination during storage under prescribed conditions of test. Alkalies participating in the expansive reactions usually are derived from the cement; under some circumstances they may be derived from other constituents of the concrete or from external sources. Two types of alkali reactivity of aggregates are recognized: (1) an alkali-silica reaction involving certain siliceous rocks, minerals, and natural or artificial glasses and (2) an alkali-carbonate reaction involving dolomite in certain calcitic dolomites and dolomitic limestones (see Standard C 294). The method is not recommended as a means to detect the latter reaction because expansions produced in the mortar bar test by the alkali-carbonate reaction (see Test Method C 586) are generally much less than those produced by the alkali-silica reaction for combinations having equally harmful effects in service. American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
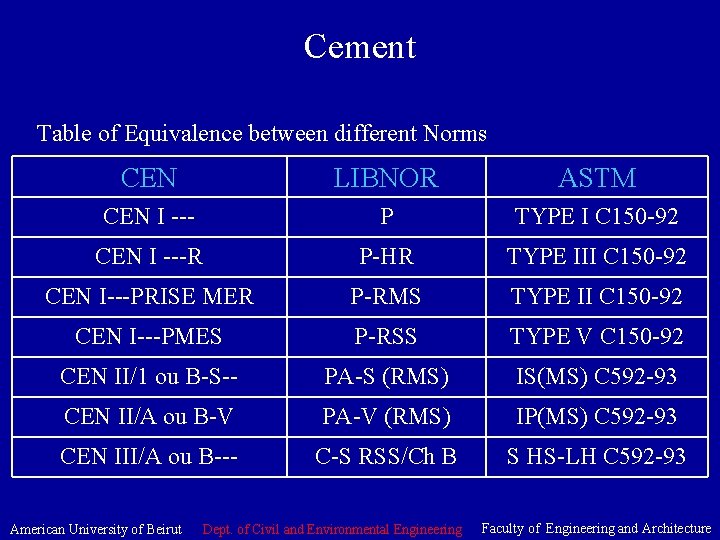
Cement Table of Equivalence between different Norms CEN LIBNOR ASTM CEN I --- P TYPE I C 150 -92 CEN I ---R P-HR TYPE III C 150 -92 CEN I---PRISE MER P-RMS TYPE II C 150 -92 CEN I---PMES P-RSS TYPE V C 150 -92 CEN II/1 ou B-S-- PA-S (RMS) IS(MS) C 592 -93 CEN II/A ou B-V PA-V (RMS) IP(MS) C 592 -93 CEN III/A ou B--- C-S RSS/Ch B S HS-LH C 592 -93 American University of Beirut Dept. of Civil and Environmental Engineering Faculty of Engineering and Architecture
- Slides: 27